Revolutionizing Soluble Polyimide Synthesis: A Safe, Scalable Route to High-Purity Diamine Monomers
The landscape of high-performance polymer synthesis is undergoing a significant transformation, driven by the urgent need for safer and more scalable manufacturing processes. A pivotal development in this sector is detailed in patent CN116162030A, which introduces a robust preparation method for α,α'-bis(4-aminophenyl)-1,4-diisopropylbenzene. This compound serves as a critical diamine monomer for synthesizing soluble polyimides, materials renowned for their exceptional mechanical strength, optical transparency, and solubility. Unlike conventional approaches that rely on hazardous peroxidation steps, this novel methodology leverages a sequence of Wittig olefination, Grignard coupling, and acid-catalyzed condensation. For R&D directors and procurement strategists, this patent represents a paradigm shift towards process intensification, offering a pathway to produce high-purity electronic and aerospace-grade materials with drastically reduced operational risks and enhanced supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sterically hindered diamines like α,α'-bis(4-aminophenyl)-1,4-diisopropylbenzene has been plagued by severe safety and efficiency bottlenecks. The traditional industrial route typically begins with 1,4-diisopropylbenzene, subjecting it to a perilous peroxidation process to introduce oxygen functionality. This step necessitates the use of molecular oxygen under pressure, creating a significant explosion hazard that demands specialized, high-cost reactor infrastructure. Furthermore, the subsequent reduction and dehydration steps often generate a complex mixture of by-products, including various isomers and oligomers, which are notoriously difficult to separate. The resulting low selectivity not only diminishes overall yield but also complicates downstream purification, requiring energy-intensive distillation or chromatography that erodes profit margins and extends production lead times.
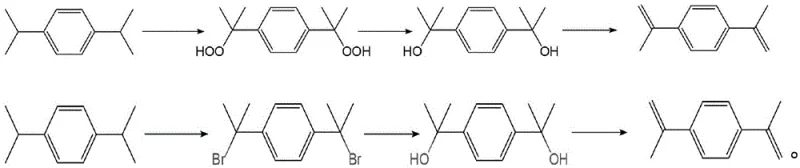
The Novel Approach
In stark contrast, the methodology disclosed in CN116162030A circumvents these inherent dangers by adopting a convergent synthetic strategy starting from 4-bromoacetophenone. This route replaces the hazardous oxidation chemistry with well-understood carbon-carbon bond-forming reactions. The initial Wittig reaction efficiently installs the necessary carbon framework, while the subsequent Grignard addition allows for precise control over the stereochemistry and substitution pattern. By avoiding the formation of unstable peroxide intermediates entirely, the process eliminates the need for high-pressure oxygen handling, thereby lowering the barrier to entry for manufacturers. This strategic pivot not only enhances workplace safety but also simplifies the equipment requirements, allowing production to proceed in standard glass-lined or stainless steel reactors commonly found in fine chemical facilities.
Mechanistic Insights into Wittig-Grignard Cascade and Acid-Catalyzed Coupling
The core of this technological breakthrough lies in the elegant orchestration of three distinct reaction mechanisms that ensure high selectivity and yield. The process initiates with a Wittig olefination, where 4-bromoacetophenone reacts with methyltriphenylphosphonium bromide in the presence of a strong base like potassium tert-butoxide. This step generates a vinyl intermediate (Compound II) with excellent regioselectivity, effectively setting the stage for the subsequent carbon skeleton expansion. Following this, the vinyl bromide undergoes magnesium insertion to form a Grignard reagent, which then attacks acetone. This nucleophilic addition is crucial as it installs the bulky isopropyl groups at the benzylic positions, creating the steric hindrance necessary for the final polyimide's solubility. The careful control of temperature during the Grignard formation prevents Wurtz-type coupling side reactions, ensuring a clean conversion to the diol precursor (Compound III).
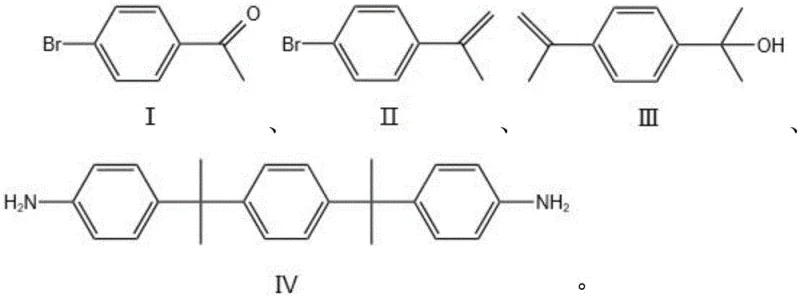
The final transformation involves an acid-catalyzed coupling between the diol precursor and aniline, mediated by strong sulfonic acids such as trifluoromethanesulfonic acid or methanesulfonic acid. This step proceeds via a carbocation mechanism where the hydroxyl groups are protonated and displaced, allowing the nucleophilic attack by aniline. The steric bulk of the isopropyl groups directs the substitution primarily to the para-position relative to the central benzene ring, minimizing ortho-substitution impurities. Moreover, the use of excess aniline acts both as a reactant and a solvent, driving the equilibrium towards the product. The final isolation is remarkably efficient; the product precipitates directly from the aqueous workup upon pH adjustment, leveraging the basicity of the amine groups to achieve high purity (>99%) without the need for column chromatography.
