Advanced Synthesis of Chiral Amine Intermediates for Commercial Scale-Up and Cost Efficiency
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral amine fragments, which serve as critical building blocks for high-value therapeutic agents such as Formoterol and Carmoterol used in asthma treatment. Patent CN112174837B introduces a transformative approach to synthesizing (R)-4-methoxy-alpha-methylphenethylamine, addressing long-standing inefficiencies in stereochemical control and process scalability. This innovation leverages a stable acridine intermediate derived from easily accessible chiral amino alcohol precursors, bypassing the need for complex resolution steps that traditionally plague this chemical space. By integrating a telescoped sequence of cyclization, selective reduction, and deprotection, the disclosed method achieves exceptional optical purity while minimizing unit operations. For global procurement and R&D teams, this represents a pivotal shift towards more sustainable and economically viable manufacturing pathways for beta-agonist intermediates. The following analysis dissects the technical merits and commercial implications of this proprietary synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral phenethylamines has relied heavily on methods that introduce significant operational hazards and economic inefficiencies, such as the imine formation and hydrogenation route depicted in earlier literature. 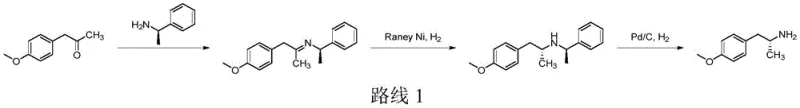 Traditional approaches often necessitate the use of pyrophoric catalysts like Raney-Nickel or expensive palladium on carbon under high-pressure hydrogenation conditions, creating substantial safety liabilities for large-scale facilities. Furthermore, strategies employing thermodynamic resolution inherently cap the maximum theoretical yield at fifty percent, leading to excessive waste generation and inflated raw material costs that erode profit margins. The requirement for multiple recrystallization steps to remove minor diastereomers further complicates the process flow, increasing solvent consumption and extending production lead times unnecessarily. Additionally, the use of chiral auxiliaries in asymmetric induction often involves costly reagents that must be stoichiometrically consumed and subsequently removed, adding layers of complexity to the downstream purification process. These cumulative factors render conventional methods less attractive for modern, cost-sensitive supply chains demanding high efficiency and safety compliance.
Traditional approaches often necessitate the use of pyrophoric catalysts like Raney-Nickel or expensive palladium on carbon under high-pressure hydrogenation conditions, creating substantial safety liabilities for large-scale facilities. Furthermore, strategies employing thermodynamic resolution inherently cap the maximum theoretical yield at fifty percent, leading to excessive waste generation and inflated raw material costs that erode profit margins. The requirement for multiple recrystallization steps to remove minor diastereomers further complicates the process flow, increasing solvent consumption and extending production lead times unnecessarily. Additionally, the use of chiral auxiliaries in asymmetric induction often involves costly reagents that must be stoichiometrically consumed and subsequently removed, adding layers of complexity to the downstream purification process. These cumulative factors render conventional methods less attractive for modern, cost-sensitive supply chains demanding high efficiency and safety compliance.
The Novel Approach
In stark contrast, the methodology outlined in CN112174837B utilizes a strategic Boc-protection strategy to facilitate the formation of a stable aziridine intermediate, which serves as a pivotal junction for stereochemical integrity. This novel pathway eliminates the need for chiral resolution entirely by leveraging the inherent chirality of the starting amino alcohol, thereby preserving the optical purity throughout the transformation without yield loss. The process is designed to be telescoped, meaning the intermediate acridine compound does not require isolation or purification before undergoing reduction ring-opening, which dramatically reduces processing time and solvent waste. By selecting mild reducing agents such as sodium borohydride or controlled hydrogenation conditions, the method mitigates the safety risks associated with combustible metal catalysts while maintaining high selectivity. This streamlined approach not only enhances the overall throughput of the manufacturing line but also ensures a consistent supply of high-purity intermediates essential for regulatory compliance in pharmaceutical production.
Mechanistic Insights into Boc-Protected Aziridine Cyclization
The core of this synthetic innovation lies in the precise construction of the aziridine ring system, which acts as a rigid template to enforce stereochemical outcomes during the subsequent ring-opening event. The reaction initiates with the activation of the hydroxyl group on the N-Boc protected amino alcohol using a sulfonyl chloride in the presence of an inorganic base within an ether solvent system. This intramolecular nucleophilic substitution proceeds with inversion of configuration or retention depending on the specific conditions, locking the chiral center into the strained three-membered ring structure of the acridine intermediate. The stability of this Boc-protected aziridine is crucial, as it prevents racemization and allows for handling without immediate degradation, a common failure point in less robust chiral intermediates. Subsequent reduction selectively cleaves the carbon-nitrogen bond of the aziridine ring, regenerating the amine functionality while preserving the established stereocenter with high fidelity. This mechanistic pathway ensures that the final product retains the desired (R)-configuration necessary for biological activity in downstream asthma medications.
Impurity control is inherently managed through the selectivity of the cyclization and reduction steps, which minimize the formation of regioisomers or over-reduced byproducts commonly seen in direct hydrogenation routes. The use of Boc protection groups serves a dual purpose: it masks the amine to prevent side reactions during cyclization and provides a handle for facile removal in the final step using acidic reagents. The purification strategy relies on salification of the final amine, which effectively precipitates the target compound while leaving organic impurities in the mother liquor, achieving purity levels exceeding ninety-nine percent without chromatographic separation. This level of chemical purity is paramount for R&D directors concerned with impurity profiles in drug substance registration files. The robustness of this mechanism against varying reaction scales ensures that the impurity spectrum remains consistent from kilogram to metric ton production, facilitating smoother technology transfer and regulatory approval processes.
