Advanced Metal-Free Synthesis of Dibenzoselenophenes for High-Performance OLED Materials
Advanced Metal-Free Synthesis of Dibenzoselenophenes for High-Performance OLED Materials
The rapid evolution of the organic optoelectronic industry demands increasingly sophisticated molecular architectures, particularly within the realm of heterocyclic compounds that serve as core building blocks for organic light-emitting diodes (OLEDs) and organic field-effect transistors (OFETs). A pivotal advancement in this sector is detailed in Chinese Patent CN112028872A, which discloses a groundbreaking synthetic methodology for constructing dibenzoselenophene compounds. This patent introduces a novel strategy that leverages TMSCN-catalyzed free radical cyclization between biarylboronic acid substrates and elemental selenium powder to form robust C-Se bonds. Unlike traditional approaches that rely heavily on scarce and costly transition metals, this innovation offers a metal-free pathway that operates efficiently under air atmosphere. For R&D directors and procurement strategists alike, this represents a significant paradigm shift, promising not only enhanced chemical purity by eliminating heavy metal residues but also a streamlined manufacturing process that drastically reduces the complexity associated with catalyst removal and waste treatment in the production of high-value electronic chemical intermediates.
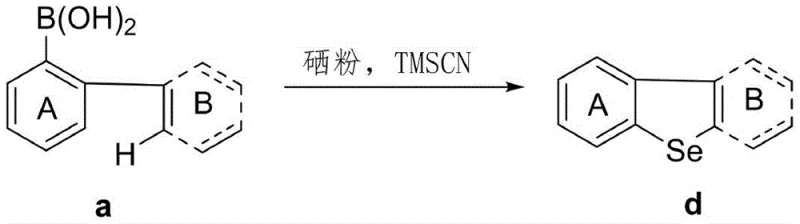
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dibenzoselenophene derivatives has been fraught with significant technical and economic challenges that hinder large-scale commercialization. Conventional methodologies predominantly rely on the use of diaryl selenide or aryl diselenide compounds as starting materials, necessitating the employment of noble metal catalysts such as palladium, silver, or transition metals like copper, titanium, and molybdenum to drive the cyclization process. These legacy routes often require严苛 reaction conditions, including stringent inert atmospheres and elevated temperatures, which increase energy consumption and operational risks. Furthermore, the reliance on precious metal catalysts introduces severe downstream processing burdens; removing trace metal impurities to meet the rigorous purity standards required for electronic materials often involves complex and expensive purification steps, such as column chromatography with specialized scavengers or recrystallization protocols that sacrifice overall yield. Additionally, the starting materials themselves, particularly specific aryl diselenides, can be difficult to source commercially or require multi-step syntheses, further inflating the cost of goods sold and creating supply chain bottlenecks for manufacturers aiming to produce consistent batches of optoelectronic intermediates.
The Novel Approach
In stark contrast to these cumbersome traditional methods, the technology disclosed in patent CN112028872A presents a remarkably elegant and efficient solution by utilizing biarylboronic acids and elemental selenium powder in the presence of a trimethylsilyl cyanide (TMSCN) catalyst. This innovative approach completely bypasses the need for transition metal catalysts, thereby eliminating the risk of metal contamination in the final product, a critical factor for semiconductor and display applications. The reaction proceeds smoothly under an air atmosphere, removing the necessity for expensive glovebox setups or rigorous nitrogen purging systems, which significantly lowers the barrier to entry for scale-up operations. Moreover, the use of biarylboronic acids as substrates offers a distinct advantage due to their commercial availability and stability compared to sensitive selenide precursors. The process is characterized by its operational simplicity, where the reaction mixture is simply stirred at moderate temperatures, followed by a straightforward workup involving filtration and concentration. This transition from complex catalytic cycles to a direct radical cyclization mechanism not only enhances atom economy but also aligns perfectly with green chemistry principles, offering a sustainable route for the cost reduction in electronic chemical manufacturing while maintaining exceptional product quality.
