Revolutionizing Acyl Azide Production: A Deep Dive into Lewis Acid-Catalyzed Efficiency and Scalability
The landscape of organic synthesis for high-value intermediates is constantly evolving, driven by the need for safer, faster, and more cost-effective methodologies. Patent CN101481327B introduces a transformative approach to the preparation of acyl azides, a critical class of compounds widely utilized in the Curtius rearrangement and peptide coupling reactions. Unlike traditional methods that rely on hazardous acyl chlorides or sluggish uncatalyzed substitutions, this invention leverages Lewis acid catalysis to drive the nucleophilic displacement of benzotriazole from acyl benzotriazoles using sodium azide. The process operates under remarkably mild conditions, typically between 0°C and 45°C, and achieves completion within a mere 0.25 to 2 hours. This represents a drastic improvement over prior art, such as the method reported by Katritzky et al., which required 16 to 20 hours of reaction time. For R&D directors and process chemists, this patent offers a robust pathway to generate high-purity intermediates with minimal operational complexity, setting a new standard for efficiency in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acyl azides has been plagued by significant safety and operational challenges that hinder large-scale production. The classical route involves the reaction of acyl chlorides with sodium azide; however, acyl chlorides are notoriously unstable, moisture-sensitive, and corrosive, posing severe risks during storage and handling in an industrial setting. Furthermore, alternative methods utilizing acyl benzotriazoles without catalysis, as seen in earlier literature, suffer from prohibitively long reaction times ranging from 16 to 20 hours. These extended durations not only bottleneck reactor occupancy but also increase the risk of product degradation for thermally sensitive substrates. Perhaps most critically, these conventional uncatalyzed routes often necessitate purification via column chromatography to achieve acceptable purity levels. Column chromatography is inherently difficult to scale, consumes vast amounts of silica and solvents, and creates substantial hazardous waste, making it economically unviable for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN101481327B fundamentally redefines the synthetic strategy by introducing a Lewis acid catalyst to activate the acyl benzotriazole substrate. By employing catalysts such as zinc chloride, aluminum chloride, or cupric chloride, the reaction barrier is significantly lowered, allowing the nucleophilic attack by the azide ion to proceed rapidly at temperatures as low as 0°C to 45°C. This catalytic acceleration reduces the reaction window from nearly a day to as little as 15 minutes (0.25 hours), dramatically increasing throughput capacity. Moreover, the workup procedure is elegantly simple, involving standard liquid-liquid extraction with diethyl ether or ethyl acetate, followed by washing with sodium carbonate and brine. This eliminates the need for chromatographic purification entirely, enabling the direct isolation of acyl azides with purities consistently exceeding 95%, and often reaching 98-99%. 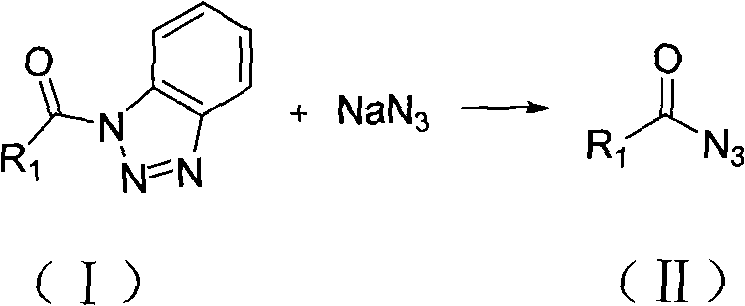
Mechanistic Insights into Lewis Acid-Catalyzed Nucleophilic Substitution
The core innovation of this process lies in the synergistic interaction between the Lewis acid catalyst and the acyl benzotriazole moiety. Mechanistically, the Lewis acid (such as AlCl3 or ZnCl2) coordinates with the carbonyl oxygen or potentially the nitrogen atoms of the benzotriazole leaving group, thereby increasing the electrophilicity of the carbonyl carbon. This activation facilitates the nucleophilic attack by the azide anion (N3-) derived from sodium azide. The presence of the catalyst stabilizes the transition state, allowing the displacement of the benzotriazole anion to occur under kinetically favorable conditions that would otherwise be inaccessible. This mechanistic pathway ensures that even sterically hindered or electronically deactivated substrates, such as p-nitrophenyl or furan derivatives, react efficiently. The ability to tune the catalyst loading between 0.1 and 1.5 equivalents allows process chemists to optimize the reaction rate against cost, providing a flexible tool for diverse substrate scopes ranging from simple alkyl chains to complex heteroaryl systems.
From an impurity control perspective, the mild thermal profile of this reaction is paramount. Acyl azides are known to be potentially unstable and can undergo premature Curtius rearrangement or decomposition if exposed to excessive heat or prolonged reaction times. By completing the conversion within 0.25 to 2 hours at temperatures not exceeding 45°C, the process minimizes the formation of thermal degradation byproducts and urea derivatives that often contaminate batches produced via slower methods. Additionally, the use of green solvents like acetone, ethanol, or ethyl acetate, often in combination with water for the azide source, reduces the environmental footprint compared to traditional chlorinated solvent-heavy protocols. The high selectivity of the Lewis acid system ensures that side reactions, such as hydrolysis of the acyl azide, are kept to a minimum, resulting in a crude product profile that is clean enough for direct crystallization or simple distillation, further validating its suitability for GMP manufacturing environments.
