Advanced Manufacturing of 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxylic Acid Intermediates
The pharmaceutical industry's relentless pursuit of effective anti-obesity therapeutics has placed significant demand on the supply chain for high-quality intermediates, specifically for cannabinoid receptor-1 (CBR1) blockers like Rimonabant Hydrochloride. A pivotal breakthrough in this domain is detailed in patent CN101456843A, which outlines a robust and industrially viable synthesis for 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxylic acid. This specific intermediate is critical for the final assembly of the active pharmaceutical ingredient, and its production efficiency directly impacts the cost and availability of the downstream medication. The disclosed methodology represents a paradigm shift from traditional multi-step sequences, offering a streamlined pathway that achieves a remarkable total yield of 60.9% and a product purity exceeding 99.5%. For global procurement teams and R&D directors, this patent data signals a move towards more sustainable and economically feasible manufacturing protocols that eliminate the bottlenecks of cryogenic reactions and expensive transition metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this complex pyrazole derivative has been plagued by inefficient methodologies that struggle to meet the rigorous demands of commercial scale-up. As illustrated in the reaction schemes of prior art, Method 1 relies on an alpha-bromination followed by condensation with ethyl acetoacetate, a process that necessitates cumbersome column chromatography purification and results in a dismal total yield of less than 20%. Furthermore, Method 2 introduces severe operational hazards by requiring ultra-low temperature conditions at -78°C and the use of costly reagents like lithium hexamethyldisilazide, capping the yield at under 30%. Other approaches, such as Method 4, depend on palladium-catalyzed cross-coupling reactions which not only inflate raw material costs due to the precious metal but also introduce complex heavy metal removal steps that complicate regulatory compliance. These legacy processes often generate oily intermediates that are notoriously difficult to purify, leading to inconsistent quality and unpredictable production timelines.
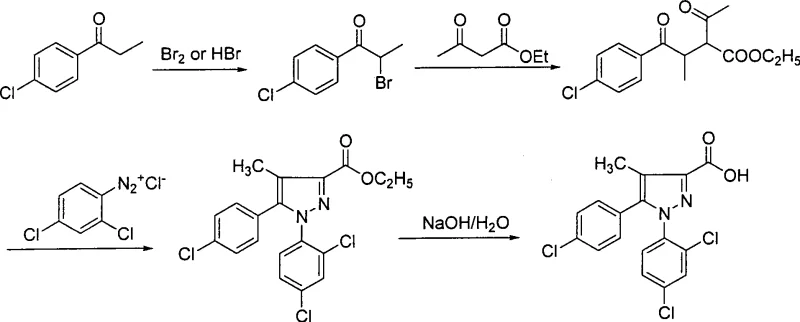
The Novel Approach
In stark contrast to these convoluted pathways, the novel approach leverages a clever strategy involving the formation of a stable furandione intermediate to drive the reaction forward with high efficiency. By reacting 4-chloropropiophenone with trimethylchlorosilane to form an enol silyl ether, followed by cyclization with oxalyl chloride, the process generates 5-(4-chlorophenyl)-4-methyl-2,3-furandione as a distinct, filterable solid. This intermediate serves as a high-quality building block that reacts cleanly with 2,4-dichlorophenylhydrazine under mild acid catalysis to close the pyrazole ring. The elimination of cryogenic requirements and the avoidance of transition metals simplify the reactor setup significantly, allowing for operation in standard glass-lined or stainless steel vessels. This route not only improves the mass balance of the reaction but also ensures that the final carboxylic acid product can be isolated with exceptional purity through simple recrystallization, making it ideally suited for reliable pharmaceutical intermediate supplier operations.
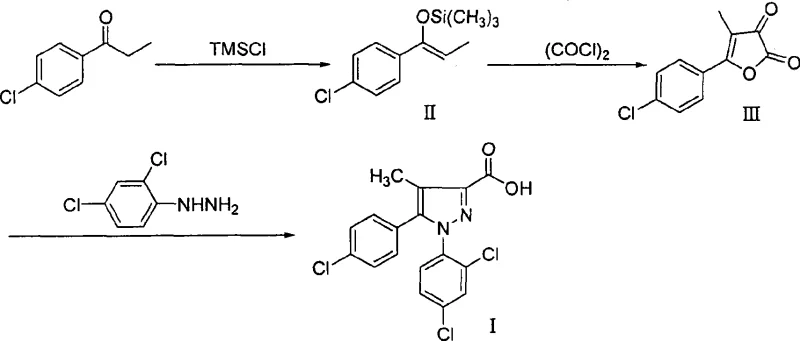
Mechanistic Insights into Enol Silyl Ether Mediated Cyclization
The core innovation of this synthesis lies in the strategic use of silyl enol ether chemistry to control regioselectivity and reactivity during the acylation step. When 4-chloropropiophenone is treated with trimethylchlorosilane in the presence of a base like triethylamine and a catalyst such as sodium iodide, it forms the corresponding enol silyl ether. This species acts as a masked enolate that is sufficiently nucleophilic to attack oxalyl chloride but stable enough to be handled without the extreme precautions required for lithiated species. The subsequent reaction with oxalyl chloride proceeds through an acylation mechanism that spontaneously cyclizes to form the furan-2,3-dione ring system. This cyclization is driven by the thermodynamic stability of the resulting heterocyclic dione and the expulsion of volatile byproducts, which helps push the equilibrium towards completion. The result is a highly crystalline intermediate with a sharp melting point range of 162.9-163.5°C, indicating a high degree of structural order and purity that is absent in the amorphous oils produced by alternative methods.
