Scalable Electrochemical C-3 Aminomethylation of Imidazo[1,2-a]pyridines for Commercial API Production
Introduction to Advanced Electrochemical Functionalization
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways to access complex heterocyclic scaffolds, particularly those with proven biological relevance. Patent CN115369430A introduces a groundbreaking synthetic methodology for the preparation of 3-aminomethyl imidazo[1,2-a]pyridine derivatives, a core structure found in numerous bioactive molecules including potential anticancer agents. This technology leverages electrochemical oxidation to achieve direct C-3 aminomethylation, bypassing the limitations of traditional thermal or photochemical methods. By utilizing electricity as a clean oxidant, this process aligns perfectly with modern green chemistry principles while maintaining high synthetic efficiency. The general structure of these valuable intermediates is depicted below, showcasing the versatility of the substitution patterns allowed at the R1, R2, R3, and R4 positions.
![General chemical structure of 3-aminomethyl imidazo[1,2-a]pyridine derivatives showing variable substitution sites](/insights/img/3-aminomethyl-imidazo-pyridine-electrochemical-synthesis-pharma-supplier-20260305005702-01.png)
For R&D directors and process chemists, the ability to functionalize the electron-rich C-3 position of the imidazo[1,2-a]pyridine ring system without harsh reagents represents a significant strategic advantage. The patent details a robust protocol that tolerates a wide array of functional groups, enabling the rapid generation of diverse libraries for drug discovery programs. Furthermore, the method's compatibility with complex amine substrates suggests immediate applicability in the late-stage functionalization of existing drug candidates, thereby accelerating lead optimization cycles. As a reliable pharmaceutical intermediate supplier, understanding such innovative technologies is crucial for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the C-3 position on imidazo[1,2-a]pyridine scaffolds has relied heavily on classical organic transformations that often suffer from significant drawbacks. Traditional approaches frequently necessitate the use of stoichiometric amounts of strong chemical oxidants or expensive transition metal catalysts, which not only increase the raw material costs but also generate substantial amounts of hazardous waste. Many existing protocols require rigorous exclusion of moisture and oxygen, demanding inert gas protection and specialized equipment that complicates scale-up efforts. Additionally, some prior art methods involve high-temperature conditions or multi-step sequences involving unstable intermediates like N-substituted glycine derivatives, leading to lower overall atom economy and reduced process safety. These factors collectively hinder the commercial viability of producing high-purity pharmaceutical intermediates on a large scale.
The Novel Approach
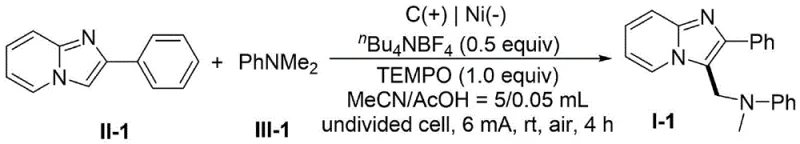
In stark contrast, the electrochemical strategy disclosed in CN115369430A offers a paradigm shift by replacing chemical oxidants with electrons. This novel approach utilizes an undivided cell setup where a constant direct current drives the oxidative coupling between the imidazo[1,2-a]pyridine derivative and an N-methyl amine. The reaction proceeds under remarkably mild conditions, typically at room temperature and open to air, eliminating the need for inert atmospheres. The general reaction scheme illustrates the simplicity of combining the heterocyclic substrate with the amine source in the presence of a supporting electrolyte and a mediator like TEMPO.
![General electrochemical reaction scheme showing the coupling of imidazo[1,2-a]pyridine with N-methyl amines](/insights/img/3-aminomethyl-imidazo-pyridine-electrochemical-synthesis-pharma-supplier-20260305005702-02.webp)
This method achieves exceptional yields, reported up to 98% in optimized examples, while producing minimal by-products. The absence of heavy metal residues simplifies downstream purification, a critical factor for meeting stringent regulatory standards in API manufacturing. By streamlining the synthetic route to a single pot operation with easily removable electrolytes, this technology significantly reduces the environmental footprint and operational complexity. For procurement managers, this translates to a more predictable and cost-effective supply chain, as the reliance on volatile specialty reagents is minimized. The scalability of electrochemical processes is well-documented, making this an ideal candidate for transitioning from gram-scale discovery to kilogram or ton-scale commercial production.
Mechanistic Insights into Electrochemical C-H Activation
The success of this transformation lies in the precise control of electron transfer at the electrode surface. Mechanistically, the anodic oxidation likely generates a reactive radical species from the N-methyl amine or facilitates the formation of an iminium ion intermediate via the TEMPO mediator. This electrophilic species then attacks the electron-rich C-3 position of the imidazo[1,2-a]pyridine ring, which is inherently activated towards electrophilic substitution due to its fused heterocyclic nature. The use of additives such as acetic acid plays a crucial role in proton management and stabilizing intermediates, ensuring the reaction proceeds smoothly without over-oxidation or decomposition of the sensitive heterocyclic core. The cathode, often made of nickel or graphite, balances the circuit by facilitating a reduction half-reaction, typically hydrogen evolution, which does not interfere with the main oxidative pathway.
