Advanced Synthetic Route for Carfentrazone-ethyl: Enhancing Purity and Industrial Scalability
The agrochemical industry continuously seeks robust manufacturing pathways that balance high yield with stringent safety and environmental standards. Patent CN103819418A presents a significant technological advancement in the synthesis of Carfentrazone-ethyl, a potent triazolinone herbicide, and its critical intermediates. This intellectual property outlines a refined methodology that transitions away from hazardous high-pressure gas reactions toward a more controlled phase transfer catalysis system. By addressing the inherent instability of difluoromethylation reagents and the waste generation of traditional reduction methods, this patent offers a blueprint for modernizing production lines. For technical directors and procurement strategists, understanding this shift is vital, as it directly impacts the reliability of the agrochemical intermediate supplier network and the overall cost structure of herbicide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
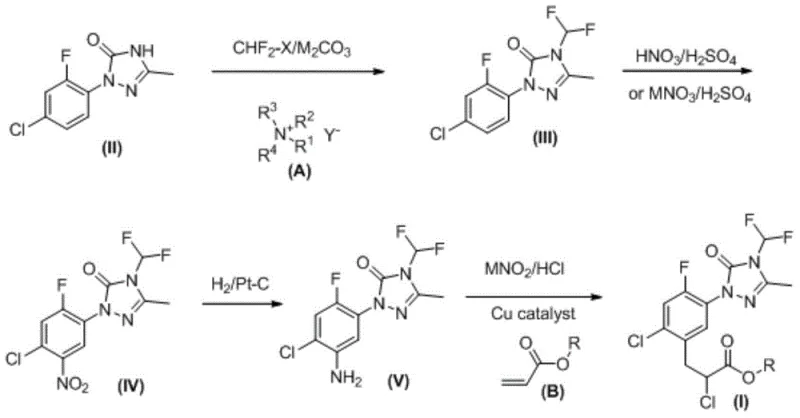
Historically, the synthesis of the key intermediate 1-(4-chloro-2-fluorophenyl)-4-difluoromethyl-3-methyl-1H-1,2,4-triazol-5-one relied on harsh conditions that posed severe industrial challenges. Prior art methods, such as those described in US5438149, necessitated the use of difluorochloromethane gas under extreme temperatures ranging from 160°C to 200°C in sealed high-pressure reactors. These conditions created a volatile environment where the acid-binding agent, typically potassium carbonate, would decompose into water upon absorbing hydrogen chloride. This generated water acted as a destructive agent, hydrolyzing the sensitive N-difluoromethyl groups and producing inorganic fluorides that corroded reactor vessels. Furthermore, the solvent DMF would thermally decompose into dimethylamine, creating foul odors and reacting with reagents, ultimately compromising the purity and color of the product while complicating subsequent nitration and reduction steps.
The Novel Approach
The methodology disclosed in CN103819418A fundamentally reengineers this critical step by introducing phase transfer catalysis to moderate the reaction environment. By employing tetrabutylammonium bromide alongside potassium carbonate in an aprotic polar solvent like DMF, the process enables efficient difluoromethylation at significantly milder temperatures and pressures. This strategic modification not only mitigates the safety risks associated with high-pressure gas handling but also preserves the integrity of the solvent and reagents. The result is a cleaner reaction profile that minimizes hydrolysis by-products and corrosion, thereby enhancing the quality of the intermediate before it even enters the nitration phase. This approach represents a paradigm shift towards safer, more sustainable cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Phase Transfer Catalysis and Pt-Catalyzed Reduction
The core innovation lies in the synergistic effect of the phase transfer catalyst (PTC) and the specific choice of reduction catalyst. In the difluoromethylation step, the quaternary ammonium salt facilitates the transport of the carbonate anion into the organic phase, enhancing nucleophilicity without requiring thermal extremes. This ensures that the difluoromethyl group is installed with high fidelity, preventing the formation of oxy-difluoromethylated by-products that are difficult to separate. Following nitration, the patent specifies the use of Platinum on Carbon (Pt/C) for the hydrogenation of the nitro group to the aniline derivative. Unlike traditional iron powder reduction, which generates tons of toxic iron sludge, or metallic nickel, which is pyrophoric and dangerous to handle on a large scale, Pt/C offers exceptional chemoselectivity. It effectively reduces the nitro group while leaving the sensitive chloro and fluoro substituents on the aromatic ring intact, a critical factor for maintaining the biological activity of the final herbicide.
Impurity control is rigorously managed through this catalytic precision. The avoidance of iron sludge eliminates a major source of heavy metal contamination that could poison downstream catalysts or remain in the final API. Additionally, the mild conditions of the PTC step prevent the thermal degradation of DMF, ensuring that dimethylamine impurities do not complicate the workup. The subsequent diazoarylation step, utilizing cuprous chloride and acrylic acid derivatives, proceeds smoothly with the high-purity aniline intermediate. This sequence ensures that the final high-purity agrochemical intermediate meets strict specifications for color and chemical composition, reducing the need for extensive recrystallization or chromatographic purification which often lowers overall yield.
