Scalable Synthesis of MHPE: A Breakthrough in Agrochemical Intermediate Manufacturing
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key intermediates, particularly for compounds like Topramezone. Patent CN112441978B introduces a transformative preparation method for 1-methyl-5-hydroxypyrazole-4-carboxylic acid ethyl ester (MHPE), a critical building block in the agrochemical sector. This technology addresses long-standing bottlenecks in traditional manufacturing by utilizing diethyl diethoxymethylmalonate (DEMME) or a mixture of DEMME and ethoxymethylene malonic acid diethyl ester (EMME) as direct raw materials. By bypassing the rigorous requirement for high-purity EMME isolation, this process offers a robust pathway for reliable agrochemical intermediate suppliers seeking to optimize their production lines. The structural integrity of the final MHPE product is paramount for downstream efficacy, and this patent ensures that the chemical architecture remains pristine throughout the synthesis.
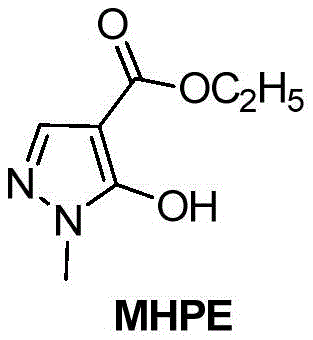
MHPE serves as a pivotal lead compound for reacting with ketals and acetals to generate a series of potent pyrazole pesticides. The ability to produce this intermediate with high fidelity and minimal impurity profiles is essential for pharmaceutical and agrochemical manufacturers. The disclosed method not only simplifies the operational workflow but also aligns with modern green chemistry principles by reducing waste associated with excessive purification steps. For R&D directors and procurement managers, understanding the nuances of this synthetic route provides a strategic advantage in securing cost-effective and high-quality supply chains for complex heterocyclic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
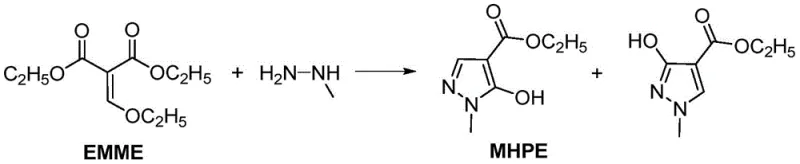
Traditionally, the synthesis of MHPE has relied heavily on the cyclization reaction of methylhydrazine with high-purity EMME. However, the preparation of EMME itself presents significant challenges, as it is typically generated via the condensation of diethyl malonate and triethyl orthoformate, a process that inevitably produces DEMME as an intermediate. In conventional workflows, achieving high yields of MHPE necessitates the complete conversion of DEMME to EMME, often requiring harsh conditions such as the use of large amounts of acetic anhydride and Lewis acid catalysts. This pursuit of purity creates a bottleneck; the separation of DEMME and EMME is notoriously difficult due to their similar boiling points, leading to low overall yields and extended processing times. Furthermore, the conventional route frequently results in the formation of isomeric by-products, such as ethyl 1-methyl-3-hydroxypyrazole-4-carboxylate, which complicates downstream purification and compromises the quality of the final active ingredient.
The Novel Approach
The innovative strategy outlined in patent CN112441978B fundamentally shifts the paradigm by accepting DEMME, or a mixture of DEMME and EMME, as the starting material. This approach recognizes that the rigorous conversion of DEMME to EMME is unnecessary for the successful formation of the pyrazole ring. By directly subjecting the DEMME/EMME mixture to amination and subsequent cyclization, the process eliminates the energy-intensive and time-consuming distillation steps required to isolate pure EMME. This modification drastically simplifies the reaction sequence, allowing for a more streamlined operation that is inherently more cost-effective. The reaction formula demonstrates how the mixture reacts with ammonia and methylhydrazine to yield the target MHPE alongside its isomer, but with optimized conditions that favor the desired product profile.