From an impurity control perspective, this mechanistic pathway offers distinct advantages for maintaining a clean profile throughout the manufacturing campaign. Because the furandione intermediate precipitates out of the reaction mixture as a solid, it can be physically separated from soluble organic impurities and unreacted starting materials via simple filtration and washing. This solid-state purification step acts as a powerful gatekeeper, preventing the carryover of contaminants into the final ring-closing step with the hydrazine. In the final condensation, the use of catalytic acids like p-toluenesulfonic acid facilitates the dehydration and aromatization of the pyrazole ring without promoting excessive side reactions or decomposition. The ability to isolate stable intermediates at each stage allows for rigorous in-process control testing, ensuring that any deviations are caught early before they impact the final batch quality, thereby guaranteeing the stringent purity specifications required for API manufacturing.
How to Synthesize 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxylic acid Efficiently
The implementation of this synthesis protocol requires careful attention to solvent selection and stoichiometry to maximize the benefits of the furandione route. The process begins with the silylation of the ketone in a polar aprotic solvent like acetonitrile, followed by the crucial cyclization step in a non-polar solvent such as diethyl ether or toluene to facilitate the precipitation of the furandione. The final condensation is typically performed in toluene with a catalytic amount of acid to drive the ring closure to completion. Detailed standard operating procedures regarding temperature profiles, addition rates, and workup techniques are essential for reproducing the reported 60.9% yield on a large scale. Operators must ensure that moisture is controlled during the silylation step to prevent hydrolysis of the silyl enol ether, while the final hydrolysis and isolation steps require precise pH adjustment to ensure maximum recovery of the carboxylic acid product.
- React 4-chloropropiophenone with trimethylchlorosilane (TMSCl) to generate the enol silyl ether intermediate.
- Cyclize the enol silyl ether with oxalyl chloride in a non-polar solvent to obtain the stable 5-(4-chlorophenyl)-4-methyl-2,3-furandione.
- Condense the furandione with 2,4-dichlorophenylhydrazine under acid catalysis to form the final pyrazole carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible economic and logistical benefits that extend far beyond simple yield improvements. By replacing expensive and hazardous reagents with commodity chemicals, the direct material cost of goods sold is significantly reduced, enhancing the overall margin profile of the intermediate. The robustness of the process, characterized by the formation of solid, filterable intermediates rather than difficult-to-handle oils, minimizes the risk of batch failures and reduces the time required for purification and quality control testing. This reliability ensures a more consistent supply of high-purity pharmaceutical intermediates, mitigating the risks of production delays that can ripple through the entire drug development timeline. Furthermore, the simplified waste stream, which avoids heavy metal contamination and complex organic residues, lowers the environmental compliance burden and associated disposal costs.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts and cryogenic cooling systems removes two of the most significant cost drivers in fine chemical synthesis. Without the need for expensive ligands or specialized low-temperature reactors, capital expenditure and operational expenses are drastically lowered. Additionally, the high atom economy of the furandione route means less raw material is wasted, further contributing to substantial cost savings in large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 4-chloropropiophenone and oxalyl chloride ensures that the supply chain is not vulnerable to the shortages often associated with specialized reagents. The ability to produce stable intermediates that can be stored or transported if necessary adds a layer of flexibility to the manufacturing schedule, allowing for better inventory management and responsiveness to fluctuating market demand for the final API.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like filtration, distillation, and crystallization that are easily transferred from the laboratory to pilot and commercial plants. The closed-loop treatment of acid gases and the recovery of solvents like toluene and acetonitrile demonstrate a commitment to green chemistry principles, reducing the environmental footprint and ensuring compliance with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this key pharmaceutical intermediate. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their broader supply networks.
Q: What is the total yield of the novel furandione route compared to conventional methods?
A: The novel route described in patent CN101456843A achieves a total yield of 60.9%, significantly outperforming conventional methods which often yield less than 20% to 40% due to purification losses and harsh conditions.
Q: How does this process improve impurity control for pharmaceutical grade intermediates?
A: The process generates a stable solid furandione intermediate (melting point 162.9-163.5°C) that can be purified by simple filtration, avoiding the difficult-to-purify oily intermediates common in other synthetic routes.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, unlike palladium-catalyzed cross-coupling methods, this route utilizes inexpensive reagents like oxalyl chloride and trimethylchlorosilane, drastically reducing raw material costs and eliminating heavy metal residue concerns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medications depends on the availability of high-quality, cost-effective intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major pharmaceutical partners without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxylic acid meets the exacting standards required for GMP manufacturing. Our commitment to process optimization allows us to deliver this critical building block with the consistency and reliability that global supply chains demand.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your development timelines and quality targets.