Mechanistic Insights into TMSCN-Catalyzed Radical Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the TMSCN catalyst, which activates elemental selenium to participate in a free radical cyclization process. Unlike traditional cross-coupling reactions that proceed through oxidative addition and reductive elimination cycles involving metal centers, this method likely involves the generation of reactive selenium species initiated by the interaction between TMSCN and selenium powder. These activated selenium species then engage with the biarylboronic acid substrate, promoting the formation of the critical carbon-selenium bond that closes the central heterocyclic ring. The radical nature of this transformation allows for a high degree of tolerance towards various functional groups, as the reaction conditions are mild enough to preserve sensitive moieties such as halogens and trifluoromethyl groups that might otherwise be compromised under harsher metallic catalytic conditions. This mechanistic distinctiveness ensures that the resulting dibenzoselenophene scaffolds retain the precise electronic properties dictated by their substituents, which is paramount for tuning the bandgap and charge transport characteristics in final optoelectronic devices.
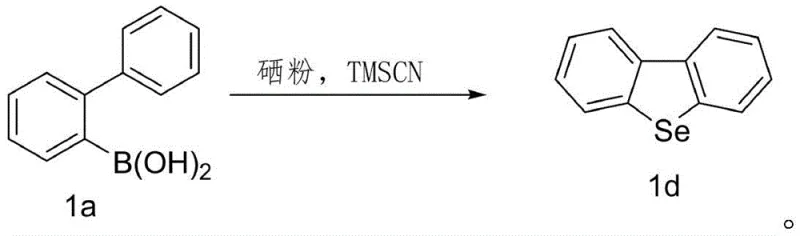
Furthermore, the impurity profile generated by this metal-free protocol is significantly cleaner compared to transition-metal catalyzed counterparts. In conventional palladium-catalyzed processes, side reactions such as homocoupling of the boronic acid or dehalogenation of the substrate can lead to complex mixtures that are difficult to separate. However, the TMSCN-mediated radical pathway demonstrated in the patent data shows remarkable selectivity, yielding the target dibenzoselenophene compounds with minimal byproduct formation. The absence of metal salts means that the crude reaction mixture primarily contains organic byproducts and unreacted starting materials, which can be easily removed through standard silica gel filtration or flash chromatography. This inherent purity advantage translates directly into higher isolated yields, with the patent reporting efficiencies reaching up to 98% for model substrates. For quality control teams, this implies a more robust and predictable manufacturing process where the risk of batch-to-batch variation caused by catalyst degradation or metal leaching is virtually non-existent, ensuring a consistent supply of high-purity OLED material intermediates.
How to Synthesize Dibenzoselenophene Efficiently
The practical implementation of this synthesis route is designed for ease of execution, making it highly attractive for process chemists looking to transfer laboratory success to pilot plant operations. The procedure involves a straightforward one-pot reaction where the biarylboronic acid substrate, TMSCN catalyst, and selenium powder are combined in dimethyl sulfoxide (DMSO) as the solvent. The choice of DMSO is critical, as the patent data indicates that other common organic solvents like toluene, dioxane, or DMF fail to support the reaction, highlighting the specific solvation effects required for the radical propagation steps. Once the reagents are mixed, the system is heated to a temperature range of 100-150°C, with optimal results observed around 140°C, and maintained under ambient air conditions for a period ranging from 4 to 48 hours depending on the specific substrate electronics. Upon completion, the workup is exceptionally simple, requiring only dilution with ether, filtration through a silica pad to remove excess selenium and polar impurities, and subsequent concentration. Detailed standardized synthesis steps see the guide below.
- Charge a reactor with biarylboronic acid substrate, TMSCN catalyst, selenium powder, and DMSO solvent.
- Stir the reaction mixture under air atmosphere at 100-150°C for 4-48 hours to facilitate radical cyclization.
- Cool to room temperature, dilute with ether, filter through silica gel, concentrate, and purify via flash chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TMSCN-catalyzed synthesis route offers compelling strategic advantages that extend far beyond mere chemical curiosity. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. By shifting away from proprietary or hard-to-source aryl diselenides and expensive noble metal catalysts, manufacturers can rely on commodity chemicals like biarylboronic acids and elemental selenium, which are available in bulk quantities from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and price volatility often associated with precious metals like palladium. Furthermore, the elimination of heavy metal catalysts removes the need for specialized waste disposal protocols required for toxic metal residues, leading to substantial cost savings in environmental compliance and waste treatment. The operational simplicity of running reactions under air rather than inert gas also reduces capital expenditure on specialized reactor equipment and lowers utility costs related to nitrogen or argon consumption, contributing to a leaner and more agile manufacturing footprint.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost inputs with low-cost alternatives. Traditional methods often consume stoichiometric or near-stoichiometric amounts of expensive ligands and catalysts, whereas this new method uses TMSCN in catalytic amounts (as low as 3 mol%), significantly lowering the material cost per kilogram of product. Additionally, the high yields reported, often exceeding 90% and reaching up to 98% for optimized substrates, mean that less raw material is wasted, improving the overall mass balance and reducing the cost of goods sold. The simplified purification process further reduces labor and solvent costs, as extensive chromatography or recrystallization steps needed to remove metal traces are no longer necessary, allowing for a more direct path from reactor to finished goods inventory.