How to Synthesize Acyl Azide Efficiently
To implement this high-efficiency synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric and procedural guidelines to maximize yield and safety. The process begins with the dissolution of the acyl benzotriazole starting material in a selected solvent system, where the solvent volume is carefully calibrated to be between 10 and 35 times the mass of the substrate to ensure adequate mixing and heat transfer. Following this, the Lewis acid catalyst is introduced, and the sodium azide is added either as a solid or in a minimal amount of water, initiating the exothermic substitution reaction. Detailed standardized operating procedures regarding the specific addition rates, temperature ramping, and quenching protocols are essential to maintain control over the reaction kinetics and ensure operator safety when handling azide salts. The comprehensive step-by-step synthesis guide below outlines the exact parameters validated across multiple embodiments in the patent data.
- Dissolve the acyl benzotriazole substrate in a suitable solvent such as acetone, THF, or ethyl acetate, ensuring a solvent volume of 10 to 35 times the mass of the substrate.
- Add the Lewis acid catalyst (e.g., ZnCl2, AlCl3, or CuCl2) and introduce an aqueous or solid solution of sodium azide, maintaining a molar ratio of substrate to azide between 1.0: 1.0 and 1.0:6.0.
- Stir the reaction mixture at a controlled temperature between 0°C and 45°C for 0.25 to 2 hours until TLC indicates complete conversion, followed by extraction and washing to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this Lewis acid-catalyzed protocol translates directly into tangible economic and logistical benefits that strengthen the overall value proposition of the supply base. The elimination of column chromatography is perhaps the most impactful cost driver, as it removes the need for expensive silica gel, large volumes of elution solvents, and the specialized labor required for flash purification. This simplification of the downstream processing significantly reduces the cost of goods sold (COGS) and shortens the batch cycle time, allowing manufacturing facilities to produce more tons of material per year with the same existing infrastructure. Furthermore, the ability to recover and recycle the benzotriazole byproduct, which is achieved with yields often exceeding 90% through simple acidification and extraction, creates a closed-loop material flow that drastically lowers raw material consumption and waste disposal fees.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the use of inexpensive, commodity-grade Lewis acid catalysts such as zinc chloride and aluminum chloride, which are readily available in bulk quantities at a fraction of the cost of precious metal catalysts. By avoiding the use of sensitive acyl chlorides, the process also mitigates the costs associated with specialized corrosion-resistant equipment and hazardous waste treatment for chloride salts. The high atom economy and the ability to use greener, cheaper solvents like acetone and ethyl acetate further contribute to a leaner manufacturing budget, ensuring that the final acyl azide intermediate is priced competitively for downstream API synthesis without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply continuity is significantly improved due to the robustness and simplicity of the reaction conditions, which are less susceptible to variations in raw material quality or minor fluctuations in environmental controls. The short reaction time of 0.25 to 2 hours means that production scheduling becomes far more predictable, reducing the risk of delays caused by extended reaction monitoring or failed batches that require reprocessing. Additionally, the starting materials, acyl benzotriazoles, are stable solids that can be stored for long periods without degradation, unlike liquid acyl chlorides, allowing manufacturers to maintain strategic stockpiles and respond rapidly to surges in demand from pharmaceutical clients without facing raw material spoilage issues.
- Scalability and Environmental Compliance: Scaling this process from gram-scale R&D to multi-ton commercial production is straightforward because the exotherm is manageable and the workup relies on standard unit operations like extraction and distillation rather than complex chromatography. The reduced solvent usage and the ability to recover benzotriazole align perfectly with modern environmental, health, and safety (EHS) regulations, minimizing the generation of hazardous waste streams. This environmental compliance not only reduces regulatory risk but also enhances the corporate sustainability profile of the supply chain, making it an attractive option for multinational corporations striving to meet their green chemistry goals and carbon reduction targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acyl azide synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on catalyst selection, substrate scope, and purification outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines or for procurement specialists assessing the quality standards of potential suppliers.
Q: What are the primary advantages of using acyl benzotriazoles over acyl chlorides for azide synthesis?
A: Acyl benzotriazoles offer superior stability and ease of handling compared to acyl chlorides, which are moisture-sensitive and difficult to store. Furthermore, the byproduct benzotriazole can be easily recovered and recycled, enhancing the overall atom economy and reducing waste disposal costs.
Q: Which Lewis acid catalysts are most effective for this transformation?
A: The patent identifies a broad range of effective Lewis acids including zinc chloride, aluminum chloride, cupric chloride, and iron trichloride. These catalysts are inexpensive, commercially available, and allow the reaction to proceed rapidly at near-room temperature.
Q: Is column chromatography required for purification in this new method?
A: No, one of the most significant breakthroughs of this method is the elimination of column chromatography. The product can be isolated through simple extraction, washing with sodium carbonate and brine, and solvent removal, yielding purities often exceeding 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acyl Azide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new pharmaceutical agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering acyl azides and other complex fine chemicals with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for GMP API synthesis. Our facility is equipped to handle the specific safety requirements of azide chemistry, providing a secure and compliant environment for the manufacture of these energetic intermediates.
We invite you to collaborate with us to leverage this advanced Lewis acid-catalyzed technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that demonstrates how switching to this catalytic route can optimize your specific supply chain economics. We encourage you to contact us today to discuss your requirements,索取 specific COA data for our catalog products, and receive detailed route feasibility assessments tailored to your unique molecular targets. Let us be your trusted partner in driving innovation and efficiency in your chemical supply chain.
