Revolutionizing 2,2-Diaryl Vinyl Alkyl Ether Production: A Scalable Green Chemistry Approach for Global Supply Chains
Revolutionizing 2,2-Diaryl Vinyl Alkyl Ether Production: A Scalable Green Chemistry Approach for Global Supply Chains
The landscape of organic synthesis for high-value intermediates is constantly evolving, driven by the dual imperatives of sustainability and economic efficiency. A groundbreaking development detailed in patent CN115536500A introduces a novel, one-pot preparation method for 2,2-diaryl vinyl alkyl ethers, a class of compounds critical for pharmaceutical and fine chemical applications. This technology represents a paradigm shift by utilizing dimethyl sulfoxide (DMSO) not merely as a solvent, but as an active carbon source that integrates directly into the product structure. By reacting readily available diaryl ketones with simple alcohols under basic conditions in an atmospheric environment, this method achieves high yields while bypassing the complex catalytic systems traditionally required. For R&D directors and procurement strategists, this innovation offers a compelling pathway to streamline manufacturing workflows, reduce dependency on precious metals, and secure a more resilient supply chain for complex vinyl ether derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl ethers has been fraught with significant technical and economic hurdles that hinder large-scale adoption. Traditional routes often rely on the addition of terminal alkynes to alcohols using expensive transition metal catalysts, such as rhodium complexes, which impose severe cost burdens and necessitate rigorous metal scavenging steps to meet pharmaceutical purity standards. Alternatively, nucleophilic substitution reactions involving haloalkenes or alkenyl esters require specialized starting materials that are not only costly but often hazardous to handle. Another prevalent method involves the reaction of ketones with specialized organic sulfur, phosphorus, or silicon reagents carrying alkoxymethyl groups. As illustrated in prior art, these approaches frequently demand strictly anhydrous and oxygen-free conditions, complicating reactor design and increasing operational expenditures. The reliance on exotic reagents and harsh conditions creates a bottleneck for industrial scalability, making the consistent supply of high-purity vinyl ethers a persistent challenge for global manufacturers.
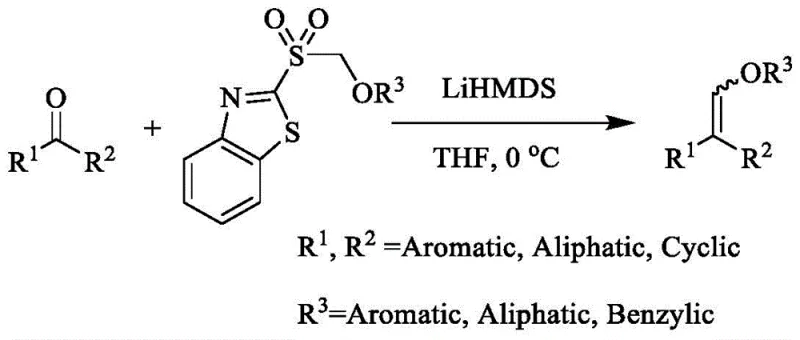
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN115536500A leverages a remarkably simple yet powerful chemical transformation. By employing common diaryl ketones and aliphatic alcohols in the presence of a base like potassium hydroxide, the process facilitates a direct coupling mediated by DMSO. This approach effectively inserts a carbon atom from the DMSO methyl group into the vinyl backbone, extending the carbon chain of the original ketone by one unit to form the desired 2,2-diaryl vinyl structure. The reaction proceeds efficiently under mild thermal conditions (60-120°C) and, crucially, tolerates the presence of air or oxygen, eliminating the need for inert gas purging. This simplicity translates directly into operational robustness, allowing for shorter reaction times—often as brief as 15 minutes—and significantly reduced energy consumption. The ability to use commodity chemicals as feedstocks fundamentally alters the cost structure of production, offering a distinct competitive advantage in the marketplace.
Mechanistic Insights into Base-Promoted DMSO Activation
From a mechanistic perspective, this transformation is a fascinating example of base-promoted C-H functionalization. The reaction initiates with the deprotonation of DMSO by the strong base (e.g., KOH) to generate a reactive dimsyl anion species. This nucleophile then attacks the carbonyl carbon of the diaryl ketone, forming a transient intermediate. Subsequent elimination and rearrangement steps, driven by the thermodynamic stability of the resulting conjugated system, lead to the formation of the vinyl ether double bond. The p-pi conjugation between the aryl groups and the newly formed double bond plays a critical role in stabilizing the product and driving the reaction forward. This mechanism avoids the formation of stable metal-carbon bonds typical in cross-coupling, thereby sidestepping the issues of catalyst poisoning and metal contamination. The elegance of this pathway lies in its atom economy regarding the solvent; DMSO serves a dual purpose, minimizing waste generation and simplifying the overall mass balance of the process.