From an impurity control perspective, the selectivity of this electrochemical method is superior to many thermal alternatives. The mild potential applied prevents the degradation of sensitive functional groups such as esters, halides, or ethers, which might otherwise decompose under harsh chemical oxidation conditions. The patent data indicates high tolerance for various substituents on both the pyridine ring and the amine component, suggesting that side reactions like polymerization or over-alkylation are effectively suppressed. This high level of chemoselectivity ensures that the final product profile is clean, reducing the burden on purification teams and increasing the overall throughput of the manufacturing process. Understanding these mechanistic nuances allows process engineers to fine-tune parameters like current density and electrolyte concentration to maximize efficiency for specific substrates.
How to Synthesize 3-Aminomethyl Imidazo[1,2-a]pyridine Efficiently
Implementing this electrochemical protocol requires careful attention to cell configuration and reagent ratios, although the procedure is fundamentally straightforward. The patent provides specific embodiments, such as Example 1, which serves as a benchmark for optimizing reaction conditions. In this representative case, a graphite anode and a nickel cathode are employed with tetrabutylammonium tetrafluoroborate as the electrolyte. The specific reaction setup for this high-yielding transformation is shown below, highlighting the precise stoichiometry and conditions used to achieve 98% yield.
- Combine the imidazo[1,2-a]pyridine substrate, N-methyl amine compound, electrolyte (e.g., nBu4NBF4), and additives (e.g., TEMPO, acetic acid) in an organic solvent like acetonitrile.
- Install a graphite anode and a nickel cathode into the undivided cell and apply a constant direct current (e.g., 6 mA) at room temperature under air atmosphere.
- Upon completion, remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target 3-aminomethyl derivative.
The detailed standardized synthesis steps for scaling this reaction are provided in the guide below, ensuring reproducibility across different laboratory or plant settings. Operators should ensure that the electrode surface area is sufficient to maintain the specified current density and that the stirring rate is adequate to prevent mass transfer limitations. The workup procedure involves simple solvent removal followed by standard silica gel chromatography, making it accessible for facilities without specialized extraction equipment. By adhering to these guidelines, manufacturers can reliably produce high-quality intermediates suitable for subsequent coupling reactions or biological testing.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this electrochemical synthesis route offers tangible benefits that extend beyond the laboratory bench, directly impacting the bottom line and supply chain resilience. For procurement managers, the elimination of expensive stoichiometric oxidants and precious metal catalysts represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The raw materials required, such as graphite electrodes and common ammonium salts, are commodity chemicals with stable pricing and widespread availability, reducing the risk of supply disruptions. Furthermore, the simplified workflow reduces labor hours and energy consumption associated with heating or cooling, contributing to a leaner operational model.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the replacement of costly chemical reagents with electricity. By avoiding the purchase of specialized oxidants and the subsequent waste disposal costs associated with them, the overall cost of goods sold (COGS) is substantially lowered. Additionally, the high yields reported minimize raw material waste, ensuring that every gram of starting material is converted into valuable product. The absence of heavy metals also removes the need for expensive scavenging resins or complex purification steps, further driving down processing costs and enhancing the overall profitability of the synthesis.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of robust, non-hazardous reagents that do not require special storage conditions or transport permits. The reaction's tolerance to air and moisture means that production does not depend on a continuous supply of nitrogen or argon, removing a potential bottleneck in facility operations. This robustness allows for more flexible scheduling and reduces the likelihood of batch failures due to environmental leaks. Consequently, suppliers can offer more consistent lead times and guarantee continuity of supply even during periods of market volatility or logistical constraints.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, allowing for a seamless transition from pilot plant to full commercial production without significant process redesign. The green nature of this synthesis aligns with increasingly strict environmental regulations, as it generates less hazardous waste and consumes fewer resources. This compliance reduces the regulatory burden and potential fines associated with waste management, making it a future-proof choice for sustainable manufacturing. Companies adopting this technology can market their products as 'green' or 'sustainably sourced,' adding value in markets where environmental stewardship is a key purchasing criterion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this electrochemical synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for potential partners and technical stakeholders. Understanding these details helps in assessing the feasibility of integrating this technology into existing production lines or research pipelines.
Q: What are the key advantages of this electrochemical method over traditional oxidation?
A: This method utilizes electricity as a traceless oxidant, eliminating the need for stoichiometric chemical oxidants or expensive transition metal catalysts. It operates under mild conditions (room temperature, air atmosphere) and achieves yields up to 98%.
Q: Is inert gas protection required for this synthesis?
A: No, the patent explicitly states that the reaction proceeds efficiently under air atmosphere without the need for inert gas protection, significantly simplifying the operational requirements for scale-up.
Q: What is the substrate scope for the amine component?
A: The method demonstrates broad tolerance, successfully reacting with various N-methyl secondary amines, N-methyl-N-alkyl amines, and even complex drug molecules like Betahistine and Tamoxifen derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminomethyl Imidazo[1,2-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern drug development. Our team of expert chemists is well-versed in adapting such cutting-edge patent technologies to meet the rigorous demands of commercial API production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from concept to market. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of 3-aminomethyl imidazo[1,2-a]pyridine meets the highest international standards.
We invite you to collaborate with us to leverage this efficient synthetic route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your timeline and optimize your budget. Let us be your partner in delivering high-quality, cost-effective pharmaceutical intermediates for the global market.
