Optimizing Wipatasvir Intermediate A Production via Convergent Synthesis for Global Supply Chains
Optimizing Wipatasvir Intermediate A Production via Convergent Synthesis for Global Supply Chains
The global demand for direct-acting antivirals (DAAs) targeting Hepatitis C Virus (HCV) continues to drive innovation in pharmaceutical intermediate manufacturing. Patent CN108358881B introduces a transformative synthetic methodology for producing Wipatasvir Intermediate A, a critical building block for pan-genotypic HCV inhibitors. This technology addresses the longstanding bottlenecks of high cost and low yield associated with previous linear synthesis routes. By shifting to a convergent strategy that utilizes inexpensive, commercially abundant starting materials such as 2-bromo-benzyl alcohol, this process offers a robust pathway for scalable production. The structural complexity of the target molecule, featuring a fused tricyclic system with specific bromination patterns, requires precise control over regioselectivity and functional group tolerance, which this new protocol achieves through optimized reaction conditions.
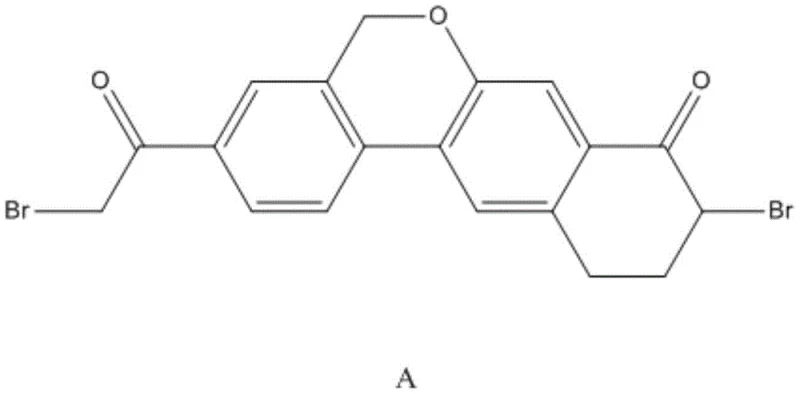
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the use of prohibitively expensive reagents found in prior art, such as potassium vinyltrifluoroborate. The disclosed route not only simplifies the operational workflow but also enhances the purity profile of the final intermediate, which is essential for downstream API synthesis. As the pharmaceutical industry seeks to reduce the cost of goods sold (COGS) for life-saving antiviral therapies, adopting such efficient manufacturing technologies becomes a strategic imperative for maintaining competitiveness in the generic and branded drug markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex biaryl intermediates like Wipatasvir Intermediate A has been plagued by inefficient linear sequences that suffer from compounding yield losses. Prior art methods, such as those described in WO2013/075029, relied heavily on costly coupling partners like potassium vinyltrifluoroborate, which drastically inflated the raw material expenditure and made industrial adoption economically unviable. Furthermore, alternative routes utilizing 2-bromo-5-iodobenzyl alcohol often encountered significant hurdles during the palladium-catalyzed ring-closing steps, resulting in suboptimal yields and difficult purification profiles due to the formation of homocoupling byproducts. These traditional approaches typically required harsh reaction conditions and multiple protection-deprotection cycles, increasing the environmental footprint and extending the overall production lead time. The reliance on scarce or specialized reagents also introduced supply chain vulnerabilities, making it difficult for manufacturers to guarantee consistent availability for large-scale commercial campaigns.
The Novel Approach
The innovative strategy outlined in patent CN108358881B fundamentally restructures the synthesis into a convergent architecture, effectively decoupling the construction of the two major molecular fragments before their final union. This approach leverages a highly efficient Suzuki-Miyaura cross-coupling reaction to join a brominated acetophenone derivative with a pinacol boronate ester derived from a tetralone scaffold. By selecting 2-bromo-benzyl alcohol as the primary feedstock, the process capitalizes on the availability and low cost of commodity chemicals, thereby achieving substantial cost reduction in pharmaceutical intermediate manufacturing. The route is designed with industrial pragmatism in mind, utilizing mild bases and standard palladium catalysts that are easily sourced and managed. This shift from linear to convergent synthesis not only improves the overall mass balance but also allows for parallel processing of intermediates, significantly compressing the critical path for production schedules.
