Advanced Catalytic Oxidation for High-Purity Alpha-Hydroxy Carbonyl Compounds: A Commercial Scale-Up Perspective
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access functionalized building blocks with high efficiency and minimal environmental impact. Patent CN103613478A introduces a groundbreaking synthetic strategy for the preparation of α-hydroxy carbonyl compounds, a critical structural motif prevalent in bioactive natural products and drug candidates. This innovation departs from traditional reliance on precious metal catalysis, instead leveraging a cost-effective cesium carbonate-mediated aerobic oxidation system. The core transformation involves the direct conversion of carbonyl precursors into their corresponding α-hydroxy derivatives under remarkably mild conditions, typically at room temperature and atmospheric pressure. 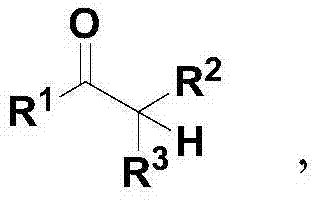 . This shift represents a paradigm change in how we approach C-H functionalization, offering a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing.
. This shift represents a paradigm change in how we approach C-H functionalization, offering a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-hydroxy carbonyl structures has been plagued by significant operational and economic hurdles. Conventional literature methods often rely heavily on expensive and complex palladium-based catalysts, which introduce severe cost pressures and supply chain vulnerabilities due to the volatility of precious metal markets. Furthermore, these traditional protocols frequently necessitate harsh reaction conditions, such as cryogenic temperatures or elevated heating, which demand energy-intensive infrastructure and specialized reactor setups. The substrate scope in many legacy methods is also notoriously narrow, failing to accommodate sterically hindered or electronically diverse molecules without significant yield penalties. Additionally, the use of stoichiometric oxidants in older pathways often generates substantial quantities of toxic waste, complicating disposal and increasing the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN103613478A utilizes a readily available inorganic base, cesium carbonate, coupled with organophosphorus reagents like triphenylphosphine or triethyl phosphite. This combination facilitates an efficient aerobic oxidation that proceeds smoothly at ambient temperatures, typically around 25°C. The elimination of precious metals drastically reduces the raw material costs and removes the need for rigorous heavy metal removal steps downstream. The reaction tolerates a wide array of solvents, with dimethyl sulfoxide (DMSO) proving particularly effective, ensuring high solubility for diverse substrates. By employing molecular oxygen or even ambient air as the oxidant, the process achieves high atom economy, producing water as the benign byproduct. This approach not only simplifies the operational workflow but also enhances the safety profile by avoiding hazardous oxidizing agents.
Mechanistic Insights into Cs2CO3-Catalyzed Aerobic Oxidation
The mechanistic elegance of this transformation lies in the synergistic interaction between the base catalyst and the organophosphorus additive. Cesium carbonate acts as a mild yet effective base to deprotonate the α-position of the carbonyl substrate, generating a nucleophilic enolate species. Simultaneously, the organophosphorus compound likely serves as an oxygen transfer mediator or stabilizes reactive intermediates, facilitating the insertion of oxygen from the aerobic environment into the carbon framework. This cooperative catalysis ensures that the reaction proceeds with high selectivity, minimizing over-oxidation or side reactions that could compromise product purity. The mild basicity of cesium carbonate is crucial, as it prevents the decomposition of sensitive functional groups that might be present in complex pharmaceutical intermediates. 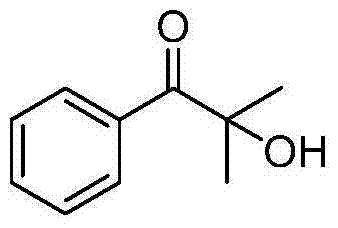 . The result is a clean reaction profile that yields high-purity products, often exceeding 80-90% isolated yield in laboratory settings, as evidenced by the extensive experimental data provided in the patent.
. The result is a clean reaction profile that yields high-purity products, often exceeding 80-90% isolated yield in laboratory settings, as evidenced by the extensive experimental data provided in the patent.
Impurity control is another critical aspect where this method excels. Traditional strong base or metal-catalyzed routes often lead to polymerization or aldol condensation byproducts, especially with enolizable ketones. However, the specific conditions outlined in this patent—utilizing polar aprotic solvents like DMSO and controlled oxygen flow—suppress these competing pathways. The reaction kinetics are tuned such that the desired hydroxylation occurs faster than potential degradation reactions. For instance, cyclic substrates such as cyclohexyl benzophenone derivatives undergo smooth conversion to their α-hydroxy counterparts without ring opening or rearrangement. 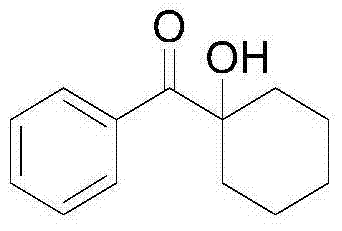 . This level of chemoselectivity is paramount for R&D teams aiming to synthesize complex drug candidates where structural integrity is non-negotiable. The ability to achieve such high purity directly from the reaction mixture significantly reduces the burden on purification processes, thereby enhancing overall process efficiency.
