Scalable Synthesis of N-Acetyl Tellurocarbamates via Acetate Catalysis for Advanced Drug Discovery
Scalable Synthesis of N-Acetyl Tellurocarbamates via Acetate Catalysis for Advanced Drug Discovery
The recent disclosure in patent CN113912526A introduces a transformative methodology for the preparation of N-acetyl tellurocarbamate compounds, a class of organotellurium species that has historically been underexplored due to synthetic challenges. This innovation addresses a critical gap in organic tellurium chemistry by providing a robust, acetate-catalyzed pathway that utilizes readily available isonitriles and symmetric ditellurides as primary building blocks. For R&D directors and process chemists, this represents a significant leap forward, as it replaces hazardous and operationally complex protocols with a mild, air-stable procedure that maintains high efficiency. The ability to efficiently introduce the N-acetyl tellurocarbamate motif into complex molecular frameworks opens new avenues for medicinal chemistry, particularly in the design of novel bioactive agents where tellurium's unique redox properties can be leveraged.
From a supply chain and procurement perspective, the strategic value of this patent lies in its reliance on commodity chemicals rather than specialized, unstable reagents. The shift away from cryogenic conditions and toxic gases not only enhances laboratory safety but also drastically reduces the infrastructure costs associated with manufacturing. By enabling the synthesis of these high-value intermediates under standard heating conditions (60-100°C) and in the presence of air, the technology lowers the barrier to entry for commercial production. This makes the reliable pharmaceutical intermediate supplier landscape more competitive, as manufacturers can now offer high-purity organotellurium compounds with improved lead times and reduced logistical burdens associated with hazardous material transport.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tellurocarbamates has been plagued by severe operational constraints that hindered their widespread adoption in industrial settings. Pioneering work by Noboru Sonoda in the early 1990s established foundational routes, yet these methods were fraught with dangers and inefficiencies. For instance, one prominent approach required the reaction of secondary amines with butyl lithium and tellurium powder at cryogenic temperatures (-78°C) under a carbon monoxide atmosphere to generate a tellurium lithium carbamate intermediate.
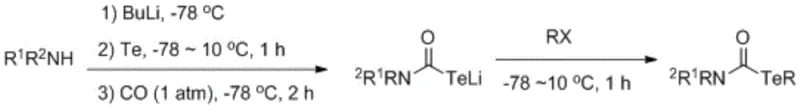
This legacy technique, while effective for simple substrates, imposed a heavy burden on process safety and equipment requirements due to the use of pyrophoric reagents and highly toxic carbon monoxide gas. Furthermore, alternative pathways involving lithium butyltelluride and carbamoyl halides were limited by narrow substrate scopes, often restricted to simple methyl or ethyl substitutions, failing to accommodate the complex aromatic systems required for modern drug discovery. These conventional methods essentially created a bottleneck, preventing the exploration of tellurium-containing scaffolds in more sophisticated pharmaceutical candidates.
The Novel Approach
In stark contrast, the methodology described in CN113912526A offers a streamlined and versatile solution that bypasses these historical hurdles. The core innovation involves the direct coupling of isonitriles and symmetric ditellurides mediated by inexpensive acetate salts, such as manganese(III) acetate. This reaction proceeds smoothly under an air atmosphere, eliminating the need for rigorous inert gas protection or specialized cryogenic cooling systems.

The general reaction scheme illustrates the elegance of this transformation, where the N-acetyl group is installed directly during the bond-forming event, creating the target tellurocarbamate structure in a single pot. This approach not only simplifies the operational workflow but also expands the chemical space accessible to chemists, allowing for the incorporation of diverse aryl, alkyl, and heterocyclic groups. By utilizing stable starting materials and benign catalytic conditions, this novel approach significantly enhances the feasibility of cost reduction in fine chemical manufacturing, making these specialized intermediates accessible for broader application.
Mechanistic Insights into Acetate-Catalyzed Telluration
The mechanistic underpinning of this transformation likely involves a radical-mediated pathway facilitated by the transition metal acetate catalyst. Manganese(III) acetate is well-known for its ability to generate radicals via single-electron transfer processes. In this context, it is hypothesized that the acetate species activates the tellurium-tellurium bond of the symmetric ditelluride, generating a tellurium-centered radical species. This reactive intermediate then attacks the electron-deficient carbon of the isonitrile group, initiating the formation of the new carbon-tellurium bond. Subsequent rearrangement and acetylation steps, potentially involving the acetate ligand itself as the acetyl source, lead to the final N-acetyl tellurocarbamate product.