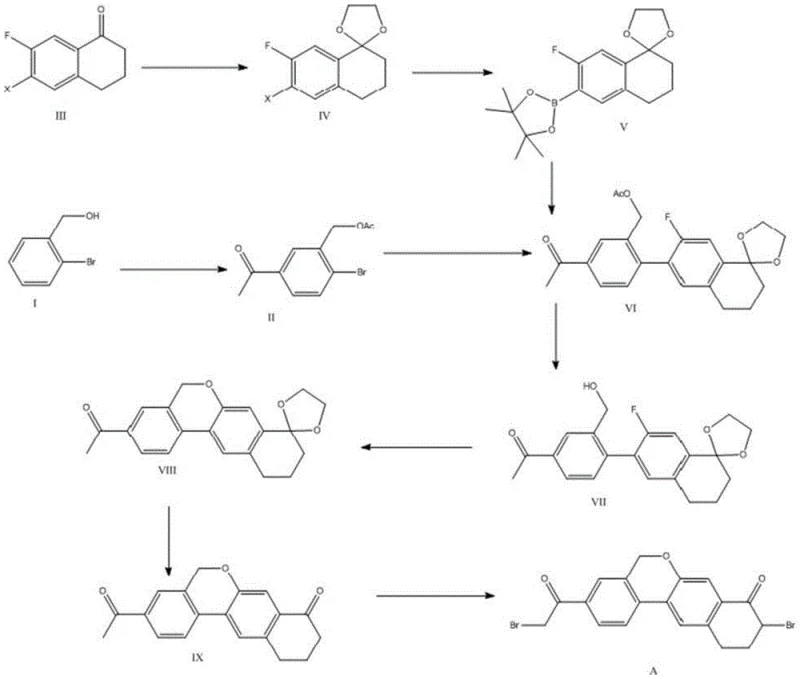
Moreover, the novel method incorporates a streamlined deprotection and cyclization sequence that avoids the use of aggressive reagents. The final bromination step employs pyridinium tribromide under controlled low-temperature conditions, ensuring high regioselectivity for the alpha-position of the ketone without affecting the sensitive ether linkages or the aromatic core. This level of chemical precision minimizes the generation of impurities, reducing the burden on downstream purification units and enhancing the overall process safety profile. For procurement teams, this translates to a more predictable and stable supply of high-quality intermediates, mitigating the risks associated with batch failures and inconsistent quality that often plague older synthetic methodologies.
Mechanistic Insights into Suzuki-Miyaura Coupling and Cyclization
The cornerstone of this synthetic advancement is the palladium-catalyzed Suzuki coupling between the acetylated aryl bromide and the boronic ester. Mechanistically, this reaction proceeds through a classic catalytic cycle involving oxidative addition of the aryl bromide to the Pd(0) species, followed by transmetallation with the organoboron compound activated by a base such as potassium carbonate or acetate. The choice of a pinacol boronate ester is particularly strategic, as this protecting group offers superior stability against protodeboronation and oxidation compared to free boronic acids, ensuring high fidelity during the coupling event. The subsequent reductive elimination regenerates the active catalyst and forms the crucial biaryl bond with excellent stereochemical integrity. This step is critical for establishing the correct spatial arrangement of the fused ring system, which is a prerequisite for the biological activity of the final antiviral agent.
Following the coupling, the synthesis employs a base-mediated intramolecular etherification to close the central heterocyclic ring. This transformation involves the nucleophilic attack of a phenolic hydroxyl group, generated in situ via hydrolytic deacetylation, onto a benzylic leaving group. The use of strong non-nucleophilic bases like potassium tert-butoxide facilitates this cyclization under relatively mild thermal conditions, preventing the degradation of the sensitive ketone functionalities present in the molecule. Impurity control is further enhanced by the specific selection of brominating agents in the final step; pyridinium tribromide acts as a solid, easy-to-handle source of electrophilic bromine, which reacts selectively with the enolizable ketone positions. This specificity prevents over-bromination or aromatic substitution, which are common side reactions in less optimized protocols, thereby securing a high-purity product suitable for stringent pharmaceutical applications.
How to Synthesize Wipatasvir Intermediate A Efficiently
The synthesis of Wipatasvir Intermediate A is achieved through a meticulously designed five-step sequence that balances reaction efficiency with operational simplicity. The process begins with the acetylation of 2-bromo-benzyl alcohol, followed by a Friedel-Crafts acylation to install the necessary ketone functionality on the aromatic ring. Simultaneously, a tetralone derivative undergoes carbonyl protection and borylation to create the coupling partner. These two fragments are then united via the aforementioned Suzuki coupling, followed by hydrolysis to reveal the phenol. The final stages involve a cascade of cyclization, deprotection, and selective bromination to yield the target molecule. This modular approach allows for the isolation and quality control of key intermediates, ensuring that any deviations can be corrected early in the process. Detailed standardized operating procedures for each transformation, including specific solvent systems, temperature ramps, and workup protocols, are essential for replicating the high yields reported in the patent literature.