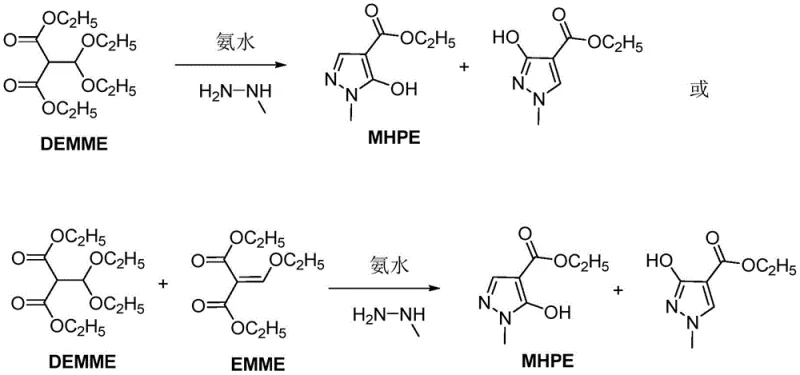
This novel approach effectively decouples the yield of the final product from the conversion rate of the intermediate DEMME. In practical terms, this means that manufacturers no longer need to chase 100% conversion in the upstream esterification step, which often leads to degradation and by-product formation. Instead, the process tolerates the presence of the diethoxymethyl group, which is transformed during the amination stage. This flexibility allows for the use of crude reaction mixtures directly from the upstream synthesis of diethyl malonate derivatives, thereby reducing raw material costs and minimizing solvent usage. For supply chain heads, this translates to a more resilient production model that is less susceptible to fluctuations in the quality of intermediate feedstocks.
Mechanistic Insights into Amination and Cyclization Reactions
The core of this technological advancement lies in the sequential amination and cyclization mechanism. In the first stage, the DEMME or DEMME/EMME mixture undergoes an amination reaction with ammonia water. This step is critical as it prepares the molecular framework for the subsequent ring closure. The reaction is conducted in an aqueous solvent at controlled temperatures ranging from 0 to 50°C, preferably between 0 and 20°C. The use of water as a solvent is particularly advantageous from an environmental and safety perspective, replacing hazardous organic solvents often found in traditional protocols. During this phase, the ethoxy groups on the malonate backbone are susceptible to nucleophilic attack by ammonia, facilitating the formation of an amidine-like intermediate that is primed for cyclization. The precise control of temperature and the dropwise addition of ammonia ensure that side reactions are minimized, preserving the integrity of the reactive centers.
Following amination, the mixture is subjected to cyclization with methylhydrazine. This step involves mixing the first reaction liquid with methylhydrazine at temperatures between -10 and 50°C, followed by heating to 40-90°C for a duration of 4 to 12 hours. The thermal energy provided in this stage drives the intramolecular condensation that forms the pyrazole ring. The mechanism likely involves the nucleophilic attack of the hydrazine nitrogen on the carbonyl carbon, followed by dehydration to establish the aromatic heterocycle. Crucially, the presence of the diethoxymethyl group in the starting material does not inhibit this cyclization; rather, it is accommodated within the reaction pathway, eventually leading to the hydroxyl-substituted pyrazole structure. The final neutralization with hydrochloric acid at low temperatures (below 30°C) precipitates the product, allowing for easy filtration and washing with ice water to achieve high purity levels exceeding 96%.
How to Synthesize 1-methyl-5-hydroxypyrazole-4-carboxylic acid ethyl ester Efficiently
The synthesis of MHPE via this patented route is designed for operational simplicity and industrial scalability. The process begins with the dissolution of the raw material mixture in water, followed by a controlled amination step that sets the stage for ring formation. Detailed standard operating procedures regarding stoichiometry, agitation speeds, and specific temperature ramps are essential for replicating the high yields reported in the patent examples. The following guide outlines the critical phases of this synthesis, ensuring that technical teams can implement this cost-reduction strategy effectively in their own facilities.
- Dissolve DEMME or a DEMME/EMME mixture in water and perform an amination reaction with ammonia water at 0-50°C to obtain the first reaction liquid.