Understanding this mechanism is crucial for impurity control and process optimization. The mild reaction conditions (60-100°C) prevent the thermal decomposition of the sensitive tellurium-carbon bond, a common issue in harsher synthetic routes. Furthermore, the use of acetate salts ensures a buffered environment that minimizes side reactions such as hydrolysis or oxidation of the tellurium center to tellurones. The compatibility with various functional groups, including esters, ethers, and sulfonamides, suggests that the radical species generated are sufficiently selective to avoid abstracting hydrogen atoms from sensitive positions on the substrate. This selectivity is paramount for maintaining high purity specifications, ensuring that the final API intermediates meet the stringent quality standards required for downstream pharmaceutical applications.
How to Synthesize N-Acetyl Tellurocarbamates Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly attractive for both laboratory scale-up and pilot plant production. The process begins by simply mixing the isonitrile, symmetric ditelluride, and the acetate catalyst in a polar aprotic solvent such as acetonitrile or DMF. The reaction vessel does not require degassing, as the protocol tolerates air, which is a significant advantage for reducing setup time and complexity. Heating the mixture to moderate temperatures drives the reaction to completion within a reasonable timeframe, typically between 2 to 12 hours depending on the specific substrate reactivity.
- Mix isonitrile, symmetric ditelluride, and acetate catalyst (e.g., Mn(OAc)3) in a solvent like acetonitrile under air atmosphere.
- Heat the reaction mixture to 60-100°C and stir for 2-12 hours until the isonitrile starting material is consumed.
- Cool the system, filter through celite, extract with dichloromethane, wash, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acetate-catalyzed protocol presents a compelling business case centered on risk mitigation and cost efficiency. The elimination of hazardous reagents like butyl lithium and carbon monoxide removes the need for specialized storage facilities and expensive safety monitoring systems, directly translating to lower overhead costs. Additionally, the stability of the raw materials—isonitriles and ditellurides—ensures a consistent supply chain, reducing the risk of production delays caused by the degradation of sensitive precursors during transport or storage.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive, specialized reagents with commodity chemicals. Acetate salts are inexpensive and widely available, contrasting sharply with the high cost of organolithium reagents and the infrastructure required to handle toxic gases. Furthermore, the ability to run reactions at elevated temperatures rather than cryogenic conditions significantly reduces energy consumption associated with cooling, leading to substantial cost savings in utility expenses. The simplified workup procedure, involving basic filtration and extraction, also minimizes labor hours and solvent usage, further optimizing the overall cost structure.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust starting materials that do not require cold-chain logistics. Isonitriles and symmetric ditellurides are stable solids or liquids that can be sourced from multiple vendors, reducing dependency on single suppliers. The tolerance of the reaction to air and moisture means that manufacturing can proceed without the strict inert atmosphere controls that often cause bottlenecks in fine chemical production. This flexibility allows for faster turnaround times and more reliable delivery schedules, ensuring that downstream drug development projects remain on track without interruption.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is straightforward due to the absence of exothermic hazards associated with pyrophoric reagents. The reaction profile is smooth and controllable, facilitating safe scale-up in standard stainless steel reactors. From an environmental perspective, the method generates less hazardous waste compared to traditional routes that produce stoichiometric amounts of lithium salts or require scrubbing of toxic gases. This alignment with green chemistry principles simplifies regulatory compliance and waste disposal, making it a sustainable choice for long-term commercial production of complex heteroatom intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on the practical aspects of adopting this method for your specific project needs.
Q: What are the advantages of this acetate-catalyzed method over traditional tellurocarbamate synthesis?
A: Unlike traditional methods requiring toxic carbon monoxide gas, cryogenic temperatures (-78°C), or pyrophoric reagents like butyl lithium, this novel protocol operates under mild conditions (60-100°C) in an air atmosphere using cheap acetate salts, significantly improving operational safety and scalability.
Q: What is the substrate scope for the R1 and R2 groups in this reaction?
A: The method demonstrates excellent functional group tolerance, accommodating alkyl, aryl, and heterocyclic aryl groups for both R1 (from isonitrile) and R2 (from ditelluride), including complex scaffolds like biphenyls, naphthyls, and methanesulfonamido-substituted phenyls.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly amenable to scale-up due to the use of stable, commercially available raw materials, the absence of sensitive reagents requiring inert gas protection, and a simplified workup procedure involving standard filtration and extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetyl Tellurocarbamate Supplier
As the demand for specialized organotellurium intermediates grows in the pharmaceutical and material science sectors, having a partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of N-acetyl tellurocarbamate meets the highest industry standards, supporting your critical drug discovery and development timelines with confidence.
We invite you to leverage our technical capabilities to optimize your supply chain for these high-value compounds. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your next breakthrough project.
