Advanced Aqueous Synthesis of Diazinium Derivatives for High-Purity Pharmaceutical Intermediates
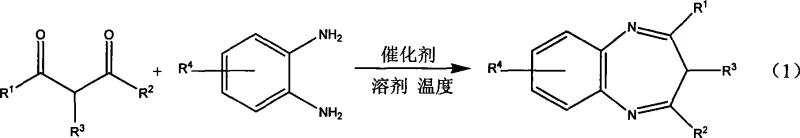
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient synthetic routes to critical heterocyclic scaffolds. Patent CN101372477A introduces a groundbreaking methodology for the synthesis of diazinium derivatives, a class of compounds with significant biological activity and utility in drug discovery. This innovation leverages phosphotungstic acid as a heterogeneous catalyst within a purely aqueous medium, marking a significant departure from traditional solvent-intensive processes. By utilizing water as the sole solvent, this technology addresses growing regulatory pressures regarding volatile organic compound (VOC) emissions while maintaining high reaction efficiency. For R&D directors and process chemists, this represents a viable pathway to streamline the production of complex nitrogen-containing heterocycles without compromising on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of diazinium rings has relied heavily on harsh reaction conditions that pose significant safety and environmental challenges. Conventional protocols often necessitate the use of phosphorus oxychloride (POCl3) under reflux conditions or microwave irradiation in organic solvents. These methods are fraught with drawbacks, including the generation of corrosive acidic waste streams and the requirement for specialized equipment to handle hazardous reagents safely. Furthermore, the reliance on organic solvents contributes to substantial operational costs related to solvent recovery and waste disposal, while the high thermal energy input can lead to substrate decomposition and lower overall selectivity. Such inefficiencies create bottlenecks in the supply chain, particularly when scaling up for commercial API manufacturing where safety and consistency are paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a benign aqueous system catalyzed by phosphotungstic acid, offering a sustainable alternative that aligns with modern green chemistry principles. This method allows for the condensation of dicarbonyl compounds with substituted o-phenylenediamines under mild thermal conditions, typically ranging from 5°C to 60°C. The use of water not only eliminates fire hazards associated with flammable organic solvents but also simplifies the workup procedure, as the product can often be extracted directly without complex quenching steps. This shift to an aqueous paradigm significantly reduces the environmental footprint of the synthesis while providing a robust platform for generating diverse diazinium libraries.
Mechanistic Insights into Phosphotungstic Acid-Catalyzed Cyclization
The efficacy of this synthesis lies in the unique properties of phosphotungstic acid (H3PW12O40), a Keggin-type heteropolyacid that exhibits strong Brønsted acidity and oxidative stability. In the aqueous medium, the catalyst facilitates the activation of the carbonyl groups in the dicarbonyl substrate, promoting nucleophilic attack by the amine groups of the o-phenylenediamine. This dual activation lowers the energy barrier for the cyclization step, allowing the reaction to proceed rapidly even at room temperature. The hydrophobic effect in water may also play a role, effectively concentrating the organic reactants in the vicinity of the catalyst clusters, thereby enhancing reaction rates and selectivity compared to homogeneous organic phases.
From an impurity control perspective, the mildness of the aqueous acidic environment is crucial for maintaining product integrity. Unlike harsh dehydrating agents like POCl3 which can induce side reactions such as chlorination or polymerization, phosphotungstic acid promotes clean condensation. The reaction conditions minimize the formation of tarry by-products, resulting in a cleaner crude profile that simplifies downstream purification. This mechanistic cleanliness is vital for pharmaceutical applications, where strict limits on genotoxic impurities and heavy metals must be adhered to, ensuring the final diazinium intermediate meets rigorous quality specifications for subsequent drug synthesis steps.
