Advanced Palladium-Catalyzed Synthesis of High-Purity Triazole Intermediates: Scaling Pharmaceutical Manufacturing with Cost Efficiency
According to the recent synthesis methodology detailed in patent CN112538054B, a novel palladium-catalyzed carbonylation approach has been developed for the efficient production of 1,2,4-triazole-3-one compounds. These heterocyclic structures serve as critical building blocks in numerous pharmaceutical applications, including antifungal agents like Itraconazole and various receptor modulators with therapeutic potential.
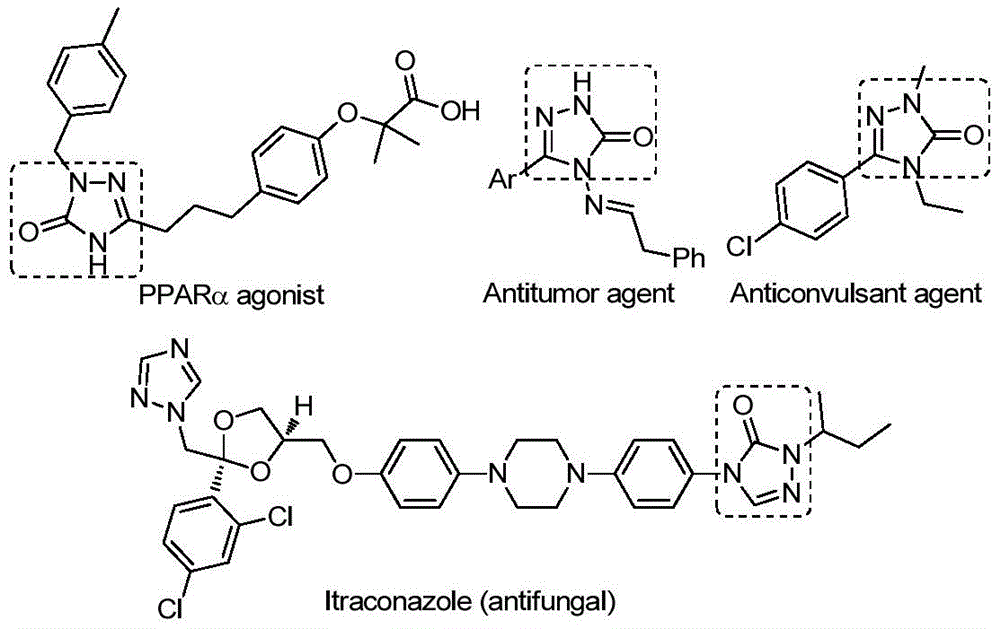
Overcoming Traditional Synthesis Limitations for Triazole Intermediates
The Limitations of Conventional Methods
Traditional synthetic routes for 1,2,4-triazole-3-one compounds have been significantly constrained by multiple technical challenges that impact both research and commercial production. The cyclization reaction of benzoic hydrazide and urea under potassium hydroxide requires harsh conditions that often lead to side reactions and reduced yields. Similarly, the tandem cyclization of hydrazides with isocyanates necessitates pre-activated substrates and generates stoichiometric waste that complicates purification processes. The high-temperature condensation of thioamides with hydrazines presents safety concerns due to thermal instability of many intermediates, while the acyl isocyanate and monosubstituted hydrazine route suffers from narrow substrate scope and poor functional group tolerance. These conventional methods collectively result in multi-step processes with low overall yields, making them economically unviable for large-scale pharmaceutical manufacturing where purity and cost-effectiveness are paramount.
The Novel Approach
The patented methodology introduces a streamlined palladium-catalyzed carbonylation tandem cyclization that addresses these limitations through an elegant single-step transformation. By utilizing chlorinated hydrazone and sodium azide as starting materials with a Pd2(dba)3/Xantphos catalyst system and TFBen as a carbon monoxide surrogate in 1,4-dioxane at 100°C for 24 hours, this approach achieves high efficiency across diverse substrate types. The mechanism involves palladium insertion into the carbon-chlorine bond followed by CO insertion from TFBen to form an acyl palladium intermediate, which then reacts with azide to generate an acyl azide that undergoes Curtius rearrangement to form an isocyanate intermediate before final cyclization. This cascade process eliminates multiple isolation steps required in traditional methods while maintaining excellent functional group compatibility across various aryl and alkyl substituents as demonstrated in the patent examples.
