Advanced Synthesis of 3,3'-(1,4-phenylene) Diglutaric Acid for High-Performance Polymer Manufacturing
Advanced Synthesis of 3,3'-(1,4-phenylene) Diglutaric Acid for High-Performance Polymer Manufacturing
The global demand for high-performance engineering plastics, specifically polyamides, polyimides, and advanced polyesters, continues to drive the need for robust and cost-effective monomer synthesis. A pivotal development in this sector is detailed in patent CN108821966B, which discloses a novel, environmentally benign method for synthesizing 3,3'-(1,4-phenylene) diglutaric acid. This compound serves as a critical building block for creating organic charge transfer compounds and high-molecular-weight materials with superior thermal and mechanical properties. The structural integrity of the target molecule, characterized by a central phenylene ring flanked by two glutaric acid moieties, is essential for the linearity and crystallinity of the resulting polymers. 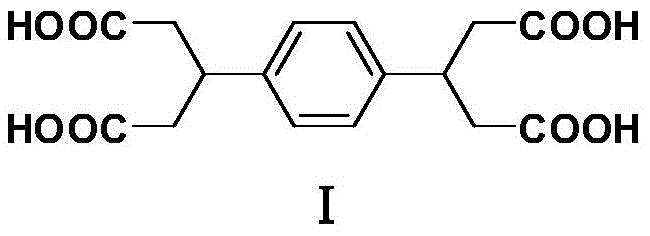 By shifting away from hazardous cyano-based precursors to abundant beta-keto esters, this technology represents a significant leap forward in green chemistry for fine chemical intermediates, offering a reliable pathway for manufacturers seeking to optimize their supply chains.
By shifting away from hazardous cyano-based precursors to abundant beta-keto esters, this technology represents a significant leap forward in green chemistry for fine chemical intermediates, offering a reliable pathway for manufacturers seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3'-(1,4-phenylene) diglutaric acid has been plagued by economic and safety inefficiencies inherent to cyano-based chemistry. Traditional routes, such as those documented in earlier literature, rely heavily on the condensation of terephthalaldehyde with ethyl cyanoacetate or cyanoacetic acid. 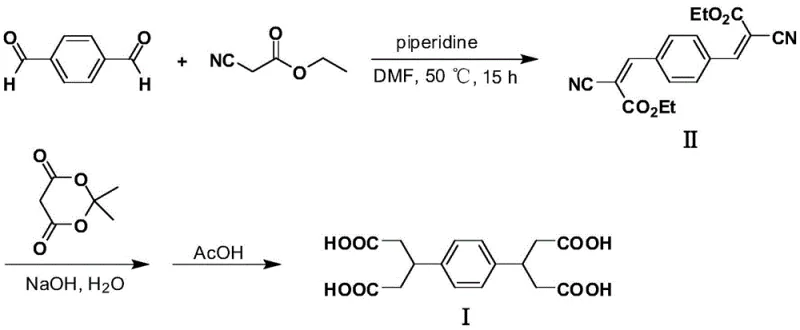 These methods suffer from severe drawbacks, primarily the high cost and toxicity associated with cyano-containing reagents. For instance, one prominent pathway requires the use of Meldrum's acid, a relatively expensive specialty chemical, alongside ethyl cyanoacetate, which poses significant handling risks due to its toxicity. Furthermore, the atom economy of these processes is poor; one specific prior art method necessitates a ten-fold molar excess of cyanoacetic acid relative to the aldehyde, creating massive waste streams and complicating the recovery of unreacted starting materials. Consequently, the total yield of these conventional routes stagnates around a mere 36%, rendering them economically unviable for large-scale commercial production of polymer additives.
These methods suffer from severe drawbacks, primarily the high cost and toxicity associated with cyano-containing reagents. For instance, one prominent pathway requires the use of Meldrum's acid, a relatively expensive specialty chemical, alongside ethyl cyanoacetate, which poses significant handling risks due to its toxicity. Furthermore, the atom economy of these processes is poor; one specific prior art method necessitates a ten-fold molar excess of cyanoacetic acid relative to the aldehyde, creating massive waste streams and complicating the recovery of unreacted starting materials. Consequently, the total yield of these conventional routes stagnates around a mere 36%, rendering them economically unviable for large-scale commercial production of polymer additives.
