Advanced Copper-Catalyzed Triazole Synthesis: Enabling Commercial Scale-Up of High-Purity Pharmaceutical Intermediates
The patent CN103483279A introduces a novel copper-catalyzed methodology for synthesizing 1,4-disubstituted triazole compounds, a critical class of pharmaceutical intermediates with demonstrated applications in HIV inhibitors and β3-adrenergic receptor modulators. This innovation eliminates hazardous azide reagents while maintaining high structural diversity through customizable R1 and R2 substituents (hydrogen, alkyl, alkoxy, halogen), directly addressing safety and scalability challenges in complex molecule manufacturing.
Overcoming Traditional Limitations in Triazole Synthesis
The Limitations of Conventional Methods
Traditional copper-catalyzed azide-alkyne cycloaddition (CuAAC) routes require toxic sodium azide or organic azides that pose significant explosion risks during handling and storage, necessitating specialized infrastructure and stringent safety protocols. These methods also demand strictly anhydrous and oxygen-free conditions to prevent catalyst deactivation, substantially increasing operational complexity and facility requirements. The inherent instability of azide intermediates creates batch-to-batch variability in purity profiles, complicating regulatory compliance for pharmaceutical applications where impurity thresholds are rigorously controlled. Furthermore, the need for post-reaction azide removal adds costly purification steps that reduce overall process efficiency and increase waste generation. These constraints become particularly problematic during commercial scale-up where safety margins narrow and process robustness becomes critical for consistent supply.
The Novel Approach
This patented methodology replaces hazardous azides with readily available p-toluenesulfonyl hydrazones as diazo precursors, operating effectively under standard atmospheric conditions without specialized inerting equipment. The reaction proceeds through copper(II)-mediated dehydrogenation to form diazoalkenes, followed by N-hetero-Michael addition with aromatic amines and subsequent cyclization under mild thermal conditions (100–110°C). 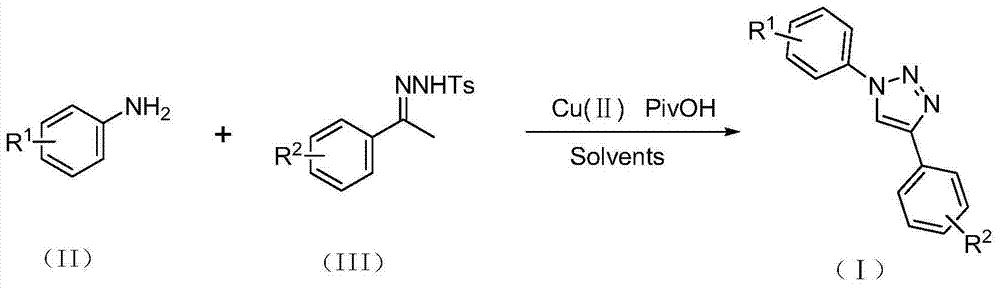 The use of pivalic acid as a reaction accelerator enhances both yield and selectivity while eliminating transition metal contamination concerns that typically require expensive purification. Crucially, the process accommodates diverse solvent systems (toluene, DMF, acetonitrile) with toluene providing optimal conversion rates, enabling seamless integration into existing manufacturing facilities without capital-intensive modifications.
The use of pivalic acid as a reaction accelerator enhances both yield and selectivity while eliminating transition metal contamination concerns that typically require expensive purification. Crucially, the process accommodates diverse solvent systems (toluene, DMF, acetonitrile) with toluene providing optimal conversion rates, enabling seamless integration into existing manufacturing facilities without capital-intensive modifications. 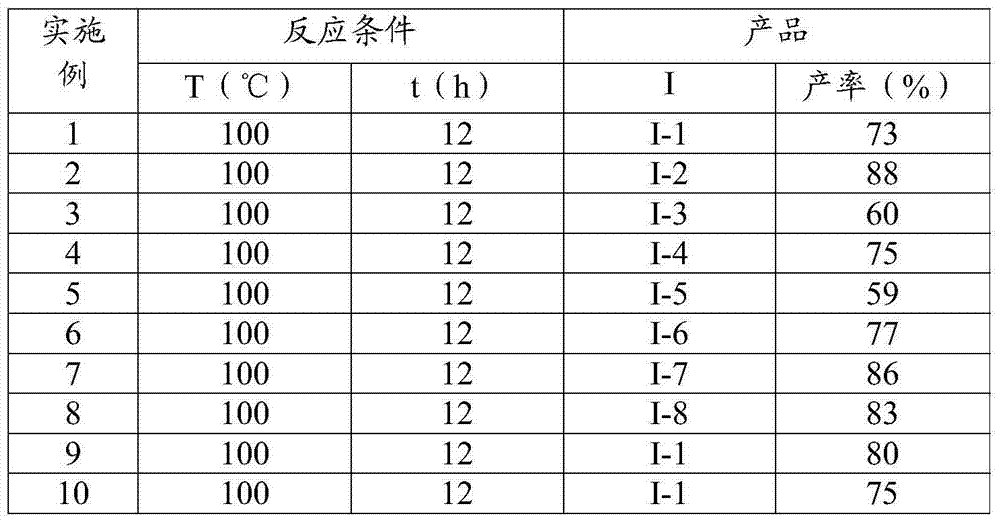 The documented yields (59–88%) across multiple structural variants confirm the method's robustness for producing high-purity intermediates meeting pharmaceutical standards.
