Advanced Photocatalytic Synthesis of 1,4-Dihydronaphthalene Structures for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex polycyclic scaffolds, particularly the 1,4-dihydronaphthalene backbone which is prevalent in bioactive natural products and lignin derivatives. A significant breakthrough in this domain is detailed in Chinese Patent CN116396315A, which discloses a novel synthetic strategy utilizing visible light photocatalysis to construct these valuable structures. This technology represents a paradigm shift from traditional thermal or stoichiometric metal-mediated processes, offering a greener and more operationally simple pathway. By leveraging an iridium-based photocatalyst in conjunction with N-heterocyclic carbene boranes, the method achieves high efficiency under remarkably mild conditions. For R&D directors and process chemists, this patent provides a critical blueprint for modernizing the production of high-purity pharmaceutical intermediates, ensuring that supply chains can meet the rigorous demands of modern drug discovery without the baggage of hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,4-dihydronaphthalene skeleton has been fraught with significant chemical and economic challenges. Traditional approaches often rely on precious transition metals that are not only expensive but also pose severe toxicity and environmental disposal issues. For instance, earlier methodologies disclosed by Mark D. Winemiller utilized osmium complexes which require harsh reaction conditions and generate toxic waste streams. As illustrated in the reaction scheme below, these routes involve multi-step sequences including oxidative decomposition, leading to overall yields that fluctuate widely between 40% and 75%, which is suboptimal for large-scale manufacturing.
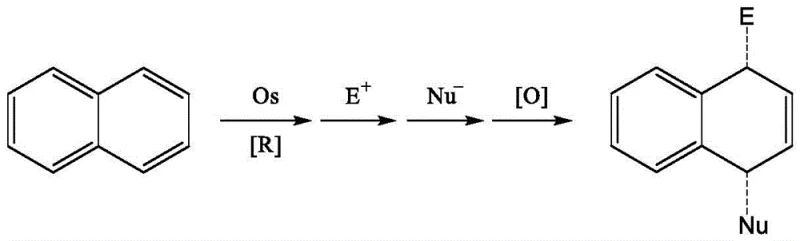
Furthermore, alternative platinum-catalyzed routes, while effective for specific substrates, often demand elevated temperatures around 80°C and strictly anhydrous conditions, limiting their utility for heat-sensitive functional groups. These conventional methods typically suffer from narrow substrate scope, meaning that slight modifications to the starting naphthalene derivative can cause the reaction to fail completely. From a procurement perspective, the reliance on stoichiometric amounts of heavy metals like osmium or specialized platinum promoters drives up the cost of goods sold (COGS) and complicates the regulatory approval process due to residual metal limits in final API products.
The Novel Approach
In stark contrast, the methodology described in CN116396315A introduces a transformative visible-light-driven protocol that circumvents these historical bottlenecks. The core innovation lies in the use of an iridium photocatalyst, specifically Ir(dF(CF3)ppy)2(dtbbpy)PF6, which activates N-heterocyclic carbene boranes to facilitate a hydrogen atom transfer (HAT) mechanism. This approach allows the reaction to proceed at near-room temperature, typically between 25°C and 45°C, drastically reducing energy consumption compared to thermal methods. The reaction scheme below highlights the elegance of this transformation, where simple 2-naphthalene derivatives are directly converted into complex 1,4-dihydronaphthalene structures in a single pot.
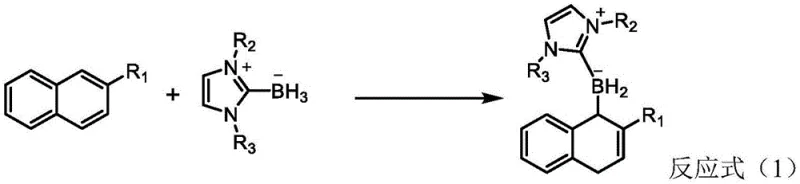
This novel route eliminates the need for dangerous strong acids or bases, relying instead on mild inorganic bases like cesium carbonate and a thiol additive, ethyl thioglycolate, to mediate the radical process. The operational simplicity is a major advantage; the reaction can be performed in standard solvents like acetonitrile under blue LED irradiation. For supply chain managers, this translates to a process that is easier to scale using flow chemistry reactors equipped with LED arrays, offering a clear path toward continuous manufacturing and improved throughput without the safety risks associated with high-pressure or high-temperature batch reactors.
Mechanistic Insights into Visible-Light Photocatalytic Hydrogen Atom Transfer
To fully appreciate the technical superiority of this method, one must understand the underlying mechanistic cycle involving the iridium photocatalyst and the N-heterocyclic carbene (NHC) borane species. Upon irradiation with blue light in the 420nm to 480nm range, the ground state iridium(III) complex is excited to a long-lived triplet state. This excited species acts as a potent oxidant, capable of accepting an electron from the NHC-borane adduct. This single-electron transfer (SET) event generates an NHC-borane radical cation, which subsequently undergoes deprotonation to form a nucleophilic boryl radical. This radical species is the key active intermediate that attacks the electron-deficient naphthalene ring system.