The Novel Approach
In stark contrast, the methodology outlined in CN108821966B introduces a transformative strategy that leverages a cascade reaction sequence to construct the carbon skeleton efficiently. This innovative approach utilizes terephthalaldehyde and ethyl acetoacetate as the sole starting materials, both of which are commodity chemicals available at a fraction of the cost of cyano-derivatives. The process bypasses the need for toxic nitriles entirely, instead employing a sophisticated tandem reaction involving Knoevenagel condensation, Michael addition, and intramolecular Aldol condensation. 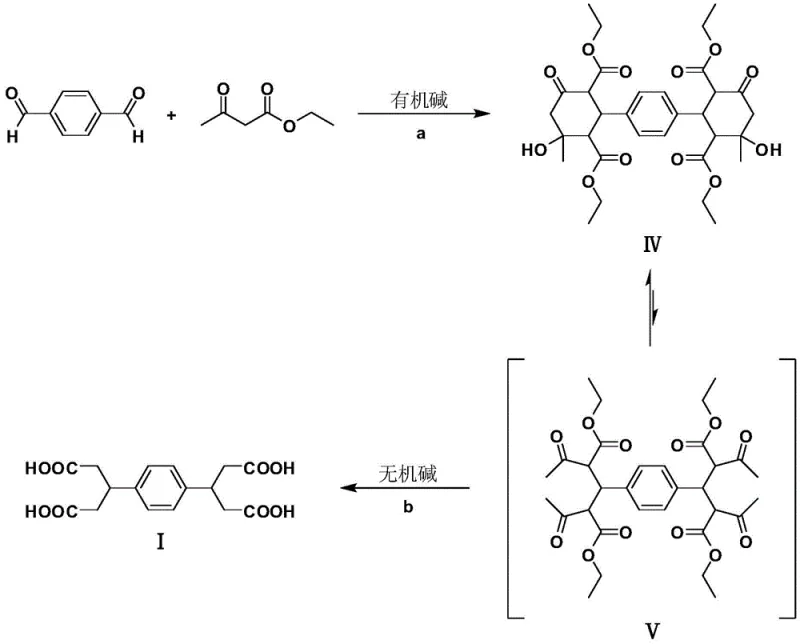 This sequence rapidly assembles a symmetrical bicyclohexanone structure (Compound IV) in high yield. Subsequently, a controlled ring-opening via retro-Aldol condensation under alkaline conditions linearizes the structure to form the desired dicarboxylic acid. This strategic redesign not only eliminates hazardous reagents but also boosts the overall process yield to an impressive 78.4%, demonstrating a clear superiority in both safety and efficiency for industrial applications.
This sequence rapidly assembles a symmetrical bicyclohexanone structure (Compound IV) in high yield. Subsequently, a controlled ring-opening via retro-Aldol condensation under alkaline conditions linearizes the structure to form the desired dicarboxylic acid. This strategic redesign not only eliminates hazardous reagents but also boosts the overall process yield to an impressive 78.4%, demonstrating a clear superiority in both safety and efficiency for industrial applications.
Mechanistic Insights into Cascade Condensation and Retro-Aldol Ring Opening
The chemical elegance of this synthesis lies in its multi-step cascade mechanism, which minimizes isolation steps and maximizes atom utilization. The reaction initiates with a double Knoevenagel condensation between the aldehyde groups of terephthalaldehyde and the active methylene groups of ethyl acetoacetate, catalyzed by an organic base such as diethylamine or DBU. This is immediately followed by a Michael addition, where the enolate species attack the conjugated double bonds formed in the initial step. The cycle concludes with an intramolecular Aldol condensation that closes the rings to form the stable, symmetrical bicyclohexanone intermediate (IV). This intermediate is chemically distinct, featuring a rigid cyclic structure that precipitates easily from the reaction medium, facilitating high-purity isolation without chromatography. The stability of Compound IV allows for flexible processing, serving as a robust platform for the subsequent transformation.
The second phase of the mechanism involves the strategic deconstruction of the cyclic intermediate to generate the linear acid. Under strong alkaline conditions, typically using sodium hydroxide or potassium hydroxide, Compound IV undergoes a retro-Aldol reaction. This base-catalyzed ring opening cleaves the carbon-carbon bonds formed during the initial cyclization, effectively reversing the Aldol step to reveal the linear tetraketone intermediate (V). Following this structural rearrangement, the substituted acetoacetate ester units within the chain undergo ketone decomposition (hydrolysis and decarboxylation). This final degradation step removes the extra carbonyl functionality introduced by the acetoacetate, leaving behind the clean glutaric acid side chains attached to the central phenylene core. This mechanistic pathway ensures that the final product, 3,3'-(1,4-phenylene) diglutaric acid, is obtained with exceptional purity, as the byproducts are primarily small, volatile molecules like ethanol and carbon dioxide, which are easily removed.
