Advanced Synthetic Route for Gastrodin Intermediate: Scalable Manufacturing & Cost Efficiency
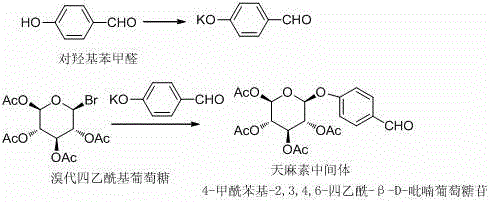
The pharmaceutical industry continuously seeks robust synthetic pathways for neuroprotective agents, particularly for gastrodin and its precursors. A pivotal advancement in this domain is detailed in patent CN111518148A, which discloses a highly efficient method for synthesizing the critical gastrodin intermediate, 4-formylphenyl-2,3,4,6-tetraacetyl-beta-D-glucopyranoside. This compound serves as a fundamental building block in the production of gastrodin, a widely used therapeutic agent for treating cerebrovascular disorders and neurological conditions. The disclosed technology represents a significant departure from legacy manufacturing protocols, offering a streamlined approach that integrates phase transfer catalysis with mild Lewis base activation. For R&D directors and procurement specialists evaluating supply chain resilience, this patent provides a blueprint for achieving superior purity profiles while drastically simplifying the operational complexity associated with glycosylation reactions. By leveraging this intellectual property, manufacturers can transition towards more sustainable and economically viable production models that meet the rigorous demands of modern regulatory environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-formylphenyl-2,3,4,6-tetraacetyl-beta-D-glucopyranoside has been plagued by significant inefficiencies that hinder large-scale commercial viability. Traditional methodologies predominantly rely on the use of potassium iodide (KI) and sodium hydroxide (NaOH) in acetone systems, which frequently result in suboptimal conversion rates and disappointing isolated yields, typically hovering between a mere 20% to 30%. These harsh alkaline conditions often promote undesirable side reactions, including the hydrolysis of acetyl protecting groups, leading to complex impurity profiles that necessitate extensive and costly purification steps. Furthermore, alternative catalytic systems employing boron trifluoride diethyl etherate have introduced their own set of logistical challenges, characterized by excessive solvent consumption and arduous post-reaction workups that generate substantial chemical waste. These legacy processes not only inflate the cost of goods sold (COGS) through material loss but also pose significant environmental compliance risks due to the generation of halogenated and heavy metal-containing waste streams, making them increasingly untenable in the context of modern green chemistry mandates.
The Novel Approach
In stark contrast to these outdated techniques, the innovative route described in the patent introduces a sophisticated phase transfer catalytic system that fundamentally alters the reaction landscape. By utilizing a combination of ammonium chloride, a phase transfer catalyst (such as tetrabutylammonium bromide), and mild Lewis bases like potassium carbonate or sodium carbonate, the process achieves a remarkable enhancement in reaction efficiency. The reaction is conducted in a biphasic or mixed solvent system involving purified water and acetone, allowing for precise temperature control between 10°C and 30°C. This mild thermal window prevents the degradation of sensitive functional groups while facilitating the nucleophilic attack of the phenoxide species on the glycosyl donor. The result is a dramatic improvement in weight yield, consistently exceeding 48% and reaching up to 54.7% in optimized examples, effectively doubling the output compared to conventional methods. This approach not only simplifies the operational workflow by eliminating the need for hazardous reagents like KI but also ensures a cleaner reaction profile that is inherently easier to scale.
Mechanistic Insights into Phase Transfer Catalyzed Glycosylation
The core of this technological breakthrough lies in the intricate interplay between the phase transfer catalyst and the Lewis base within the aqueous-organic interface. In this mechanism, the Lewis base, such as potassium hypochlorite or carbonate, acts to deprotonate the p-hydroxybenzaldehyde, generating a reactive phenoxide anion. However, in a traditional heterogeneous system, this anion would remain largely sequestered in the aqueous phase, inaccessible to the organic-soluble bromotetraacetyl glucose. The phase transfer catalyst, typically a quaternary ammonium salt, bridges this gap by transporting the phenoxide anion into the organic phase as a lipophilic ion pair. Once in the organic milieu, the phenoxide executes a nucleophilic substitution at the anomeric carbon of the sugar moiety. The presence of ammonium chloride further stabilizes the reaction environment, potentially acting as a buffer to moderate the basicity and prevent the saponification of the acetate esters on the glucose ring. This delicate balance ensures that the glycosidic bond is formed with high beta-selectivity while preserving the integrity of the acetyl protecting groups, which is critical for the downstream synthesis of high-purity gastrodin.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over strong base catalysis. In traditional NaOH-mediated reactions, the high concentration of hydroxide ions often leads to partial deacetylation, resulting in mono-, di-, or tri-acetylated byproducts that are structurally similar to the target molecule and difficult to separate via crystallization. The novel method's use of weaker Lewis bases and buffered conditions significantly suppresses these hydrolytic side reactions. Furthermore, the avoidance of iodide ions eliminates the risk of iodine-related impurities or oxidation byproducts that can complicate the purification process. The reaction mixture, upon completion, can be simply concentrated under reduced pressure until turbidity appears, followed by cooling and filtration. This straightforward workup suggests that the crude product possesses a high degree of purity, which is subsequently refined through a single ethanol recrystallization step to achieve HPLC purities greater than 99.6%, demonstrating the robustness of the impurity rejection mechanism inherent in this catalytic system.
