Scalable Iodide-Catalyzed Synthesis of Alpha-Keto Amides for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign pathways to access critical structural motifs. Patent CN102603552A introduces a transformative preparation method for alpha-keto amides, a privileged scaffold found in numerous protease inhibitors and bioactive natural products. This technology leverages an iodide-catalyzed oxidative coupling strategy that operates under remarkably mild conditions, specifically at room temperature and under an air atmosphere. By replacing hazardous high-pressure carbonylation processes with a simple mixing protocol involving methyl ketones, amines, and tert-butyl hydroperoxide, this invention addresses long-standing safety and cost inefficiencies in intermediate manufacturing. The significance of this development lies in its ability to streamline the supply chain for complex amide derivatives while adhering to the principles of green chemistry, making it an attractive option for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-keto amides has relied heavily on palladium-catalyzed carbonylation reactions involving halogenated benzenes, carbon monoxide, and secondary amines. These traditional routes present severe operational challenges, primarily due to the requirement for high-pressure carbon monoxide gas, which poses significant safety risks regarding toxicity and explosion hazards in a plant setting. Furthermore, the reliance on precious metal catalysts like palladium not only inflates raw material costs but also necessitates rigorous downstream purification steps to remove trace heavy metals to meet stringent pharmaceutical regulatory standards. Alternative multi-step transformations often involve cumbersome protection-deprotection sequences and specialized substrates that limit the overall atom economy and increase waste generation. These factors collectively contribute to extended lead times and higher manufacturing overheads, creating bottlenecks for procurement teams aiming to secure reliable supplies of high-purity intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN102603552A utilizes a direct oxidative coupling of methyl ketones with amines mediated by inexpensive iodide catalysts. This novel approach eliminates the need for gaseous carbon monoxide and precious metals entirely, conducting the reaction in common organic solvents or even water under ambient air conditions. The process demonstrates exceptional versatility, accommodating a wide range of substrates including aryl methyl ketones, heterocyclic ketones, and various primary or secondary amines. By operating at room temperature for periods ranging from 2 to 48 hours, the method significantly reduces energy consumption associated with heating or cooling reactors. The simplicity of the workup procedure, involving quenching with sodium thiosulfate and standard extraction, further enhances its appeal for commercial scale-up. This shift from high-risk carbonylation to safe oxidative coupling represents a paradigm shift in how alpha-keto amides can be manufactured efficiently.
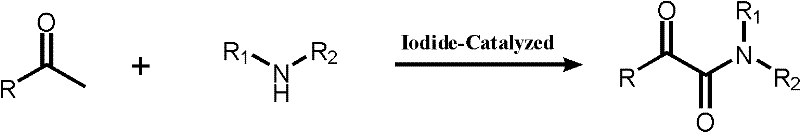
Mechanistic Insights into Iodide-Catalyzed Oxidative Coupling
The core of this innovation lies in the unique role of the iodide species, which acts as a redox mediator to activate the alpha-C-H bond of the methyl ketone. In the presence of tert-butyl hydroperoxide (TBHP), the iodide catalyst facilitates the generation of reactive iodine intermediates that abstract hydrogen atoms, leading to the formation of an alpha-keto species in situ. This activated ketone then undergoes nucleophilic attack by the amine component to form the desired alpha-keto amide bond. The catalytic cycle is sustained by the continuous regeneration of the active iodide species by the oxidant, ensuring high turnover numbers even with low catalyst loadings. This mechanism avoids the formation of stable organometallic complexes typical of palladium chemistry, thereby simplifying the reaction profile and reducing the likelihood of catalyst deactivation by substrate impurities. The compatibility with air suggests that molecular oxygen may also play a synergistic role in the reoxidation of the catalyst, further enhancing the sustainability of the process.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions associated with harsh acidic or basic conditions often found in classical amidation protocols. The mild oxidative environment prevents the degradation of sensitive functional groups such as esters, nitriles, and halogens, which are frequently present in advanced pharmaceutical intermediates. For instance, the patent data confirms successful conversion of substrates containing bromo, chloro, and trifluoromethyl groups without observing dehalogenation or hydrolysis byproducts. The use of sodium thiosulfate during the quenching phase effectively reduces any residual oxidizing iodine species to harmless iodide ions, preventing oxidative damage to the final product during isolation. This high level of chemoselectivity ensures that the resulting alpha-keto amides possess the high purity profiles required for downstream drug substance synthesis, reducing the burden on analytical quality control laboratories.
