Advanced Manganese-Catalyzed Asymmetric Oxidation for Scalable Chiral Oxindole Spiro-Indanone Production
The pharmaceutical industry continuously seeks robust methodologies for constructing complex chiral scaffolds, particularly spiro-oxindole derivatives which are prevalent in bioactive natural products and drug candidates. A significant breakthrough in this domain is detailed in patent CN111072549A, which discloses a novel synthesis method for chiral oxindole spiro-indanone compounds. This technology leverages a chiral tetradentate nitrogen ligand manganese complex to directly catalyze the asymmetric oxidation of benzyl C-H bonds. Unlike traditional methods that often suffer from harsh conditions and poor atom economy, this innovation utilizes liquid-phase hydrogen peroxide as a green oxidant under mild temperatures ranging from -30°C to 0°C. The result is a highly enantioselective process that delivers target molecules with exceptional purity, addressing critical needs for reliable pharmaceutical intermediate suppliers seeking sustainable and efficient manufacturing routes.
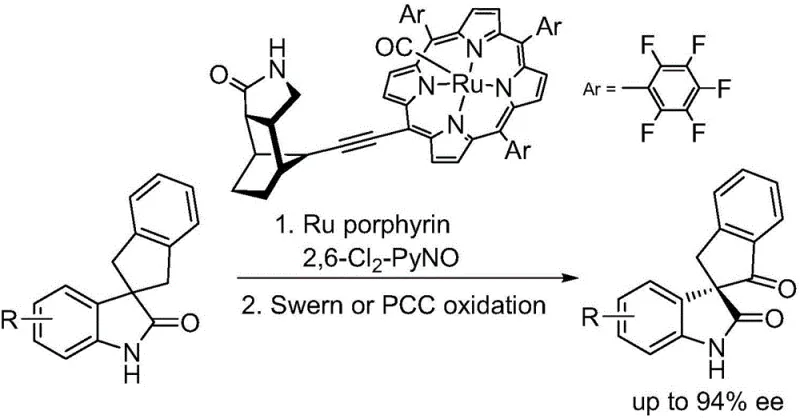
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral oxindole spiro-indanone compounds has relied heavily on transition metal catalysts such as ruthenium porphyrins, often requiring stoichiometric amounts of specialized oxidants like 2,6-dichloropyridine N-oxide. As illustrated in prior art, these conventional pathways frequently exhibit low conversion rates, necessitating additional oxidation steps using reagents like Swern oxidation or Pyridinium Chlorochromate (PCC). These secondary steps not only introduce toxic chromium waste and hazardous sulfur byproducts but also drastically increase the operational complexity and cost of goods sold. Furthermore, the requirement for multiple purification stages to remove heavy metal residues and side products creates significant bottlenecks in supply chain continuity, making it difficult for procurement managers to secure consistent volumes of high-quality intermediates without incurring substantial downstream processing expenses.
The Novel Approach
The methodology presented in the patent data revolutionizes this landscape by employing a biomimetic manganese catalyst system that enables direct, single-step asymmetric oxidation. By utilizing hydrogen peroxide, a commercially abundant and environmentally benign oxidant, the process eliminates the need for toxic co-oxidants and subsequent harsh oxidation treatments. This streamlined approach not only simplifies the workflow but also enhances the overall yield and enantiomeric excess, with reported values reaching up to 92% ee in optimized examples. The ability to achieve such high stereocontrol under mild conditions represents a paradigm shift for cost reduction in API manufacturing, allowing producers to bypass expensive and dangerous reagent handling protocols while maintaining rigorous quality standards required for clinical applications.
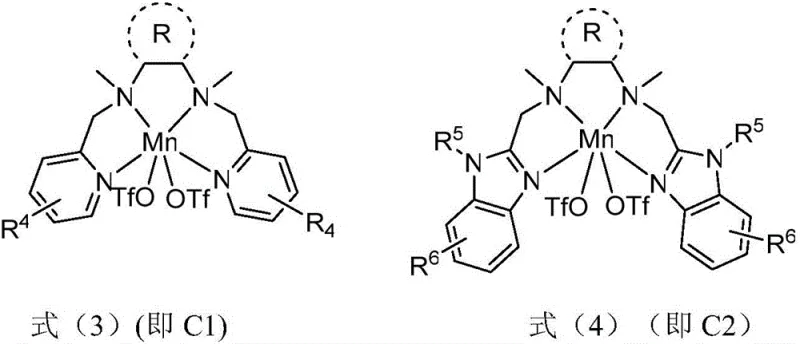
Mechanistic Insights into Mn-Catalyzed Asymmetric C-H Oxidation
The core of this technological advancement lies in the unique structure of the chiral manganese complex, specifically designed with a tetradentate nitrogen ligand framework that mimics the active sites of biological enzymes. As depicted in the catalyst structures, the coordination environment around the manganese center is finely tuned to facilitate the activation of the benzylic C-H bond with high spatial precision. The chiral diamine backbone of the ligand induces a specific steric environment that directs the approach of the oxidant, ensuring that oxygen insertion occurs selectively at the pro-chiral center. This precise control is critical for minimizing the formation of unwanted diastereomers and regioisomers, thereby simplifying the purification process and maximizing the recovery of the desired enantiomer.