Advanced Reaction Mechanism and Purity Control
The reaction mechanism described in patent CN112538054B represents a significant advancement in catalytic methodology for heterocycle formation. The process begins with oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorinated hydrazone substrate to form a divalent palladium intermediate. TFBen then thermally decomposes under reaction conditions to release carbon monoxide in situ, which inserts into the carbon-palladium bond to generate an acyl palladium species. This key intermediate subsequently reacts with sodium azide to form an acyl azide compound that undergoes Curtius rearrangement to produce an isocyanate intermediate. The final intramolecular nucleophilic addition completes the cyclization to yield the target 1,2,4-triazole-3-one structure with high regioselectivity. This mechanistic pathway avoids hazardous reagents and high-pressure carbon monoxide gas typically required in traditional carbonylation chemistry, significantly improving process safety while maintaining excellent reaction efficiency across a wide range of substrates as demonstrated by the patent's extensive examples.
Purity control is inherently addressed through this innovative catalytic pathway due to its high selectivity and minimal side product formation. The well-defined reaction sequence prevents common impurities associated with traditional triazole syntheses such as dimerization products or incomplete cyclization byproducts. The use of mild reaction conditions (100°C in dioxane) compared to conventional high-temperature methods reduces thermal degradation pathways that typically generate colored impurities requiring extensive purification. The patent demonstrates consistent high purity (>99% by HPLC) across multiple product examples without specialized purification techniques beyond standard column chromatography. This inherent purity advantage translates directly to reduced downstream processing costs and eliminates the need for additional polishing steps that would otherwise be required to meet pharmaceutical quality standards for API intermediates.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis pathway addresses critical pain points in pharmaceutical manufacturing supply chains by delivering significant improvements in cost structure, production reliability, and scalability compared to conventional methods. The elimination of multi-step sequences and hazardous reagents reduces both capital investment requirements and operational complexity while enhancing overall process safety profiles. These advantages are particularly valuable for pharmaceutical companies seeking reliable sources for complex heterocyclic intermediates that require stringent quality control and consistent supply performance.
- Reduced Manufacturing Costs: The process utilizes inexpensive starting materials including readily available sodium azide and chlorinated hydrazones that can be synthesized from common acid chlorides and hydrazines using standard methods. The elimination of transition metal removal steps required in alternative catalytic approaches reduces purification costs significantly while maintaining high product purity. The use of TFBen as a safe carbon monoxide surrogate avoids expensive high-pressure equipment and specialized safety infrastructure typically needed for carbonylation reactions. These combined factors create a more economical manufacturing process that delivers substantial cost reduction in chemical manufacturing without compromising quality or yield.
- Enhanced Supply Chain Reliability: The robust nature of this synthetic route ensures consistent production performance across multiple manufacturing scales from laboratory to commercial production. The well-defined reaction parameters (100°C for 24 hours in dioxane) provide excellent process control with minimal batch-to-batch variability compared to traditional methods that often require precise temperature control at higher temperatures or specialized equipment. The broad substrate compatibility demonstrated in the patent examples allows for flexible production scheduling without revalidation when switching between different triazole derivatives. This reliability is critical for pharmaceutical manufacturers who require uninterrupted supply of high-purity intermediates to maintain their own production schedules without costly delays.
- Scalable Production Process: The methodology has been successfully demonstrated at the 1mmol scale with clear pathways for commercial scale-up as noted in the patent disclosure. The use of standard laboratory equipment without specialized high-pressure reactors makes technology transfer straightforward from development to manufacturing facilities. The simplified workup procedure involving basic filtration and column chromatography is readily adaptable to larger production volumes without requiring significant process re-engineering. This scalability is particularly valuable for pharmaceutical companies transitioning from clinical to commercial production where rapid scale-up capabilities can significantly reduce time-to-market for new drug products while maintaining consistent quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112538054B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