- Perform acetylation on 2-bromo-benzyl alcohol followed by Friedel-Crafts acylation to generate the key aromatic ketone intermediate.
- Execute a Suzuki-Miyaura coupling between the brominated aromatic ketone and a pinacol boronate ester derived from tetralone derivatives.
- Complete the synthesis through hydrolytic deacetylation, intramolecular etherification cyclization, and final selective bromination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary driver of value is the substitution of exotic, high-cost reagents with bulk commodity chemicals. By eliminating the dependency on specialized fluoroborates and iodinated precursors, manufacturers can secure raw materials from a broader supplier base, reducing the risk of supply disruptions and price volatility. This diversification of the supply chain is critical for maintaining continuous production lines, especially in the context of global health initiatives where consistent drug availability is paramount. Furthermore, the simplified workflow reduces the number of unit operations and solvent exchanges, leading to lower utility consumption and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic reduction in raw material expenses. Replacing expensive coupling partners with 2-bromo-benzyl alcohol and tetralone derivatives lowers the input cost per kilogram of the final intermediate significantly. Additionally, the high efficiency of the Suzuki coupling and the one-pot nature of several steps minimize solvent usage and labor hours. The avoidance of complex purification steps, such as extensive chromatography, further contributes to cost savings by reducing the consumption of silica gel and eluents. These factors combine to create a leaner manufacturing model that improves the overall margin profile for the finished API.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures a robust and resilient supply chain. Unlike proprietary reagents that may have single-source limitations, the key inputs for this synthesis are produced by multiple chemical manufacturers globally. This redundancy protects against geopolitical or logistical shocks that could otherwise halt production. Moreover, the stability of the intermediates, particularly the pinacol boronate ester, allows for strategic stockpiling and flexible scheduling. The process conditions are also compatible with standard stainless steel reactors, meaning that existing manufacturing infrastructure can be utilized without the need for costly capital investments in specialized equipment.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is designed for seamless scale-up. The use of mild reaction temperatures and the avoidance of hazardous reagents like elemental bromine or strong mineral acids reduce the operational risk profile. The waste streams generated are less toxic and easier to treat, facilitating compliance with increasingly stringent environmental regulations. The high atom economy of the convergent synthesis means less waste is generated per unit of product, aligning with green chemistry principles. This sustainability advantage is becoming a key differentiator for suppliers seeking to partner with major pharmaceutical companies that have aggressive carbon reduction and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Wipatasvir Intermediate A using this patented methodology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of the route. Understanding these details is crucial for technical teams evaluating the potential for technology transfer and commercial implementation.
Q: What are the primary cost advantages of this new synthetic route for Wipatasvir Intermediate A?
A: The process replaces expensive reagents like potassium vinyltrifluoroborate and complex iodinated starting materials with readily available 2-bromo-benzyl alcohol and tetralone derivatives, significantly lowering raw material costs.
Q: How does the convergent synthesis strategy improve overall yield compared to linear methods?
A: By synthesizing two major fragments independently and joining them via a high-efficiency Suzuki coupling, the route minimizes cumulative yield losses typical of long linear sequences, thereby boosting total throughput.
Q: Is the final bromination step safe for large-scale industrial production?
A: Yes, the method utilizes pyridinium tribromide under mild conditions (0-10°C), avoiding hazardous elemental bromine and ensuring safer handling and easier waste treatment at commercial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Wipatasvir Intermediate A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against Hepatitis C. Our technical team has thoroughly analyzed the synthetic route described in CN108358881B and is fully prepared to execute this convergent strategy at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of material. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Wipatasvir Intermediate A meets the exacting standards required for API synthesis. We are committed to delivering not just a product, but a partnership built on technical excellence and supply chain security.
We invite pharmaceutical innovators and generic manufacturers to collaborate with us to optimize their supply chains for HCV therapeutics. By leveraging our expertise in process chemistry and scale-up engineering, we can help you realize the full cost-saving potential of this advanced synthetic route. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your project timelines and regulatory filings.
