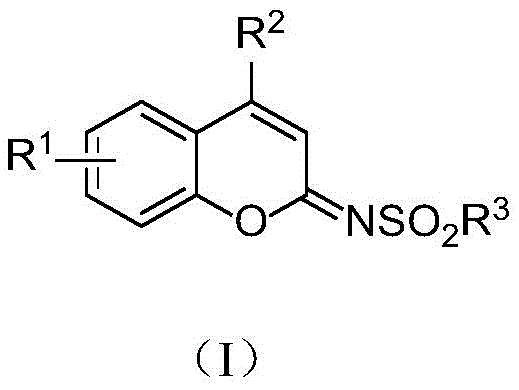
Advanced Copper-Catalyzed Synthesis of N-Sulfonyl Imine Coumarin Derivatives for Pharmaceutical Applications
Advanced Copper-Catalyzed Synthesis of N-Sulfonyl Imine Coumarin Derivatives for Pharmaceutical Applications
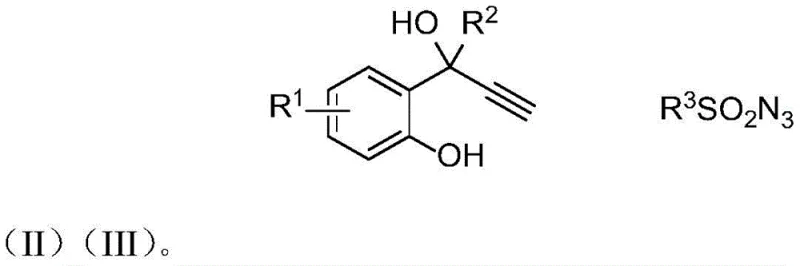
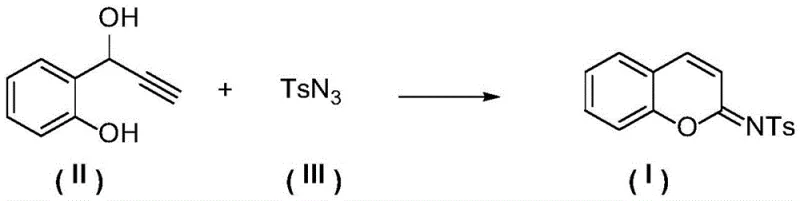
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and agrochemical industries, particularly regarding the efficient production of heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN112442005B, which discloses a novel preparation method for N-sulfonyl imine coumarin derivatives. These compounds are not merely academic curiosities; they represent a critical class of intermediates with potent biological activities, including anticoagulant, antitumor, and antibacterial properties. The patent outlines a robust synthetic strategy that leverages click chemistry principles to construct the coumarin core with exceptional efficiency. By utilizing a copper-catalyzed cascade reaction involving 2-(1-hydroxy-2-propyn-1-yl)aryl phenols and sulfonyl azides, this technology offers a streamlined pathway to high-value chemical building blocks. For R&D directors and procurement specialists alike, understanding the nuances of this methodology is essential for securing reliable supply chains and optimizing manufacturing costs in the production of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of functionalized coumarin derivatives has often relied on classical condensation reactions such as the Pechmann condensation or Knoevenagel condensation. While these methods are well-established, they frequently suffer from significant drawbacks that hinder their utility in modern, high-throughput drug discovery and process chemistry. Conventional routes often require harsh acidic or basic conditions, elevated temperatures, and prolonged reaction times, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, many traditional strategies involve multi-step sequences to introduce specific substituents at the 3- or 4-positions of the coumarin ring, resulting in lower overall yields and increased waste generation. The reliance on stoichiometric amounts of reagents and the generation of hazardous byproducts also pose substantial environmental and safety challenges, complicating the regulatory approval process for new drug applications. These inefficiencies create bottlenecks in the supply chain, driving up the cost of goods and extending lead times for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN112442005B introduces a transformative one-pot synthetic route that dramatically simplifies the construction of the N-sulfonyl imine coumarin scaffold. This novel approach utilizes a copper-catalyzed tandem reaction that seamlessly integrates cycloaddition, ring-opening rearrangement, nucleophilic addition, and dehydration into a single operational step. By reacting readily available 2-(1-hydroxy-2-propyn-1-yl)aryl phenols with sulfonyl azides in the presence of a copper catalyst and an amine ligand, the process achieves high atom economy and generates harmless byproducts, primarily nitrogen gas. The mild reaction conditions, typically ranging from room temperature to moderate heating, preserve the integrity of diverse functional groups, allowing for the synthesis of a broad library of derivatives with varying electronic and steric properties. This methodological shift not only enhances the purity of the final product but also significantly reduces the operational complexity, making it an ideal candidate for both laboratory-scale optimization and industrial manufacturing.
Mechanistic Insights into Copper-Catalyzed Cyclization and Rearrangement
The success of this synthetic transformation hinges on the intricate interplay between the copper catalyst, the alkyne substrate, and the sulfonyl azide reagent. The mechanism is believed to initiate with the activation of the terminal alkyne by the copper(I) species, forming a copper-acetylide intermediate. This activated species then undergoes a [3+2] cycloaddition with the sulfonyl azide, a classic click chemistry reaction, to generate a triazole intermediate. However, unlike standard CuAAC reactions that stop at the triazole, this system is designed to proceed further. The proximal phenolic hydroxyl group acts as an internal nucleophile, attacking the electron-deficient centers within the transient triazole or ketenimine species formed upon nitrogen extrusion. This triggers a cascade of ring-opening and ring-closing events, ultimately leading to the formation of the stable coumarin imine structure. The presence of the amine ligand is crucial in stabilizing the copper center and modulating its Lewis acidity, ensuring that the reaction proceeds smoothly without the precipitation of inactive copper species or the formation of side products such as Glaser coupling dimers.