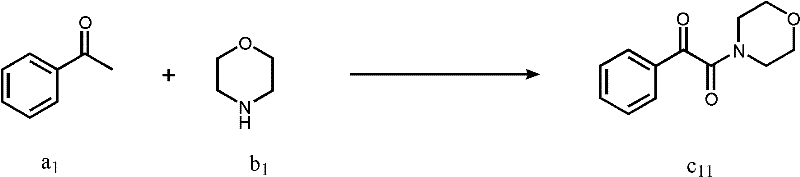
How to Synthesize Alpha-Keto Amides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and mixing protocols to maximize yield and safety. The general procedure involves charging a reaction vessel with the methyl ketone substrate, the chosen amine, an iodide catalyst such as potassium iodide or tetrabutylammonium iodide, and a solvent like isopropanol or dichloromethane. Tert-butyl hydroperoxide is then added as the terminal oxidant, and the mixture is stirred vigorously to ensure homogeneous contact between the phases. The reaction progress is typically monitored until the starting material is consumed, after which the mixture is treated with aqueous sodium thiosulfate to neutralize excess oxidant and iodine. Following extraction with ethyl acetate, the organic layer is dried and concentrated, and the crude residue is purified via silica gel chromatography or recrystallization to afford the pure alpha-keto amide. Detailed standardized synthesis steps are provided in the guide below.
- Configure a reaction system comprising a methyl ketone substrate, an amine compound (primary or secondary), an iodide-based catalyst, tert-butyl hydroperoxide as the oxidant, and a suitable organic solvent.
- Stir the reaction mixture under air atmosphere at room temperature for a duration ranging from 2 to 48 hours to facilitate the oxidative coupling.
- Quench the reaction with sodium thiosulfate solution, extract the organic phase with ethyl acetate, and purify the crude product via silica gel column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodide-catalyzed technology offers substantial strategic benefits beyond mere technical feasibility. The elimination of palladium catalysts and high-pressure carbon monoxide equipment translates directly into reduced capital expenditure and lower operating costs. By removing the dependency on precious metals, manufacturers can mitigate the volatility associated with commodity pricing of rhodium and palladium, leading to more stable long-term contracts. Furthermore, the ability to run reactions at room temperature under air pressure removes the need for specialized high-pressure autoclaves, allowing production to be scheduled on general-purpose glass-lined or stainless steel reactors. This flexibility enhances asset utilization rates and shortens the turnaround time between batches, enabling suppliers to respond more rapidly to fluctuating market demands for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with commodity iodide salts results in a drastic reduction in raw material costs per kilogram of product. Additionally, the simplified workup procedure eliminates the need for specialized scavenger resins or complex filtration systems required to remove trace heavy metals to ppm levels. This streamlining of the downstream processing phase reduces solvent consumption and waste disposal fees, contributing to a leaner overall cost structure. The economic efficiency is further amplified by the high yields reported across diverse substrates, minimizing the loss of valuable starting materials and maximizing the output from each production campaign.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as acetophenones and common amines ensures a robust supply base that is less susceptible to geopolitical disruptions affecting rare metal mining. The mild reaction conditions reduce the risk of unplanned shutdowns due to equipment failure or safety incidents related to high-pressure gas handling. Consequently, suppliers can offer more reliable delivery schedules and maintain higher safety stock levels without incurring excessive storage costs. This stability is crucial for pharmaceutical companies managing just-in-time inventory strategies for their active pharmaceutical ingredient (API) production lines, ensuring continuity of supply for life-saving medications.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, including the use of air as an oxidant source and the generation of minimal hazardous waste, align perfectly with increasingly stringent environmental regulations. Scaling this reaction from grams to tons does not require fundamental changes in the reaction engineering, as the heat transfer requirements are minimal due to the ambient temperature operation. This ease of scale-up reduces the time and cost associated with process validation and technology transfer between sites. Moreover, the avoidance of toxic carbon monoxide simplifies permitting processes and reduces the regulatory burden on manufacturing facilities, facilitating faster approval for commercial production in regions with strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodide-catalyzed synthesis platform. These answers are derived directly from the experimental data and claims presented in the patent literature to provide accurate guidance for process development teams. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing portfolio and supply chain strategy.
Q: What are the safety advantages of this iodide-catalyzed method over traditional carbonylation?
A: Unlike conventional methods requiring high-pressure carbon monoxide gas and palladium catalysts, this protocol operates at atmospheric pressure using air and liquid oxidants, significantly reducing explosion risks and toxicity hazards.
Q: Does this synthesis method support diverse functional groups on the ketone substrate?
A: Yes, the patent demonstrates excellent functional group tolerance, successfully accommodating halogens, esters, ethers, cyano groups, and various heterocycles like thiophene and pyridine without protecting groups.
Q: How does the catalyst loading impact the economic feasibility of this process?
A: The process utilizes inexpensive iodide salts (e.g., KI, NaI) or quaternary ammonium iodides instead of precious metals like palladium, drastically lowering catalyst costs and eliminating the need for expensive heavy metal removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Keto Amide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver high-value intermediates to the global market. Our technical team has extensively evaluated the iodide-catalyzed oxidation pathway described in CN102603552A and validated its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of alpha-keto amide meets the exacting standards required by top-tier pharmaceutical clients.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain resilience. Let us partner together to drive innovation and efficiency in the production of complex pharmaceutical intermediates.