The subsequent steps involve a radical addition to the aromatic system followed by a hydrogen atom transfer from the thiol co-catalyst, ethyl thioglycolate, to restore aromaticity in the non-reduced ring and finalize the 1,4-dihydro structure. The role of the thiol is critical; it serves as a hydrogen donor to quench the carbon-centered radical intermediate, regenerating the thiyl radical which then closes the catalytic cycle. This intricate balance of redox potentials ensures that side reactions, such as over-reduction or polymerization, are minimized. The presence of cesium carbonate is also mechanistically vital, as it facilitates the deprotonation step of the borane radical cation, thereby accelerating the formation of the active boryl radical species and improving the overall turnover number of the photocatalyst.
From an impurity control perspective, this mechanism offers distinct advantages over ionic pathways. Because the reaction proceeds via discrete radical intermediates rather than highly reactive carbocations or carbanions, the formation of rearrangement byproducts is significantly suppressed. The mild conditions prevent the degradation of sensitive functional groups such as esters and amides, which might otherwise hydrolyze under the harsh acidic or basic conditions required by older methods. This results in a cleaner crude reaction profile, simplifying downstream purification and increasing the isolated yield of the target high-purity pharmaceutical intermediate.
How to Synthesize 1,4-Dihydronaphthalene Derivatives Efficiently
Implementing this photocatalytic protocol requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the precise weighing of the iridium photocatalyst at a loading of approximately 1 mol% relative to the substrate. The reaction mixture, comprising the 2-naphthalene derivative, the NHC-borane, the base, and the thiol additive in acetonitrile, must be rigorously degassed to exclude oxygen, which can quench the excited state of the photocatalyst and inhibit radical propagation. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining consistency.
- Combine the 2-naphthalene derivative, N-heterocyclic carbene borane, iridium photocatalyst, and cesium carbonate in a Schlenk bottle under inert atmosphere.
- Add ethyl thioglycolate and acetonitrile solvent, then degas the solution thoroughly to remove oxygen.
- Irradiate the mixture with blue LEDs (420-480nm) at 40°C for 24 hours, followed by silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers compelling strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive and regulated heavy metals like osmium and platinum with a catalytic amount of an organic-soluble iridium complex and commodity chemicals like ethyl thioglycolate, the direct material costs are significantly reduced. Furthermore, the elimination of stoichiometric metal reagents removes the need for costly metal scavenging steps during purification, which traditionally adds time and expense to the manufacturing process.
- Cost Reduction in Manufacturing: The shift from stoichiometric heavy metal reagents to a catalytic photocatalytic system fundamentally alters the cost structure of producing 1,4-dihydronaphthalene intermediates. Since the iridium catalyst is used in minute quantities (around 1 mol%) and the other reagents are inexpensive bulk chemicals, the variable cost per kilogram of product is substantially lowered. Additionally, the mild reaction temperature eliminates the need for energy-intensive heating or cooling systems, leading to reduced utility costs. The simplified workup procedure, which avoids complex metal removal protocols, further decreases labor and solvent consumption, resulting in a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: Relying on exotic or tightly controlled reagents creates vulnerability in the supply chain, whereas the reagents for this process are widely available from multiple global suppliers. Ethyl thioglycolate, cesium carbonate, and acetonitrile are commodity chemicals with stable pricing and abundant supply, mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output even when sourcing from different vendors. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The use of visible light LEDs as the energy source aligns perfectly with modern green chemistry principles and sustainability goals. Unlike thermal processes that generate significant carbon footprints through energy consumption, LED irradiation is highly energy-efficient and generates minimal heat, simplifying thermal management during scale-up. The absence of toxic heavy metal waste streams simplifies environmental compliance and waste disposal, reducing the regulatory burden on the manufacturing site. This eco-friendly profile not only lowers operational costs related to waste treatment but also enhances the corporate sustainability image, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the safety advantages of this photocatalytic method compared to traditional osmium routes?
A: Unlike traditional methods requiring toxic osmium reagents or harsh oxidative conditions, this protocol operates at mild temperatures (25-45°C) using visible light, significantly reducing operational hazards and waste disposal costs.
Q: Can this synthesis method accommodate diverse functional groups on the naphthalene ring?
A: Yes, the method demonstrates broad substrate tolerance, successfully converting 2-naphthalene derivatives bearing esters, amides, cyano, and ketone groups into the corresponding 1,4-dihydronaphthalene products with good yields.
Q: Is the iridium photocatalyst recoverable or required in stoichiometric amounts?
A: The process utilizes the iridium complex in catalytic amounts (approximately 1 mol%), which is highly efficient compared to stoichiometric metal reagents used in older methodologies, thereby lowering raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dihydronaphthalene Supplier
The technological advancements detailed in CN116396315A underscore the potential for producing high-value 1,4-dihydronaphthalene intermediates with unprecedented efficiency and safety. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global supply chains. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of our pharmaceutical intermediates meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this photocatalytic method can optimize your budget. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects, ensuring a seamless transition from development to commercial supply.