. This level of chemoselectivity is paramount for R&D teams aiming to synthesize complex drug candidates where structural integrity is non-negotiable. The ability to achieve such high purity directly from the reaction mixture significantly reduces the burden on purification processes, thereby enhancing overall process efficiency.
How to Synthesize Alpha-Hydroxy Carbonyl Compounds Efficiently
Implementing this synthetic route in a production environment requires adherence to specific operational parameters to maximize yield and safety. The process begins with the precise weighing of the carbonyl substrate and the organophosphorus reagent, which are then dissolved in the chosen solvent, preferably DMSO for optimal performance. The addition of cesium carbonate initiates the catalytic cycle, and the introduction of oxygen is managed carefully to maintain a steady supply of the oxidant without creating explosive mixtures. While laboratory examples utilize Schlenk tubes and oxygen balloons, industrial scale-up would employ standard stirred tank reactors equipped with spargers for efficient gas-liquid mass transfer. The reaction progress is monitored via standard analytical techniques such as HPLC or TLC until the starting material is fully consumed.
- Mix the carbonyl substrate with an organophosphorus compound (such as triethyl phosphite or triphenylphosphine) in a polar aprotic solvent like DMSO.
- Add cesium carbonate as the catalyst and introduce oxygen or air into the reaction system, maintaining stirring at room temperature (25°C).
- Upon completion, quench the reaction with ethyl acetate, wash with brine, separate the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing scarce and expensive palladium catalysts with abundant cesium carbonate and common phosphorus reagents, companies can mitigate the risks associated with geopolitical instability affecting precious metal prices. This substitution leads to a more predictable and stable cost structure for the final intermediate. Furthermore, the use of air or oxygen as the oxidant eliminates the need to procure, store, and handle hazardous chemical oxidants, reducing both logistical complexity and regulatory compliance costs related to hazardous material transport.
- Cost Reduction in Manufacturing: The economic implications of switching to this cesium carbonate-catalyzed system are profound. The removal of precious metal catalysts eliminates a major cost driver, while the mild reaction conditions significantly lower energy consumption by removing the need for heating or cooling systems. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces solvent usage and labor time. These factors collectively contribute to a substantially lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commodity chemicals that are widely available from multiple global suppliers. Unlike specialized ligands or catalysts that may have long lead times, cesium carbonate and triethyl phosphite are standard inventory items for most chemical distributors. This availability ensures that production schedules are not disrupted by raw material shortages. Moreover, the robustness of the reaction means that minor variations in reagent quality do not critically impact the outcome, providing a buffer against supply chain fluctuations and ensuring consistent delivery timelines to downstream customers.
- Scalability and Environmental Compliance: From a scalability perspective, the reaction's simplicity is its greatest asset. The absence of exothermic hazards associated with strong oxidants and the operation at near-ambient temperatures make the transition from kilogram to tonne scale straightforward. The process aligns well with modern environmental, health, and safety (EHS) standards by minimizing waste generation and avoiding toxic heavy metals. This 'green' profile facilitates easier permitting for new manufacturing lines and enhances the corporate sustainability narrative, which is increasingly important for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aerobic oxidation technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using Cesium Carbonate over Palladium catalysts for this transformation?
A: Cesium carbonate offers a significant cost advantage as a non-precious metal base compared to expensive palladium complexes. Furthermore, it operates effectively under mild aerobic conditions without requiring stringent inert atmospheres or extreme temperatures, simplifying the overall process safety and equipment requirements.
Q: Does this synthetic method support a broad range of substrate structures?
A: Yes, the methodology demonstrates exceptional substrate tolerance. It is effective for acyclic ketones, cyclic ketones, aromatic ketones, and even heterocyclic substrates. The mild reaction conditions prevent the degradation of sensitive functional groups often found in complex pharmaceutical intermediates.
Q: How does this process impact environmental compliance and waste generation?
A: By utilizing molecular oxygen or air as the terminal oxidant, the process generates water as the primary byproduct rather than stoichiometric amounts of heavy metal waste or toxic oxidants. This aligns with green chemistry principles and reduces the burden on wastewater treatment facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Carbonyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN103613478A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this cesium carbonate-catalyzed route are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-hydroxy carbonyl compounds meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of complex intermediates or process optimization for existing product lines, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with NINGBO INNO PHARMCHEM can drive efficiency and reliability in your supply chain.