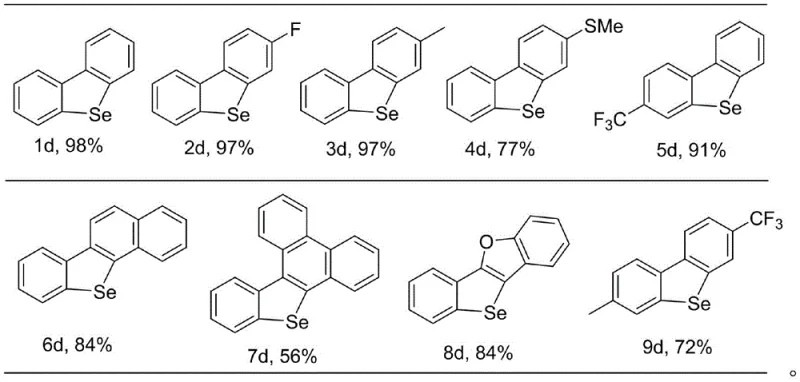
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for downstream device manufacturers who operate on tight production schedules. The robustness of this synthetic method, evidenced by its tolerance to air and moisture, ensures that production is less susceptible to interruptions caused by equipment failure or gas supply issues. The broad substrate scope demonstrated in the patent, covering various substituted benzene rings and fused aromatic systems, means that a single manufacturing platform can be adapted to produce a wide library of dibenzoselenophene derivatives without needing to requalify entirely new process chemistries. This flexibility allows suppliers to respond rapidly to changing market demands for specific OLED host or transport materials, ensuring continuity of supply even when product specifications evolve.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, but the straightforward nature of this radical cyclization suggests excellent scalability potential. The use of DMSO, a high-boiling polar solvent, facilitates heat transfer and mixing in large reactors, while the absence of pyrophoric reagents like butyllithium (used in some older methods) enhances operational safety. From an environmental perspective, the atom economy is superior because the selenium powder is incorporated directly into the product without generating stoichiometric metal salt waste. This aligns with increasingly stringent global regulations on hazardous waste discharge and supports corporate sustainability goals, making the final product more attractive to eco-conscious multinational corporations seeking green chemistry solutions in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing lines or for sourcing high-quality intermediates from external partners who utilize this advanced chemistry.
Q: What are the primary advantages of this TMSCN-catalyzed method over traditional palladium-catalyzed routes?
A: The primary advantage is the complete elimination of expensive and toxic transition metal catalysts such as palladium, silver, or copper. This significantly reduces raw material costs and simplifies the purification process by removing the need for rigorous heavy metal scavenging steps, which is critical for electronic grade materials.
Q: Is this synthesis method scalable for industrial production of OLED intermediates?
A: Yes, the method demonstrates excellent scalability potential due to its operational simplicity. It utilizes readily available starting materials like biarylboronic acids and selenium powder, operates under air atmosphere without stringent inert gas protection, and achieves high yields up to 98%, making it highly suitable for commercial scale-up.
Q: What is the functional group tolerance of this radical cyclization strategy?
A: The strategy exhibits broad functional group compatibility. It successfully tolerates various substituents including halogens (fluorine, chlorine), alkyl groups (methyl, tert-butyl), alkoxy groups, and trifluoromethyl groups on both aromatic rings, allowing for the diverse derivatization required in fine-tuning optoelectronic properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzoselenophene Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with a chemically proficient manufacturer is essential for securing a competitive edge in the market. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge synthetic methodologies like the TMSCN-catalyzed radical cyclization to deliver superior dibenzoselenophene intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of major display and lighting manufacturers without compromising on quality. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of heavy metal contaminants and ensure consistent batch-to-batch performance, which is critical for the reliability of final optoelectronic devices.
We invite industry leaders to collaborate with us to optimize their material sourcing strategies and reduce overall production costs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and purity requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our advanced metal-free synthesis capabilities can enhance your supply chain resilience and drive innovation in your product development pipeline.