How to Synthesize α,α'-bis(4-aminophenyl)-1,4-diisopropylbenzene Efficiently
Implementing this synthesis requires precise control over stoichiometry and thermal conditions to maximize the yield of each intermediate. The process is designed to be telescoped where possible, minimizing solvent swaps and handling time. For the Wittig step, maintaining the reaction temperature between -10°C and 0°C is critical to prevent ylide decomposition, while the Grignard formation requires careful initiation to avoid runaway exotherms. The final coupling step benefits from elevated temperatures (80°C to 160°C) depending on the specific acid catalyst chosen, with triflic acid allowing for milder conditions. Detailed standard operating procedures regarding reagent grades, solvent drying, and quenching protocols are essential for reproducible results at scale.
- Perform Wittig reaction on 4-bromoacetophenone using methyltriphenylphosphonium bromide and potassium tert-butoxide to generate the vinyl intermediate.
- Convert the vinyl intermediate into a Grignard reagent using magnesium, followed by nucleophilic addition to acetone to form the diol precursor.
- React the diol precursor with excess aniline in the presence of a sulfonic acid catalyst (e.g., triflic acid) to yield the final diamine product via precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented route offers compelling economic and logistical advantages that extend beyond mere technical feasibility. The shift away from hazardous peroxidation chemistry fundamentally alters the risk profile of the manufacturing process, leading to lower insurance premiums and reduced regulatory compliance burdens. By utilizing commodity chemicals like 4-bromoacetophenone and acetone as starting materials, the supply chain becomes more resilient against raw material volatility compared to relying on specialized oxidized intermediates. Furthermore, the simplified purification protocol, which relies on crystallization rather than complex distillation trains, significantly reduces energy consumption and waste generation, aligning with modern sustainability goals and reducing the total cost of ownership for the production facility.
- Cost Reduction in Manufacturing: The elimination of high-pressure oxidation equipment represents a substantial capital expenditure saving, as manufacturers can utilize existing standard reactor fleets instead of investing in specialized safety infrastructure. Additionally, the high selectivity of the Wittig and Grignard steps minimizes the formation of hard-to-remove by-products, thereby reducing the consumption of solvents and adsorbents typically required for purification. The ability to recover and recycle excess aniline from the final step further contributes to raw material efficiency, driving down the variable cost per kilogram of the final diamine monomer.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified because the key starting material, 4-bromoacetophenone, is a widely available bulk chemical produced by numerous global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague specialty intermediate markets. Moreover, the robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment maintenance or safety shutdowns, ensuring a consistent and reliable flow of material to downstream polymer manufacturers who depend on just-in-time delivery for their own production lines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively without the need for exotic catalysts or extreme conditions that often fail upon translation from lab to plant. The absence of heavy metal catalysts and the use of aqueous workups for product isolation significantly reduce the load on wastewater treatment facilities, facilitating easier compliance with stringent environmental regulations. This 'green' profile not only future-proofs the manufacturing site against tightening emissions standards but also enhances the marketability of the final polyimide products to eco-conscious end-users in the electronics and automotive sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and mechanistic details outlined in the patent documentation, providing clarity for stakeholders evaluating the technology for potential licensing or procurement partnerships. Understanding these nuances is critical for assessing the fit of this intermediate within your specific application portfolio.
Q: Why is the new Wittig-Grignard route safer than traditional peroxidation methods?
A: Traditional methods often involve the peroxidation of 1,4-diisopropylbenzene, which requires handling hazardous oxygen and unstable peroxide intermediates under high pressure. The new route utilizes standard organic transformations (Wittig and Grignard) that operate under mild conditions without explosive risks.
Q: What are the purity specifications achievable with this synthesis method?
A: The patent demonstrates that the final product can achieve a purity of over 99% through simple recrystallization from ethanol, avoiding complex chromatographic separations typically required for isomeric mixtures in older processes.
Q: How does this process impact the cost of soluble polyimide manufacturing?
A: By starting from commercially available 4-bromoacetophenone and eliminating the need for specialized high-pressure oxidation equipment, the process significantly reduces capital expenditure and raw material costs, making high-performance polyimides more economically viable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable α,α'-bis(4-aminophenyl)-1,4-diisopropylbenzene Supplier
As the demand for high-performance soluble polyimides continues to surge in flexible electronics and advanced composites, securing a stable supply of high-quality diamine monomers is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in complex organic synthesis to deliver solutions that meet the rigorous demands of modern material science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your growth from pilot trials to full-scale manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of α,α'-bis(4-aminophenyl)-1,4-diisopropylbenzene meets the exacting standards required for next-generation polymer applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can become a strategic asset for your organization.