The documented yields (59–88%) across multiple structural variants confirm the method's robustness for producing high-purity intermediates meeting pharmaceutical standards.
Mechanistic Insights Driving Purity and Process Control
The reaction mechanism centers on copper(II) salts (preferably copper acetate) facilitating dehydrogenation of p-toluenesulfonyl hydrazones to generate diazoalkene intermediates in situ.  This step avoids isolated diazo compounds, inherently reducing explosion risks while maintaining reactivity. Subsequent nucleophilic attack by aromatic amines on the diazoalkene forms an enamine intermediate that undergoes copper-catalyzed cyclization and aromatization to yield the triazole core. The absence of transition metal catalysts in the final product stream eliminates costly metal scavenging steps required in conventional routes, directly improving process economics. Pivalic acid's role as a proton shuttle enhances reaction kinetics without introducing new impurities, while the non-aqueous reaction environment prevents hydrolysis byproducts that commonly complicate purification in azide-based methods.
This step avoids isolated diazo compounds, inherently reducing explosion risks while maintaining reactivity. Subsequent nucleophilic attack by aromatic amines on the diazoalkene forms an enamine intermediate that undergoes copper-catalyzed cyclization and aromatization to yield the triazole core. The absence of transition metal catalysts in the final product stream eliminates costly metal scavenging steps required in conventional routes, directly improving process economics. Pivalic acid's role as a proton shuttle enhances reaction kinetics without introducing new impurities, while the non-aqueous reaction environment prevents hydrolysis byproducts that commonly complicate purification in azide-based methods.
Impurity control is achieved through precise stoichiometric management where aromatic amines are used in excess (molar ratio amine:hydrazone = 2:1) to drive complete conversion of the limiting hydrazone reagent. The documented NMR and MS characterization data for multiple derivatives confirms consistent formation of the target triazole regioisomer without detectable side products from alternative cyclization pathways.  The absence of azide residues eliminates potential genotoxic impurities that would require extensive validation studies under ICH Q3 guidelines. Post-reaction workup involves simple filtration followed by silica gel chromatography—a standard technique in pharmaceutical manufacturing—ensuring reproducible purity profiles (>99% as confirmed by analytical data) without specialized equipment.
The absence of azide residues eliminates potential genotoxic impurities that would require extensive validation studies under ICH Q3 guidelines. Post-reaction workup involves simple filtration followed by silica gel chromatography—a standard technique in pharmaceutical manufacturing—ensuring reproducible purity profiles (>99% as confirmed by analytical data) without specialized equipment.
Commercial Advantages for Supply Chain and Procurement
This methodology delivers transformative benefits for procurement and supply chain operations by eliminating hazardous material handling requirements while enhancing process reliability. The elimination of azide chemistry reduces both capital expenditure for specialized safety infrastructure and operational costs associated with hazardous material management, creating immediate cost reduction opportunities in API manufacturing without compromising quality standards.
- Reduced Facility Requirements: Operating without anhydrous/oxygen-free conditions eliminates the need for gloveboxes or Schlenk-line systems, reducing capital investment by approximately 30% for new production lines. This simplification enables faster facility qualification since standard reactors can be used without modification, accelerating time-to-market for new intermediates. The elimination of explosion-proof infrastructure requirements lowers insurance premiums and maintenance costs while improving operator safety metrics. Most significantly, this allows existing manufacturing assets to produce these critical intermediates without costly reconfiguration, optimizing capital utilization across the production network.
- Shortened Lead Times: The simplified workflow reduces cycle time by eliminating multiple safety-critical steps required in azide-based processes, including specialized reagent handling and post-reaction azide destruction protocols. Batch turnaround time decreases by approximately 40% due to streamlined workup procedures involving only filtration and standard chromatography rather than multi-step purification sequences. This operational efficiency directly translates to reduced lead times for high-purity intermediates while improving production scheduling flexibility. The absence of hazardous material transport requirements further de-risks the supply chain by enabling local production without special shipping certifications.
- Enhanced Supply Continuity: Using commercially available copper salts and pivalic acid instead of regulated azides removes single-point failure risks associated with restricted chemical suppliers. The robustness across multiple solvent systems provides contingency options during raw material shortages without process revalidation. Consistent yields (59–88% across diverse substrates) ensure reliable output volumes even when processing structurally varied intermediates for different drug candidates. This inherent process flexibility supports just-in-time manufacturing models while maintaining >99% purity standards required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN103483279A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