How to Synthesize Carfentrazone-ethyl Efficiently
The synthesis protocol detailed in the patent provides a clear, scalable pathway for producing Carfentrazone-ethyl with industrial viability. The process begins with the preparation of the triazolone backbone, followed by the critical difluoromethylation under phase transfer conditions. Subsequent nitration and catalytic hydrogenation steps are optimized to maximize throughput while minimizing waste. The final coupling with acrylic acid derivatives completes the molecular architecture. For process chemists looking to implement this route, the detailed standardized synthesis steps see the guide below.
- Perform difluoromethylation of 1-(4-chloro-2-fluorophenyl)-3-methyl-1H-1,2,4-triazole-5-one using difluorochloromethane, potassium carbonate, and tetrabutylammonium bromide in DMF.
- Conduct nitration of the resulting intermediate using a mixture of fuming nitric acid and sulfuric acid to introduce the nitro group.
- Execute catalytic hydrogenation reduction using Pt/C catalyst to convert the nitro group to an amino group with high chemoselectivity.
- Finalize the synthesis via diazoarylation using sodium nitrite, HCl, and acrylic acid/ethyl acrylate with a cuprous chloride catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible operational benefits that extend beyond simple yield metrics. The elimination of high-pressure, high-temperature reactors reduces capital expenditure on specialized equipment and lowers maintenance costs associated with corrosion. Furthermore, the shift from stoichiometric metal reductions to catalytic hydrogenation drastically simplifies waste management protocols. By removing the generation of iron sludge and avoiding pyrophoric materials, facilities can achieve higher throughput with reduced downtime for cleaning and safety inspections. This operational efficiency directly supports the commercial scale-up of complex agrochemical intermediates, ensuring a steady flow of material to formulation plants.
- Cost Reduction in Manufacturing: The transition to phase transfer catalysis allows for the use of standard reactor vessels rather than expensive high-pressure autoclaves, significantly lowering infrastructure costs. Additionally, the recovery and reuse of the platinum catalyst, combined with the elimination of costly waste disposal fees for iron sludge, creates a leaner cost structure. The improved purity of intermediates also reduces solvent consumption during purification, further driving down variable production costs without compromising quality standards.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply continuity. By replacing hazardous pyrophoric nickel and corrosive high-pressure gas processes with safer, ambient-pressure alternatives, the risk of unplanned shutdowns due to safety incidents is minimized. The robustness of the Pt/C reduction ensures consistent batch-to-batch quality, reducing the likelihood of rejected shipments. This reliability is essential for maintaining reducing lead time for high-purity agrochemical intermediates in a global market where timing is critical for planting seasons.
- Scalability and Environmental Compliance: The process is inherently greener, aligning with increasingly strict global environmental regulations regarding heavy metal discharge and volatile organic compound (VOC) emissions. The absence of iron sludge and the containment of DMF decomposition products simplify wastewater treatment requirements. This environmental compatibility facilitates easier permitting for capacity expansion, allowing manufacturers to scale production from pilot batches to multi-ton volumes with fewer regulatory hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on why this method represents a superior choice for modern manufacturing.
Q: How does the new phase transfer catalysis method improve safety over conventional high-pressure difluoromethylation?
A: The novel method utilizes tetrabutylammonium bromide as a phase transfer catalyst, allowing the reaction to proceed at significantly lower temperatures and pressures compared to the traditional 160-200°C high-pressure conditions. This eliminates the risk of solvent decomposition and reactor corrosion associated with high-temperature hydrolysis by-products.
Q: Why is Platinum (Pt) preferred over Iron (Fe) or Nickel (Ni) for the reduction step?
A: Platinum catalysts offer superior chemoselectivity, preventing unwanted dehalogenation side reactions common with other metals. Furthermore, unlike iron powder which generates massive amounts of hazardous sludge, or pyrophoric nickel which poses fire risks, Pt/C is safer, easier to recover, and aligns with stricter environmental compliance standards.
Q: What are the purity advantages of this synthetic route for downstream processing?
A: By avoiding the formation of inorganic fluorides and amine by-products typical of high-temperature DMF decomposition, this route yields intermediates with superior color and purity. This reduces the burden on downstream purification steps, ensuring a more consistent quality for the final herbicide active ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carfentrazone-ethyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global agrochemical market. Our technical team has extensively analyzed routes like CN103819418A to ensure our production capabilities align with the highest standards of efficiency and safety. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver stringent purity specifications required by top-tier herbicide formulators. Our rigorous QC labs ensure that every batch of Carfentrazone-ethyl intermediate meets the precise chemical profiles necessary for optimal field performance.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into how our adoption of catalytic technologies reduces total landed costs. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments tailored to your specific volume requirements and quality standards.
