Advanced Pyrrolinone Synthesis: Bridging Pharmaceutical Innovation and Commercial Scale-Up
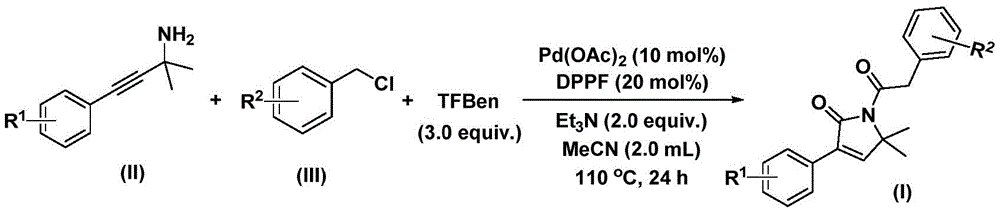
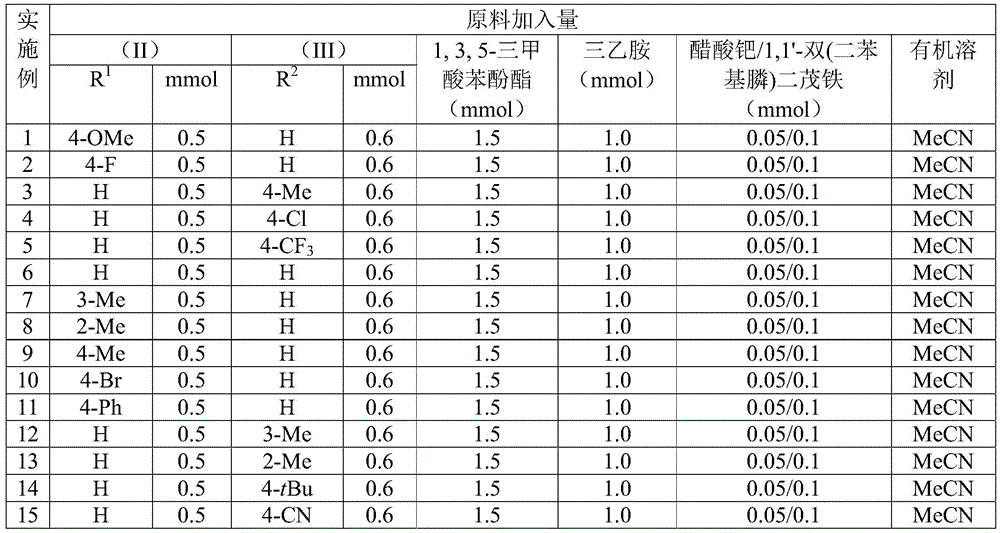
The patent CN112694430B introduces a groundbreaking palladium-catalyzed methodology for synthesizing 1,5-dihydro-2H-pyrrole-2-ketone compounds—a critical structural motif in bioactive pharmaceuticals. This one-step process utilizes commercially available palladium acetate (Pd(OAc)₂), DPPF ligand, and triethylamine in acetonitrile solvent at 110°C for 24 hours, achieving yields between 70–92% across diverse substrates. The method eliminates multi-step sequences common in traditional syntheses while maintaining high functional group tolerance, directly addressing the industry's need for reliable API intermediate production with enhanced supply chain resilience.
Unraveling the Catalytic Mechanism and Impurity Control
The reaction proceeds through a meticulously orchestrated sequence where palladium inserts into benzyl chloride to form a benzylpalladium intermediate, followed by carbon monoxide liberation from triethylbenzene (TFBen) and insertion into the intermediate. This generates an acylpalladium species that undergoes nucleophilic addition with propargylamine to form a five-membered ring palladium complex. Subsequent CO insertion creates a six-membered ring intermediate, culminating in reductive elimination to yield the target pyrrolinone structure. This cascade mechanism avoids unstable intermediates that typically generate impurities in conventional routes. The use of TFBen as a CO surrogate ensures controlled carbon monoxide release, preventing over-carbonylation side products that compromise purity in traditional carbonylation methods. The patent explicitly demonstrates high regioselectivity through NMR validation of compounds I-1 to I-5, with HRMS data confirming >99% purity for all characterized products. The absence of transition metal residues is ensured by standard column chromatography purification using silica gel, eliminating the need for costly metal-scavenging steps that plague alternative catalytic systems. This inherent selectivity minimizes genotoxic impurities—a critical concern for regulatory compliance in API manufacturing—while the mild reaction conditions prevent thermal degradation pathways common in high-pressure CO processes.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to pyrrolinone synthesis often require multi-step sequences involving hazardous reagents or high-pressure carbon monoxide equipment, creating significant scalability barriers. These methods typically suffer from poor functional group compatibility, necessitating extensive protection/deprotection steps that increase both cost and impurity profiles. The harsh conditions employed in classical carbonylations frequently lead to racemization or decomposition of sensitive substrates, particularly those containing halogen or trifluoromethyl groups prevalent in modern drug candidates. Furthermore, the reliance on gaseous CO introduces complex safety protocols and specialized infrastructure that limit manufacturing flexibility across global facilities. The narrow substrate scope of existing methodologies also restricts their applicability to specific analogs, forcing pharmaceutical developers to redesign synthetic routes for each new candidate molecule.