From an impurity control perspective, this mechanism offers distinct advantages. The intramolecular nature of the cyclization step ensures high regioselectivity, minimizing the formation of isomeric byproducts that are difficult to separate. Moreover, the driving force of nitrogen gas evolution renders the initial cycloaddition step essentially irreversible, pushing the equilibrium towards the desired product. The choice of solvent, particularly dichloromethane as highlighted in the patent examples, plays a pivotal role in solubilizing the polar intermediates while maintaining the stability of the catalyst system. Detailed analysis of the reaction profile suggests that the dehydration step, which finalizes the aromatic coumarin system, is facilitated by the thermal energy provided during the reaction or by the inherent acidity of the reaction medium. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters such as temperature and stoichiometry to maximize yield and purity, ensuring that the final API intermediate meets stringent quality specifications required for clinical trials.
How to Synthesize N-Sulfonyl Imine Coumarin Efficiently
Implementing this synthesis in a practical setting requires careful attention to the molar ratios of the reactants and the choice of catalytic system. The patent data demonstrates that a molar ratio of the propargylic phenol to the sulfonyl azide between 1:1 and 1:3 is optimal, with a slight excess of the azide often driving the reaction to completion. The catalyst loading is remarkably low, with effective turnover observed at ratios as low as 1:0.05 (substrate to copper), although slightly higher loadings may be used to accelerate the kinetics. The reaction is typically conducted in dichloromethane, though other solvents like chloroform or THF can be employed depending on the solubility of the specific substrates. Post-reaction workup is straightforward, involving simple solvent removal followed by purification via silica gel chromatography using standard ethyl acetate and petroleum ether mixtures. This simplicity underscores the practicality of the method for generating diverse libraries of compounds for biological screening.
- Prepare the reaction mixture by combining 2-(1-hydroxy-2-propyn-1-yl)aryl phenol, sulfonyl azide, copper catalyst (e.g., CuI), and amine ligand in an organic solvent like dichloromethane.
- Stir the mixture at temperatures ranging from 25°C to 120°C for 1 to 24 hours to facilitate cycloaddition and rearrangement.
- Upon completion, cool the system, remove the solvent, and purify the crude residue via silica gel column chromatography to isolate the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the manufacturing process. By consolidating multiple synthetic steps into a single pot, the method eliminates the need for intermediate isolation, drying, and purification stages, which are traditionally resource-intensive and time-consuming. This reduction in unit operations directly correlates to a significant decrease in labor costs, equipment usage time, and energy consumption. Furthermore, the use of inexpensive and commercially available starting materials, such as simple aryl phenols and sulfonyl azides, ensures a stable and cost-effective raw material supply base. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, enhancing batch-to-b consistency and reducing the risk of costly production failures.
- Cost Reduction in Manufacturing: The elimination of transition metal scavenging steps, often required when using palladium or other precious metals, represents a major cost saving. Since the copper catalyst used here is inexpensive and the reaction generates harmless nitrogen gas as a byproduct, the waste treatment costs are substantially lowered. The high atom economy ensures that a greater proportion of the raw material mass ends up in the final product, minimizing waste disposal fees and maximizing material efficiency. Additionally, the mild conditions reduce the energy burden on the facility, as there is no need for extreme heating or cryogenic cooling, further contributing to a leaner cost structure for the production of these high-value intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like cuprous iodide and triethylamine mitigates the risk of supply chain disruptions associated with specialized or scarce reagents. The scalability of the reaction has been demonstrated across various substrate scopes, indicating that the process can be reliably transferred from gram-scale laboratory synthesis to kilogram or ton-scale commercial production without significant re-engineering. This scalability ensures that suppliers can meet fluctuating demand from pharmaceutical clients without long lead times. The simplicity of the workup procedure also means that production throughput can be increased, allowing for faster turnaround times on custom synthesis orders and improving overall supply chain agility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The generation of nitrogen gas as the primary byproduct avoids the accumulation of toxic heavy metal waste or hazardous organic sludge. The use of dichloromethane, while requiring proper handling, is a standard solvent with well-established recovery and recycling protocols in modern chemical plants. The high purity of the crude product reduces the solvent volume required for recrystallization or chromatography, lowering the overall solvent footprint. These factors collectively simplify the regulatory compliance process, facilitating faster approvals for new manufacturing sites and ensuring long-term sustainability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the operational parameters and expected outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this copper-catalyzed method over traditional coumarin synthesis?
A: This method utilizes mild reaction conditions and a one-pot procedure, eliminating the need for harsh reagents and complex multi-step sequences often found in conventional routes, thereby improving atom economy and operational simplicity.
Q: Which catalyst and ligand system provides the optimal yield for this transformation?
A: Experimental data indicates that Cuprous Iodide (CuI) combined with Triethylamine (Et3N) as the ligand delivers superior catalytic performance and highest product yields compared to other copper salts or nitrogenous ligands.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process features simple post-treatment procedures such as evaporation and standard column chromatography, along with high atom economy and harmless byproducts, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Sulfonyl Imine Coumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medicines. Our technical team has extensively analyzed the potential of the copper-catalyzed synthesis described in CN112442005B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from discovery to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-sulfonyl imine coumarin delivered meets the highest industry standards. We understand that consistency and reliability are paramount in the pharmaceutical supply chain, and we are committed to being a partner you can trust.
We invite you to discuss how this advanced synthetic route can optimize your specific project requirements. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and timeline. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to initiate a discussion on route feasibility assessments for your proprietary targets. Let us help you accelerate your drug development pipeline with efficient, scalable, and cost-effective chemical solutions.