- Mix the first reaction liquid with methylhydrazine at -10 to 50°C, then heat to 40-90°C for cyclization to form the second reaction liquid.
- Neutralize the second reaction solution with hydrochloric acid at temperatures below 30°C, cool to 0-5°C, and filter to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this DEMME-based synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage is the drastic simplification of the upstream supply chain. By removing the requirement for high-purity EMME, companies can source cheaper, cruder intermediate mixtures directly from bulk chemical suppliers, thereby reducing the number of processing steps and the associated logistical overhead. This consolidation of steps leads to a significant reduction in manufacturing costs, as energy consumption for distillation and the usage of expensive dehydrating agents like acetic anhydride are largely eliminated. Furthermore, the shortened reaction time enhances throughput, allowing facilities to produce larger volumes of MHPE within the same operational window, effectively increasing asset utilization without the need for capital expansion.
- Cost Reduction in Manufacturing: The elimination of the rigorous purification step for EMME represents a major cost saving opportunity. In traditional methods, driving the conversion of DEMME to EMME to completion requires excess reagents and energy-intensive distillation to separate close-boiling components. By accepting the mixture, this new method avoids these costly unit operations entirely. Additionally, the use of water as a primary solvent reduces the expense associated with purchasing, recovering, and disposing of organic solvents. The overall process efficiency is improved, leading to lower variable costs per kilogram of MHPE produced, which directly improves the margin profile for the final herbicide product.
- Enhanced Supply Chain Reliability: Relying on pure EMME creates a single point of failure in the supply chain, as its production is complex and sensitive to process deviations. By shifting to a process that tolerates DEMME, manufacturers gain flexibility in raw material sourcing. Suppliers can provide the intermediate mixture with broader specifications, reducing the risk of batch rejections and supply interruptions. This robustness ensures a more consistent flow of materials to the production line, reducing lead time for high-purity agrochemical intermediates and enabling better inventory management. The ability to use a mixture also means that production can continue even if the upstream conversion rates fluctuate, providing a buffer against operational variability.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on aqueous chemistry and straightforward temperature controls. The reduction in hazardous reagents, such as large quantities of acetic anhydride and Lewis acids, simplifies waste treatment and lowers the environmental footprint of the facility. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the costs associated with environmental health and safety (EHS) management. The high yield of over 90% and purity of over 96% ensure that the process is not only environmentally friendlier but also commercially viable for commercial scale-up of complex pyrazole derivatives, meeting the stringent quality standards required by global agrochemical markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel MHPE synthesis method. These insights are derived directly from the experimental data and claims presented in patent CN112441978B, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of transitioning from conventional EMME-based routes to this more efficient DEMME-inclusive methodology.
Q: What is the primary advantage of using DEMME over pure EMME for MHPE synthesis?
A: Using DEMME or a DEMME/EMME mixture eliminates the need for difficult and costly purification steps to isolate high-purity EMME, significantly shortening reaction time and reducing production costs while maintaining yields above 90%.
Q: What are the typical yield and purity specifications for MHPE produced via this method?
A: According to patent CN112441978B, this novel preparation method consistently achieves a product yield of more than 90% and a product purity exceeding 96%, making it highly suitable for industrial-scale agrochemical production.
Q: How does this process impact the supply chain for Topramezone intermediates?
A: By utilizing easily available raw materials and bypassing the complex conversion of DEMME to pure EMME, this method enhances supply chain reliability and reduces lead times for high-purity agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-methyl-5-hydroxypyrazole-4-carboxylic acid ethyl ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the competitive agrochemical landscape. Our team of expert chemists has extensively analyzed the potential of the DEMME-based route described in CN112441978B and is fully prepared to assist clients in integrating this technology into their supply chains. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of MHPE meets the exacting standards required for Topramezone synthesis.
We invite you to collaborate with us to leverage these process improvements for your manufacturing needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production volume and current raw material sources. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your supply of high-purity 1-methyl-5-hydroxypyrazole-4-carboxylic acid ethyl ester is both economically optimized and technically secure for the long term.