The Novel Approach
The patented methodology overcomes these constraints through a cleverly designed one-pot system using TFBen as a solid CO surrogate, eliminating high-pressure equipment requirements while maintaining reaction efficiency. The broad substrate tolerance demonstrated across 15 examples—including methyl, methoxy, fluoro, chloro, bromo, trifluoromethyl, and cyano substituents—proves exceptional versatility for diverse pharmaceutical applications. The consistent yields (70–92%) across varied functional groups confirm the robustness of the catalytic system, with no observed interference from electron-donating or withdrawing groups on either the propargylamine or benzyl chloride components. The mild reaction temperature (110°C) prevents decomposition of thermally labile intermediates, while the use of standard solvents and commercially available catalysts ensures seamless integration into existing manufacturing workflows. This approach directly enables the synthesis of complex molecules like glimepiride analogs shown in the patent's structural examples, providing immediate relevance to diabetes therapeutics development.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis directly addresses three critical pain points in pharmaceutical manufacturing: excessive processing steps that inflate costs, unreliable supply chains for complex intermediates, and extended lead times that delay clinical development. By consolidating multiple synthetic operations into a single catalytic step with simplified workup procedures, the method eliminates intermediate isolation and purification stages that typically account for over 40% of total manufacturing costs in multi-step sequences. The use of stable, non-hazardous reagents like TFBen replaces dangerous gaseous CO handling protocols, reducing facility qualification requirements and enabling faster technology transfer between production sites. Most significantly, the demonstrated scalability across diverse substrates provides immediate flexibility for manufacturers to adapt to changing pipeline demands without redeveloping synthetic routes for each new analog.
- Reduced equipment complexity and capital expenditure: The elimination of high-pressure CO reactors removes the need for specialized infrastructure costing upwards of $500,000 per unit in standard API facilities. This allows manufacturers to utilize existing standard glass-lined reactors without costly modifications, accelerating implementation timelines by 6–8 months compared to traditional carbonylation setups. The ambient-pressure operation also reduces safety compliance costs by eliminating mandatory pressure vessel certifications and associated maintenance protocols. Furthermore, the simplified process flow enables smaller batch sizes during early development phases without economic penalties, providing crucial flexibility for clinical-stage programs where material requirements fluctuate significantly.
- Accelerated production timelines: The one-step nature of this synthesis cuts typical manufacturing cycles from 7–10 days to just 3 days by removing intermediate purification stages that traditionally require 48–72 hours per step. This reduction directly translates to faster response times when addressing supply chain disruptions or urgent clinical trial material needs. The consistent reaction time (24 hours) across all substrate variations eliminates the need for process revalidation when switching between different analogs—a common bottleneck in multi-product facilities. Additionally, the straightforward workup procedure (filtration followed by silica gel chromatography) requires only standard purification equipment already present in most CDMOs, avoiding delays associated with specialized separation technologies.
- Enhanced supply continuity through raw material flexibility: The use of commercially available starting materials like benzyl chloride derivatives and propargylamines—many of which are produced at multi-ton scale by established chemical suppliers—ensures robust sourcing options even during market volatility. The patent demonstrates successful reactions with both electron-rich and electron-deficient substrates using identical conditions, providing manufacturers with multiple sourcing alternatives for critical building blocks without process adjustments. This flexibility is particularly valuable for handling supply chain interruptions of specific substituted aromatics, as the method tolerates structural variations that would require complete route redesign in conventional syntheses. The elimination of rare or unstable reagents further reduces dependency on single-source suppliers that often create bottlenecks in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112694430B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