How to Synthesize (R)-4-Methoxy-alpha-methylphenethylamine Efficiently
Implementing this synthesis requires careful attention to solvent selection and stoichiometry to maximize the efficiency of the cyclization and ring-opening sequences. The process begins with the reaction of the protected amino alcohol with sulfonyl chloride and base, followed by a reduction step that can be tuned using either hydride sources or catalytic hydrogenation depending on facility capabilities. Detailed standardized synthetic steps see the guide below, which outlines the specific molar ratios and temperature profiles optimized in the patent examples to ensure reproducibility. Operators must ensure strict control over the quenching procedures in the reduction step to manage exotherms safely, particularly when using reactive hydride reagents. The final deprotection and salification stage is critical for achieving the required physical form and purity specifications for commercial distribution. Adhering to these parameters allows manufacturers to replicate the high yields and optical purity reported in the intellectual property documentation.
- React N-Boc protected amino alcohol with sulfonyl chloride and inorganic base in ether solvent to form the stable acridine intermediate.
- Perform reduction ring-opening of the acridine compound using sodium borohydride or hydrogen to obtain the N-Boc protected chiral amine.
- Deprotect the intermediate using an acidic reagent followed by salification purification and dissociation to isolate the target chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented process offers profound advantages by fundamentally altering the cost structure and risk profile associated with chiral amine production. The elimination of resolution steps means that the theoretical yield ceiling is removed, allowing for significantly reduced raw material consumption per kilogram of finished product. This efficiency translates directly into lower variable costs and a smaller environmental footprint, aligning with corporate sustainability goals that are increasingly important for multinational corporations. Furthermore, the avoidance of pyrophoric catalysts like Raney-Nickel reduces the need for specialized safety infrastructure and hazardous waste disposal protocols, lowering overhead expenses related to plant maintenance and compliance. The ability to telescope steps without intermediate purification shortens the overall cycle time, enhancing the responsiveness of the supply chain to fluctuating market demands. These factors combine to create a more resilient and cost-effective supply source for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for expensive chiral resolving agents and reducing the number of isolation steps required. Eliminating the resolution step effectively doubles the potential yield compared to racemic synthesis followed by separation, drastically improving the utilization rate of starting materials. Additionally, the use of common commercial reducing agents instead of precious metal catalysts reduces the dependency on volatile commodity markets for palladium or platinum. The simplified workup procedures decrease solvent usage and energy consumption for distillation and drying, contributing to a leaner manufacturing cost base. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality or purity of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials and avoiding specialized chiral catalysts that may have long lead times, the supply chain becomes more robust against disruptions. The synthetic route is less sensitive to variations in raw material quality due to the purifying nature of the salification step in the final stage. This reliability ensures consistent delivery schedules for downstream customers who depend on just-in-time inventory models for their own production lines. The scalability of the reaction conditions from laboratory to industrial scale minimizes the risk of technology transfer failures, ensuring that supply commitments can be met reliably over the long term. Consequently, procurement managers can secure long-term contracts with greater confidence in the continuity of supply.
- Scalability and Environmental Compliance: The reduction in solvent volume and the elimination of heavy metal catalysts simplify the waste treatment process, making it easier to meet stringent environmental regulations. The process generates fewer byproducts that require complex disposal methods, reducing the overall environmental impact of the manufacturing facility. Scalability is enhanced by the exothermic control inherent in the chosen reagents, allowing for safe operation in large reactors without extensive cooling infrastructure. This alignment with green chemistry principles not only reduces compliance costs but also enhances the brand reputation of the supply chain partners. Ultimately, this creates a sustainable manufacturing model that is future-proofed against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthetic route. These answers are derived directly from the experimental data and advantageous effects described in the patent documentation to ensure accuracy. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs. The focus remains on practical considerations such as yield, purity, and operational safety that drive decision-making in the fine chemical sector.
Q: How does this new method improve upon traditional resolution techniques?
A: Unlike thermodynamic resolution which often suffers from maximum 50% theoretical yield, this asymmetric induction route avoids resolution steps entirely, significantly improving raw material utilization and reducing waste generation.
Q: What are the safety advantages regarding catalyst usage in this process?
A: The process allows for the use of sodium borohydride or controlled hydrogenation, avoiding the pyrophoric risks associated with Raney-Ni and reducing reliance on expensive palladium catalysts found in conventional hydrogenation routes.
Q: Is intermediate purification required between the synthetic steps?
A: No, the first two steps of the route do not require purification; the intermediate product is directly fed into the next step, which drastically simplifies the operational workflow and reduces solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-4-Methoxy-alpha-methylphenethylamine Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to deliver high-quality chiral intermediates for the global pharmaceutical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for drug substance synthesis. We understand the critical nature of chiral purity in asthma medications and are committed to maintaining the optical integrity of the product throughout the supply chain. Partnering with us means gaining access to a technically proficient team capable of optimizing this route for maximum efficiency and cost-effectiveness.
We invite you to contact our technical procurement team to discuss how this innovative process can benefit your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this superior synthetic method. Our experts are available to provide specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to enhance your supply chain resilience and drive down manufacturing costs through scientific innovation.