How to Synthesize 4-Formylphenyl-2,3,4,6-tetraacetyl-beta-D-glucopyranoside Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the phase transfer components and the thermal profile of the addition sequence. The process begins with the preparation of the aqueous phase containing the aldehyde and catalytic species, followed by the controlled addition of the glycosyl donor dissolved in acetone. Maintaining the temperature within the specified 10-30°C range during the exothermic addition is crucial to prevent runaway reactions that could compromise stereochemical control. The extended reaction time of 3 to 10 hours post-addition ensures complete consumption of the starting materials, maximizing the overall yield. Following the reaction, the strategic addition of purified water prior to concentration aids in precipitating inorganic salts and facilitates the isolation of the organic product. This standardized protocol minimizes operator variability and ensures consistent batch-to-batch quality, making it an ideal candidate for technology transfer to commercial manufacturing suites.
- Prepare Reactor A with p-hydroxybenzaldehyde, water, phase transfer catalyst, ammonium chloride, and Lewis base, stirring at 10-30°C.
- Dissolve bromotetraacetyl glucose in acetone in Reactor B and slowly drip into Reactor A, maintaining temperature for 3-10 hours.
- Add purified water, concentrate under reduced pressure until turbid, cool to 0-5°C, filter, wash, and recrystallize with ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible economic and operational benefits. The primary driver of value is the substantial increase in process yield, which effectively reduces the raw material consumption per kilogram of finished intermediate. By nearly doubling the yield compared to legacy methods, the demand for expensive starting materials like bromotetraacetyl glucose is significantly curtailed, leading to a direct reduction in variable manufacturing costs. Additionally, the elimination of potassium iodide and strong caustic sodas removes the need for specialized handling equipment and reduces the burden on wastewater treatment facilities, as the effluent stream is less laden with heavy halides and extreme pH adjusters. This simplification of the chemical bill of materials (BOM) enhances supply chain security by relying on commodity chemicals like ammonium chloride and carbonates, which are globally available and less prone to market volatility than specialized catalysts.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the drastic improvement in mass efficiency. Traditional routes with yields of 20-30% imply that nearly 70-80% of the input mass is lost to waste or byproducts, representing a massive sunk cost in raw materials. By elevating yields to the 50% range, the effective cost per unit of product is nearly halved regarding material inputs. Furthermore, the removal of KI and the reduction in solvent usage for workup lower the utility and disposal costs associated with production. The simplified purification process, requiring only water washing and ethanol recrystallization, reduces the consumption of energy-intensive distillation steps and chromatography resins, contributing to a leaner and more cost-effective manufacturing operation that maximizes margin potential.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche reagents or complex catalytic systems that have limited supplier bases. This novel method mitigates such risks by utilizing a robust cocktail of reagents including ammonium chloride, acetone, and common carbonates, all of which are produced at a massive global scale with stable pricing. The operational simplicity of the reaction, which tolerates a broad temperature window and does not require stringent anhydrous conditions or inert atmospheres, reduces the likelihood of batch failures due to minor procedural deviations. This robustness ensures that production schedules can be met consistently, reducing lead times for high-purity pharmaceutical intermediates and providing a reliable buffer against market fluctuations or logistical disruptions in the supply of specialized chemicals.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat transfer and mixing challenges that are absent in the laboratory, but this methodology is inherently designed for scale-up. The use of water as a co-solvent improves heat capacity and safety, minimizing the risk of thermal runaways in large reactors. From an environmental standpoint, the process aligns perfectly with increasingly stringent global regulations regarding chemical emissions. By avoiding boron trifluoride and iodide salts, the facility avoids the generation of hazardous waste streams that require expensive incineration or specialized neutralization. The aqueous workup allows for easier separation of organic and inorganic phases, facilitating solvent recovery and recycling. This 'green' profile not only reduces regulatory compliance costs but also enhances the corporate sustainability metrics of the manufacturing entity, making it a preferred partner for environmentally conscious multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gastrodin intermediate synthesis. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, aiming to clarify the operational advantages and chemical rationale behind the new method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production, as it highlights the specific improvements in yield, purity, and process safety that distinguish it from incumbent technologies.
Q: How does the new method improve yield compared to traditional KI/NaOH processes?
A: Traditional methods using KI and sodium hydroxide often suffer from low conversion rates and yields typically ranging between 20-30%. The novel phase-transfer catalytic method described in patent CN111518148A utilizes milder Lewis bases and ammonium chloride, significantly reducing side reactions and improving weight yields to over 50%.
Q: What are the environmental benefits of avoiding boron trifluoride diethyl etherate?
A: Conventional catalysis using boron trifluoride diethyl etherate requires large volumes of solvents and generates complex post-treatment waste streams. The new aqueous-acetone system eliminates the need for hazardous Lewis acids, simplifying waste treatment and aligning with modern clean production standards.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It operates at mild temperatures (10-30°C), uses readily available reagents like ammonium chloride and carbonates, and involves simple workup procedures such as suction filtration and ethanol recrystallization, making it ideal for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gastrodin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader value chain of neuroprotective drug development. Our technical team has thoroughly analyzed the pathway described in CN111518148A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this laboratory innovation to full industrial maturity. We are equipped with state-of-the-art reactor systems capable of maintaining the precise thermal controls necessary for this phase-transfer catalyzed glycosylation, ensuring that the high yields and purity specifications observed in the patent are replicated consistently at scale. Our rigorous QC labs employ advanced HPLC and NMR techniques to verify the structural integrity and beta-selectivity of every batch, guaranteeing that the 4-formylphenyl-2,3,4,6-tetraacetyl-beta-D-glucopyranoside we supply meets the most stringent international pharmacopoeia standards.
We invite procurement leaders and R&D strategists to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By transitioning to this optimized synthetic route, your organization can realize significant reductions in COGS while securing a more sustainable supply source. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and to discuss route feasibility assessments for your long-term supply agreements. Let us partner with you to engineer a more efficient and profitable supply chain for your gastrodin projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
