Scalable Synthesis of Octahydro-4a,8-epoxypyrido Azepine Derivatives for Advanced Pharmaceutical Manufacturing
Introduction to Advanced Azepine Synthesis Technology
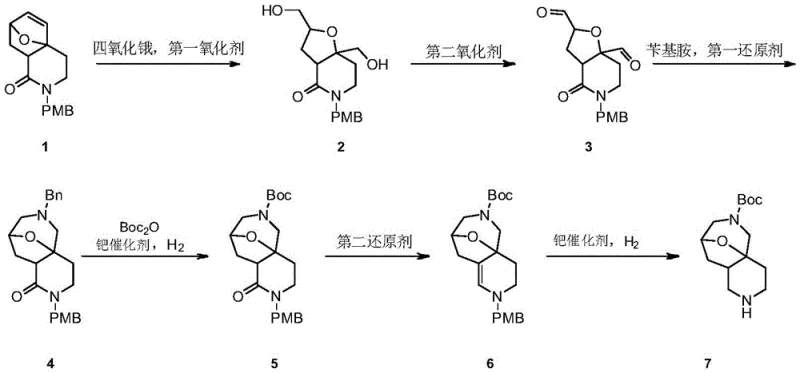
The pharmaceutical industry continuously demands more efficient pathways for constructing complex heterocyclic scaffolds, particularly bridged azepines which serve as critical cores for various bioactive molecules. Patent CN113214291B discloses a groundbreaking six-step synthesis method for octahydro-4a,8-epoxypyrido[4,3-c]azepine-6(5H)-tert-butyl formate, a highly valuable intermediate in medicinal chemistry. This novel approach addresses the longstanding challenges of low overall yields and hazardous reaction conditions associated with traditional routes. By leveraging catalytic osmium tetroxide oxidation and strategic reductive amination cascades, the process achieves a streamlined workflow that is inherently safer and more economically viable for large-scale production. The methodology emphasizes the use of commercially available raw materials and robust reaction conditions, making it an ideal candidate for industrial adoption by forward-thinking chemical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused azepine systems has relied on multi-step sequences involving harsh reagents and unstable intermediates that degrade rapidly upon isolation. Conventional strategies often require stoichiometric amounts of toxic heavy metal oxidants, leading to significant environmental burdens and costly waste disposal protocols. Furthermore, traditional routes frequently suffer from poor stereocontrol, resulting in complex mixtures of diastereomers that are difficult to separate, thereby dragging down the overall process efficiency. The reliance on cryogenic conditions for key cyclization steps in older literature further complicates scale-up, requiring specialized equipment that increases capital expenditure. These factors collectively create a bottleneck for procurement teams seeking reliable sources of high-purity intermediates at competitive price points.
The Novel Approach
In stark contrast, the disclosed invention introduces a rational design that maximizes atom economy and operational simplicity. The route initiates with a highly stereoselective dihydroxylation using catalytic osmium tetroxide regenerated by N-methylmorpholine-N-oxide (NMO), drastically reducing the loading of the toxic metal. Subsequent oxidative cleavage is performed under mild aqueous conditions using sodium periodate, avoiding the need for anhydrous environments that drive up solvent costs. A key innovation lies in the third step, where reductive amination and cyclization occur in a concerted manner, effectively building the complex bridged skeleton in a single operation. This telescoping of reactions not only saves time but also prevents the degradation of sensitive aldehyde intermediates, ensuring a higher throughput of quality material for downstream processing.
Mechanistic Insights into Catalytic Oxidation and Reductive Cyclization
The core of this synthetic strategy relies on the precise manipulation of oxidation states to unlock latent reactivity within the bicyclic precursor. The initial dihydroxylation proceeds via a [3+2] cycloaddition of osmium tetroxide to the alkene, forming an osmate ester that is hydrolyzed to release the vicinal diol. The use of NMO as a co-oxidant is critical here, as it rapidly re-oxidizes the reduced osmium species back to the active Os(VIII) state, allowing the catalyst to turnover hundreds of times. This catalytic cycle ensures that the reaction remains kinetically favorable without accumulating toxic osmium waste. Following this, the glycol cleavage by periodate generates a reactive dialdehyde species in situ, which is immediately poised for nucleophilic attack, demonstrating a sophisticated understanding of transient intermediate stability.