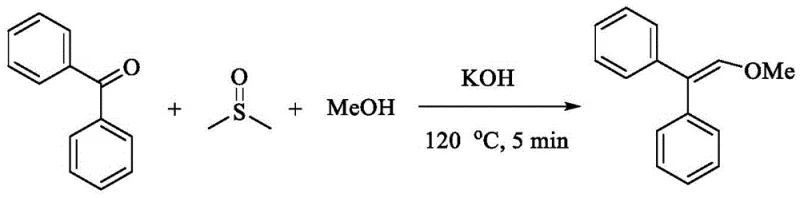
Furthermore, the impurity profile of this reaction is exceptionally clean, a factor of paramount importance for R&D teams focused on regulatory compliance. The mild basic conditions and the absence of transition metals mean that side reactions such as homocoupling or over-oxidation are minimized. The patent data indicates that a wide variety of substituents on the aromatic rings—including electron-donating methoxy groups and electron-withdrawing halogens—are well-tolerated without significant degradation in yield. This robustness suggests that the electronic properties of the substrate have a limited impact on the rate-determining step, likely due to the high reactivity of the dimsyl anion. Consequently, purification is straightforward, typically requiring only standard silica gel chromatography, which facilitates the isolation of products with high purity specifications suitable for sensitive downstream applications in drug discovery and material science.
How to Synthesize 2,2-Diaryl Vinyl Alkyl Ether Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it accessible for both laboratory-scale optimization and pilot plant operations. The protocol involves charging a reaction vessel with the diaryl ketone, the chosen alcohol, and a stoichiometric amount of base, followed by the addition of DMSO. The mixture is then heated to the optimal temperature range, typically around 100°C, for a short duration. Detailed standard operating procedures regarding exact molar ratios, workup protocols, and purification parameters are essential for reproducibility. For a comprehensive guide on executing this transformation with maximum efficiency and safety, please refer to the standardized synthesis steps outlined below.
- Combine diaryl ketone, alcohol, and potassium hydroxide (KOH) in a reaction vessel containing dimethyl sulfoxide (DMSO) as both solvent and reagent.
- Heat the mixture to 100°C under an air or oxygen atmosphere for approximately 15 minutes to facilitate the base-promoted coupling.
- Upon completion, quench with saturated brine, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift away from precious metal catalysts and specialized reagents towards commodity chemicals fundamentally de-risks the supply chain. By relying on diaryl ketones and simple alcohols, manufacturers can source raw materials from a broad base of suppliers, mitigating the risk of shortages or price volatility associated with niche intermediates. Furthermore, the one-pot nature of the reaction reduces the number of unit operations required, leading to lower capital expenditure on equipment and reduced labor costs per batch. The environmental profile is also significantly improved, aligning with increasingly stringent global regulations on solvent use and waste disposal, which can translate into lower compliance costs and enhanced corporate sustainability metrics.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as rhodium or nickel, along with their associated ligands, results in substantial direct material savings. Additionally, the use of DMSO as both solvent and reagent reduces the total volume of chemicals required, lowering waste treatment costs. The short reaction time further contributes to cost efficiency by maximizing reactor throughput and reducing utility consumption for heating and stirring. These factors combine to create a highly economical process that improves overall profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the key inputs—diaryl ketones and alcohols—are widely available bulk chemicals with established global supply networks. This reduces dependency on single-source vendors for exotic catalysts or unstable reagents. The robustness of the reaction conditions, particularly the tolerance to air and moisture, minimizes the risk of batch failures due to environmental fluctuations, ensuring consistent delivery schedules. This reliability is crucial for maintaining continuous production lines and meeting the just-in-time delivery expectations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple thermal requirements and lack of hazardous reagents. Scaling from gram to kilogram or tonnage levels does not require complex engineering controls for pyrophoric materials or high-pressure hydrogenation. Moreover, the reduced generation of heavy metal waste simplifies effluent treatment and aligns with green chemistry principles. This ease of scale-up accelerates time-to-market for new products and ensures long-term viability in a regulatory environment that increasingly penalizes environmentally burdensome manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific capabilities and constraints outlined in the patent literature. Understanding these nuances is vital for assessing the fit of this technology within your existing manufacturing portfolio. The answers reflect the proven performance of the method across diverse substrate classes and its operational flexibility under standard industrial conditions.
Q: What are the primary advantages of this DMSO-based method over transition metal catalysis?
A: Unlike conventional rhodium or nickel-catalyzed methods, this process eliminates the need for expensive transition metals and complex ligands. It utilizes commodity chemicals (ketones, alcohols, DMSO) and operates under mild aerobic conditions, drastically reducing raw material costs and simplifying downstream metal removal processes.
Q: Does this method support a wide range of substrate functional groups?
A: Yes, the protocol demonstrates excellent tolerance for various substituents on the diaryl ketone, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like halogens. It is also compatible with heterocyclic ketones such as thiophene and quinoline derivatives.
Q: Is the reaction suitable for large-scale industrial manufacturing?
A: Absolutely. The one-pot nature, short reaction time (approx. 15 mins), and use of non-toxic, inexpensive solvents make it highly amenable to scale-up. The absence of stringent anhydrous or oxygen-free requirements further enhances its operational feasibility for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Diaryl Vinyl Alkyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this DMSO-mediated synthesis for the production of high-value vinyl ether intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific thermal and separation requirements of this process, guaranteeing stringent purity specifications through our rigorous QC labs. We are committed to delivering consistent quality and supply continuity, leveraging our deep technical expertise to optimize this green chemistry route for your specific application needs.
We invite you to explore how this innovative method can enhance your product portfolio and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for reliable, high-purity pharmaceutical intermediate supply.