How to Synthesize 3,3'-(1,4-phenylene) Diglutaric Acid Efficiently
The operational simplicity of this patented method makes it highly attractive for process chemists aiming to establish a reliable supply of high-purity polymer intermediates. The synthesis is divided into two distinct stages: the formation of the cyclic precursor and its subsequent conversion to the final acid. The first stage requires precise control of the molar ratio between terephthalaldehyde and ethyl acetoacetate, optimally maintained between 1:4 and 1:6, to ensure complete conversion while minimizing side reactions. The use of organic bases like DBU accelerates the cascade reaction, allowing it to proceed under mild reflux conditions in ethanol. The second stage involves treating the isolated intermediate with a concentrated inorganic base solution, followed by acidification to precipitate the final product. This straightforward workflow eliminates the need for complex extraction protocols or hazardous solvents, streamlining the path from raw materials to finished goods.
- Condense terephthalaldehyde with ethyl acetoacetate using an organic base catalyst to form the symmetrical bicyclohexanone intermediate (IV).
- Subject the intermediate (IV) to alkaline conditions using inorganic bases like sodium hydroxide to induce ring-opening via retro-Aldol condensation.
- Perform ketone decomposition and acidification to isolate the final 3,3'-(1,4-phenylene) diglutaric acid product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this new synthesis route offers tangible benefits that extend beyond mere chemical curiosity. The replacement of specialized, high-cost reagents like Meldrum's acid and cyanoacetic acid with bulk commodity chemicals fundamentally alters the cost structure of the manufacturing process. Ethyl acetoacetate is produced on a massive global scale for various industries, ensuring a stable supply and consistent pricing, unlike the niche cyano-compounds which are subject to volatile market fluctuations and stricter regulatory controls. This transition to widely available raw materials significantly mitigates supply chain risks, ensuring continuity of production even during periods of raw material scarcity. Furthermore, the elimination of toxic cyanide groups simplifies waste management and reduces the environmental compliance burden, leading to substantial operational cost savings in effluent treatment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic reduction in raw material costs and the elimination of expensive purification steps. By avoiding the use of ten-fold excesses of cyanoacetic acid required in older methods, the new route minimizes material waste and the associated costs of recovering and recycling unreacted starting materials. The high yield of the intermediate (over 80%) and the final product (over 95% in the second step) means that less feedstock is required to produce the same amount of output, directly improving the gross margin. Additionally, the simplified workup, which relies on filtration and crystallization rather than column chromatography or complex extractions, reduces labor hours and solvent consumption, further driving down the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Dependence on toxic, regulated substances like cyanoacetic acid often introduces bottlenecks in the supply chain due to stringent transportation and storage regulations. By switching to non-toxic, non-regulated starting materials, manufacturers can streamline logistics and reduce lead times for raw material delivery. The robustness of the reaction conditions, which do not require extreme temperatures or pressures, also enhances equipment reliability and reduces downtime. This stability ensures that production schedules can be met consistently, providing downstream polymer manufacturers with a dependable source of critical monomers. The ability to source ingredients globally without regulatory hurdles adds a layer of resilience to the supply network, protecting against regional disruptions.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns perfectly with modern green chemistry principles, making it easier to scale up to multi-ton production without facing prohibitive environmental permits. The aqueous workup and the generation of benign byproducts simplify the wastewater treatment process, reducing the load on industrial sewage facilities. The high atom economy of the cascade reaction ensures that the majority of the input mass is incorporated into the final product, minimizing solid waste generation. This environmental efficiency not only lowers disposal costs but also enhances the corporate social responsibility profile of the manufacturer, appealing to end-users in the automotive and electronics sectors who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its adoption for commercial production. The following questions address common inquiries regarding the feasibility, safety, and quality parameters of the process described in patent CN108821966B. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear picture of the technology's capabilities. Addressing these points helps clarify how this method outperforms legacy technologies in terms of both operational safety and economic viability.
Q: What are the primary advantages of the new synthesis route over traditional cyano-based methods?
A: The new route eliminates the use of toxic and expensive cyanoacetic acid and Meldrum's acid, replacing them with cheap, non-toxic ethyl acetoacetate. Additionally, it drastically improves the total yield from approximately 36% in prior art to over 78%.
Q: Is this synthesis method scalable for industrial production of polymer intermediates?
A: Yes, the method utilizes common commodity chemicals and standard reaction conditions (reflux in ethanol/aqueous base), making it highly suitable for commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Q: How does the purification process compare to previous methods?
A: The post-treatment is significantly simplified. The intermediate (IV) precipitates directly from the reaction mixture as crystals, and the final product is obtained via simple acidification and filtration, avoiding complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-(1,4-phenylene) Diglutaric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation polymers and electronic materials. Our technical team has thoroughly analyzed the pathway described in CN108821966B and validated its potential for large-scale implementation. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,3'-(1,4-phenylene) diglutaric acid meets the exacting standards required for high-performance polymer synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this superior intermediate into your supply chain. Let us collaborate to drive innovation and efficiency in your polymer manufacturing operations.