Impurity control is meticulously managed through the selection of specific reducing agents and protecting group strategies. In the reductive amination step, sodium triacetoxyborohydride is employed due to its chemoselectivity; it reduces the iminium ion formed between the aldehyde and benzylamine but leaves other functional groups untouched. This selectivity prevents the formation of over-reduced byproducts that often plague similar transformations. Furthermore, the strategic use of the p-methoxybenzyl (PMB) group on the lactam nitrogen provides orthogonal protection that remains stable during the acidic and basic conditions of the intermediate steps but can be cleanly removed in the final hydrogenation. This orthogonal protection scheme ensures that the final product emerges with a clean impurity profile, minimizing the burden on downstream purification units.
How to Synthesize Octahydro-4a,8-epoxypyrido Azepine Efficiently
The execution of this synthesis requires careful attention to reaction parameters to maximize the reported yields. The process begins with the oxidation of the starting alkene in acetone at ambient temperature, followed by a straightforward workup that allows the crude diol to proceed directly to cleavage. The subsequent oxidative cleavage in methanol-water generates the dialdehyde, which is extracted and concentrated without further purification to preserve yield. The critical cyclization step involves the addition of benzylamine at low temperatures to control exotherms, followed by the slow addition of the reducing agent to ensure complete conversion to the bridged amine.
- Perform catalytic dihydroxylation of the starting alkene using osmium tetroxide and NMO to form the diol intermediate.
- Execute oxidative cleavage of the diol using sodium periodate to generate the dialdehyde species.
- Conduct reductive amination with benzylamine followed by cyclization to construct the bridged azepine core.
- Protect the amine functionality with Boc anhydride while simultaneously removing benzyl groups via catalytic hydrogenation.
- Reduce the lactam carbonyl to a methylene group using lithium aluminum hydride under controlled anhydrous conditions.
- Finalize the synthesis by removing the remaining protecting group via palladium-catalyzed hydrogenation to yield the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers tangible benefits that extend beyond simple chemistry. The reliance on commodity chemicals such as acetone, methanol, and sodium periodate means that the supply chain is not vulnerable to the volatility of exotic reagent markets. The ability to use crude intermediates directly in subsequent steps significantly reduces solvent consumption and waste generation, aligning with modern green chemistry mandates and lowering disposal costs. Moreover, the short reaction times, often completed within 4 to 16 hours per step, enhance equipment utilization rates, allowing manufacturers to produce larger batches in the same timeframe compared to legacy processes.
- Cost Reduction in Manufacturing: The implementation of catalytic rather than stoichiometric oxidation dramatically lowers the raw material cost per kilogram of product. By eliminating the need for expensive chiral auxiliaries or cryogenic cooling for the majority of the sequence, the operational expenditure is significantly reduced. The high overall yield achieved through telescoped steps means less starting material is required to produce the same amount of final API intermediate, directly improving the margin structure for the end user.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent batch-to-batch quality, which is paramount for maintaining regulatory compliance in pharmaceutical supply chains. The use of standard hydrogenation and filtration equipment means that the process can be transferred to multiple contract manufacturing organizations without requiring specialized custom reactors. This flexibility mitigates the risk of supply disruptions caused by equipment bottlenecks or single-source dependencies.
- Scalability and Environmental Compliance: The process avoids the generation of heavy metal sludge associated with stoichiometric oxidants, simplifying wastewater treatment and environmental permitting. The solvents used throughout the synthesis are readily recyclable, further contributing to a sustainable manufacturing footprint. This alignment with environmental, social, and governance (ESG) goals makes the resulting intermediate more attractive to major pharmaceutical companies striving to reduce their Scope 3 emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis pathway. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation to assist technical teams in evaluating feasibility.
Q: What are the key safety advantages of this synthesis route compared to traditional methods?
A: This process utilizes catalytic amounts of osmium tetroxide rather than stoichiometric quantities, significantly reducing toxicity risks. Furthermore, the use of mild oxidants like NMO and sodium periodate avoids hazardous exothermic reactions common in older protocols.
Q: How does this method improve the overall yield for industrial scale-up?
A: By integrating a telescoped three-step sequence with a cumulative yield of over 73%, the route minimizes material loss during isolation. The robust nature of the intermediates allows for direct use of crude products in subsequent steps, enhancing throughput.
Q: Is the final purification process compatible with GMP standards?
A: Yes, the final steps involve standard column chromatography and crystallization techniques using common solvents like methanol and ethyl acetate, ensuring the removal of impurities to meet stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octahydro-4a,8-epoxypyrido Azepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the plant. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of octahydro-4a,8-epoxypyrido azepine derivative meets the highest industry standards. Our commitment to technical excellence allows us to troubleshoot complex synthesis issues rapidly, providing our partners with a secure and dependable source of supply.
We invite you to discuss how our advanced manufacturing capabilities can optimize your specific supply chain requirements. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify how our optimized process can deliver superior value for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