How to Synthesize Diazinium Derivatives Efficiently
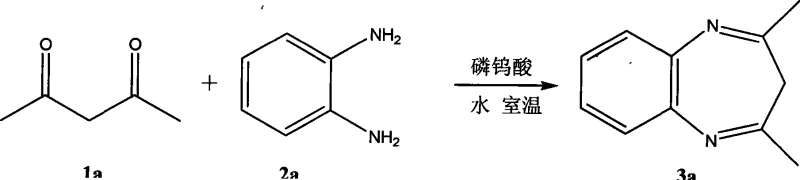
Implementing this synthesis protocol requires precise control over stoichiometry and reaction parameters to maximize yield and reproducibility. The process begins with the preparation of an aqueous slurry containing the dicarbonyl compound and the substituted o-phenylenediamine in a molar ratio of approximately 1:1.1. A catalytic amount of phosphotungstic acid, typically between 0.005 and 0.015 equivalents relative to the substrate, is then introduced to initiate the transformation. The mixture is stirred vigorously to ensure adequate mass transfer between the organic substrates and the aqueous catalyst phase. Temperature control is flexible, allowing operators to tune the reaction rate from 5°C for sensitive substrates up to 60°C for faster throughput, with reaction times varying from 2 to 12 hours depending on the specific electronic nature of the substituents.
- Mix dicarbonyl compound and substituted o-phenylenediamine in pure water with 0.005-0.015 equivalents of phosphotungstic acid catalyst.
- Stir the reaction mixture at temperatures between 5°C and 60°C for 2 to 12 hours depending on substrate reactivity.
- Extract the product with ethyl acetate, wash with saturated sodium carbonate and chloride solutions, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this aqueous catalytic system offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive and hazardous organic solvents drastically reduces raw material costs and mitigates the risks associated with solvent price volatility and supply disruptions. Furthermore, the simplified workup procedure, which avoids complex neutralization of corrosive reagents, translates into reduced labor hours and lower utility consumption per batch. These operational efficiencies contribute to a more resilient supply chain capable of responding quickly to market demands for key pharmaceutical intermediates without the burden of excessive waste management overheads.
- Cost Reduction in Manufacturing: The replacement of toxic organic solvents and harsh reagents with water and a reusable heteropolyacid catalyst leads to significant cost savings in waste treatment and raw material procurement. By avoiding the use of phosphorus oxychloride, facilities eliminate the need for specialized corrosion-resistant equipment and costly scrubbing systems for acidic off-gases. Additionally, the high atom economy of the condensation reaction ensures that a greater proportion of input materials are converted into valuable product, minimizing material loss and maximizing return on investment for large-scale production runs.
- Enhanced Supply Chain Reliability: Utilizing water as the primary reaction medium removes dependencies on petrochemical-derived solvents, the supply of which can be subject to geopolitical and market fluctuations. The raw materials required, specifically dicarbonyls and phenylenediamines, are commodity chemicals with stable global supply chains, ensuring consistent availability for continuous manufacturing operations. This stability allows for better long-term planning and inventory management, reducing the risk of production stoppages due to raw material shortages and enabling more reliable delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The inherent safety of running exothermic reactions in water makes this process highly scalable from laboratory benchtop to multi-ton industrial reactors without the need for extensive re-engineering. The absence of flammable solvents lowers insurance premiums and simplifies regulatory compliance regarding fire safety and environmental discharge permits. As global regulations on VOC emissions tighten, adopting this water-based technology positions manufacturers as leaders in sustainability, facilitating easier approval processes for new facilities and enhancing the corporate reputation among environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this phosphotungstic acid-catalyzed synthesis. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process robustness and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this methodology into existing production workflows or developing new synthetic routes for diazinium-based active ingredients.
Q: What are the advantages of using phosphotungstic acid over traditional catalysts?
A: Phosphotungstic acid acts as a robust heteropolyacid catalyst that functions efficiently in pure water, eliminating the need for toxic organic solvents like phosphorus oxychloride and reducing environmental hazards.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method utilizes water as a solvent and mild temperatures (5°C-60°C), making it inherently safer and easier to scale compared to refluxing microwave methods or hazardous solvent systems.
Q: What yields can be expected with this aqueous protocol?
A: Experimental data indicates high yields ranging from 72% to 94% depending on the specific substrates and reaction temperature, demonstrating excellent efficiency for pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazinium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity diazinium intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against exacting standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced aqueous synthesis can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in delivering high-quality chemical solutions.