Furthermore, the reaction mechanism benefits from the synergistic effect of acid additives, such as organic carboxylic acids, which assist in stabilizing the high-valent manganese-oxo species responsible for the hydrogen atom transfer. This stabilization prevents non-selective radical pathways that typically lead to racemic mixtures or over-oxidation byproducts. By maintaining the reaction temperature between -30°C and 0°C, the kinetic energy of the system is controlled to favor the lower-energy transition state associated with the desired chiral pathway. This mechanistic understanding allows R&D teams to fine-tune reaction parameters for diverse substrates, ensuring that the synthesis of complex pharmaceutical intermediates remains robust even when scaling from gram to kilogram quantities.
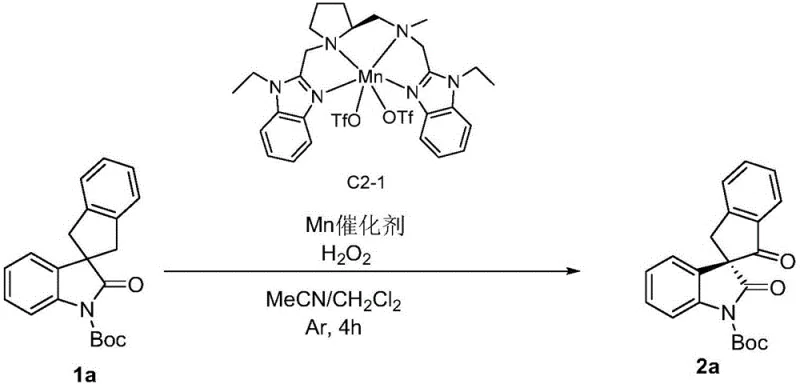
How to Synthesize Chiral Oxindole Spiro-Indanone Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to maximize both yield and enantioselectivity. The process begins with the preparation of a homogeneous solution containing the oxindole spiro indene precursor, the specific manganese catalyst variant (such as C2-1), and an acid additive in a suitable solvent system like dichloromethane or acetonitrile. The detailed standardized synthetic steps provided in the patent examples serve as a foundational guide for optimizing the molar ratios of oxidant to substrate, which typically range from 1:3 to 1:10. Adhering to these protocols ensures that the reactive manganese-oxo species is generated in situ at a rate that matches the substrate consumption, preventing decomposition and maintaining high efficiency throughout the transformation.
- Prepare a uniformly mixed reaction system containing the oxindole spiro indene precursor, chiral Mn catalyst, acid additive, and solvent at temperatures between -30°C and 0°C.
- Slowly add liquid phase hydrogen peroxide (oxidant) to the reaction mixture over a period of 2 hours while maintaining strict temperature control.
- Quench the reaction with sodium thiosulfate, followed by extraction, drying, and purification via column chromatography to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this manganese-catalyzed oxidation technology offers transformative benefits regarding cost structure and operational reliability. The shift away from precious metal catalysts like ruthenium and toxic oxidants significantly lowers the raw material input costs, while the elimination of multi-step oxidation sequences reduces labor and utility consumption. Moreover, the use of hydrogen peroxide generates water as the primary byproduct, drastically simplifying waste treatment protocols and reducing the environmental compliance burden associated with heavy metal disposal. These factors collectively contribute to a more resilient supply chain capable of meeting the stringent demands of global pharmaceutical markets without the volatility associated with scarce reagent availability.
- Cost Reduction in Manufacturing: The replacement of expensive ruthenium porphyrins and hazardous oxidants with economical manganese complexes and hydrogen peroxide leads to substantial savings in direct material costs. Additionally, the removal of downstream Swern or PCC oxidation steps eliminates the need for specialized equipment and cryogenic conditions often required for those reactions, further driving down capital and operational expenditures. This streamlined process flow allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins through improved process efficiency.
- Enhanced Supply Chain Reliability: Hydrogen peroxide and manganese salts are commodity chemicals with stable global supply chains, unlike specialized nitroxide oxidants which may face sourcing bottlenecks. The mild reaction conditions (-30°C to 0°C) are easily achievable with standard industrial cooling systems, reducing the risk of batch failures due to temperature excursions. This robustness ensures consistent delivery schedules and reduces the lead time for high-purity intermediates, enabling pharmaceutical partners to accelerate their own development timelines with confidence in the continuity of supply.
- Scalability and Environmental Compliance: The atom economy of using hydrogen peroxide as the terminal oxidant aligns perfectly with green chemistry principles, generating minimal waste and avoiding the generation of toxic chromium or sulfur byproducts. This environmental profile facilitates easier regulatory approval for commercial scale-up, as the process inherently meets stricter emission and effluent standards. Consequently, manufacturers can scale production from pilot plants to multi-ton facilities with reduced permitting hurdles, ensuring long-term viability and sustainability for the production of complex chiral building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric oxidation technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on the practical application of the manganese catalyst system. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of this Mn-catalyzed method over traditional Ruthenium porphyrin oxidation?
A: The Mn-catalyzed method utilizes benign hydrogen peroxide instead of toxic nitroxide oxidants and eliminates the need for subsequent Swern or PCC oxidation steps, significantly reducing environmental impact and operational complexity.
Q: What level of enantioselectivity can be achieved with this synthesis route?
A: The process demonstrates high enantioselectivity, consistently achieving ee values ranging from 60% to 92% depending on the specific substrate and catalyst variant used.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the mild reaction conditions (-30°C to 0°C) and the use of inexpensive, safe oxidants like hydrogen peroxide make this route highly amenable to commercial scale-up with reduced safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxindole Spiro-Indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies in securing a competitive edge in the pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical identity. By leveraging our expertise in process optimization, we can adapt the manganese-catalyzed oxidation protocol to meet your specific volume and quality requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your project needs. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits of switching to this greener, more efficient route. Our experts are ready to provide specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
