Efficient One-Step Copper-Catalyzed Synthesis of Aryl-Substituted Homoallyl Alcohols for Commercial Scale-Up
Introduction to Advanced Intermediate Synthesis
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, cost-effective, and scalable methodologies. A significant breakthrough in this domain is detailed in patent CN109678673B, which discloses a novel synthetic method for aryl-substituted homoallyl alcohols. These compounds serve as critical building blocks in the construction of complex organic architectures, including tetrahydrofurans and various halogenated derivatives essential for drug discovery. The patented approach utilizes a copper-catalyzed coupling between aryl boron compounds and alkenyl epoxides, offering a distinct advantage over legacy methods by operating under mild conditions with exceptional regio- and stereoselectivity. This technological leap addresses long-standing challenges in the industry, such as the reliance on harsh reagents and multi-step sequences, positioning it as a vital tool for modern pharmaceutical intermediate manufacturing.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl-substituted homoallyl alcohols has been plagued by inefficiencies that hinder large-scale production. Traditional routes, such as the Wittig reaction described in literature by Sigman and Suero, involve a cumbersome multi-step sequence. This process necessitates the protection of hydroxyl groups using TBSCl, followed by the formation of phosphonium salts and subsequent reaction at cryogenic temperatures ranging from -20°C to -78°C using strong bases like n-BuLi. Such conditions are not only energy-intensive but also pose significant safety hazards and operational complexities in a plant setting. Furthermore, alternative pathways like olefin metathesis using Grubbs' catalyst suffer from the high cost of Ruthenium-based catalysts and the limited commercial availability of diverse substituted styrenes, restricting the structural diversity accessible to chemists.
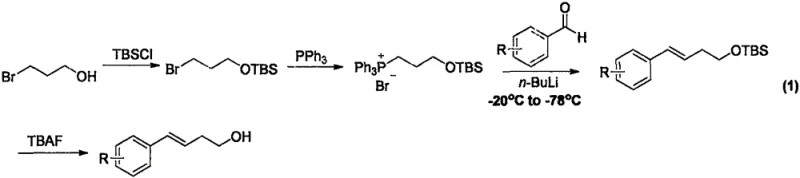
The Novel Approach
In stark contrast, the methodology outlined in CN109678673B streamlines the entire process into a single, robust step. By leveraging a copper catalytic system comprising cuprous chloride (CuCl) and a ligand such as TMEDA, the reaction proceeds efficiently at a moderate temperature of 60°C. This approach completely bypasses the need for protecting groups, cryogenic cooling, or expensive noble metal catalysts. The use of readily available aryl boronates and vinyl epoxides as starting materials ensures a broad substrate scope and simplifies the supply chain logistics. This shift from a three-step, hazard-prone protocol to a direct coupling reaction represents a paradigm shift in how these valuable intermediates are produced, offering substantial improvements in both economic viability and operational safety for fine chemical manufacturers.
Mechanistic Insights into Copper-Catalyzed Epoxide Ring Opening
The core of this innovation lies in the copper-catalyzed cross-coupling mechanism, which facilitates the nucleophilic attack of the aryl group onto the vinyl epoxide. The catalytic cycle likely involves the transmetallation of the aryl boron species to the copper center, followed by the regioselective ring-opening of the epoxide. This specific pathway is engineered to favor the formation of the thermodynamically stable E-alkene geometry, ensuring high stereochemical purity without the need for extensive chromatographic separation of isomers. The choice of ligands, such as 1,3-diketones or dinitrogen ligands like TMEDA, plays a crucial role in stabilizing the active copper species and enhancing the reaction rate. This mechanistic precision allows for the tolerance of a wide array of functional groups that would typically be incompatible with stronger nucleophiles or reducing agents used in older methods.
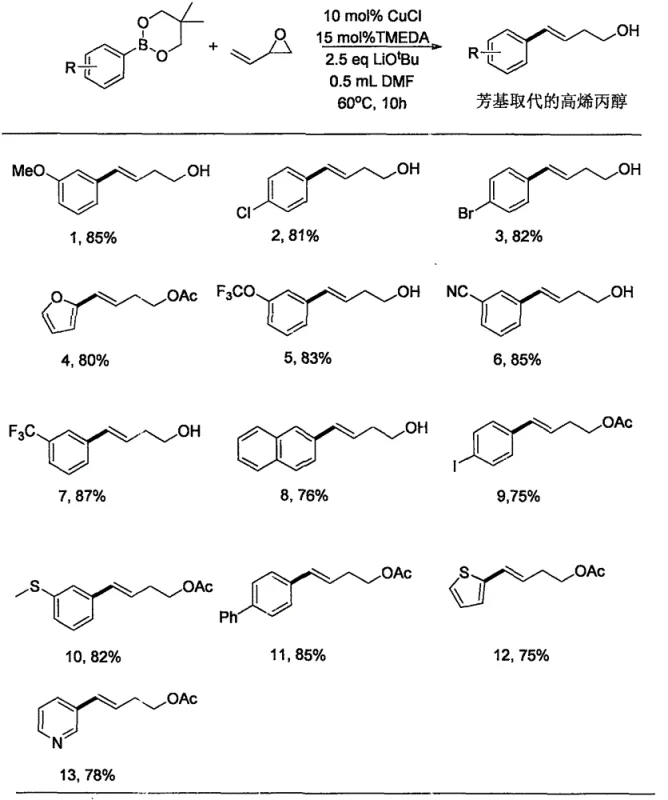
Furthermore, the reaction exhibits remarkable chemoselectivity, leaving sensitive moieties such as esters, nitriles, and halides intact. For instance, substrates containing bromo or chloro substituents on the aromatic ring undergo coupling smoothly without competing side reactions like dehalogenation. This level of control is paramount for R&D directors aiming to synthesize complex libraries of analogs for structure-activity relationship (SAR) studies. The ability to introduce diverse aryl groups while maintaining the integrity of the homoallylic alcohol backbone underscores the versatility of this catalytic system. It effectively solves the problem of low total yields associated with sequential synthetic routes, providing a direct line to high-purity products suitable for immediate downstream processing in API manufacturing.
How to Synthesize Aryl-Substituted Homoallyl Alcohol Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring standard laboratory equipment and avoiding specialized cryogenic setups. The protocol involves mixing the copper catalyst, ligand, and base in a solvent like DMF under an inert atmosphere, followed by the addition of the boronate and epoxide substrates. Heating the mixture to 60°C for approximately 10 hours drives the reaction to completion with high conversion rates. Workup procedures are straightforward, involving simple aqueous quenching and extraction, which facilitates rapid isolation of the crude product. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized guide below.
- Prepare the reaction mixture by combining cuprous chloride (10 mol%), TMEDA ligand (15 mol%), lithium tert-butoxide base (2.5 eq), and arylneopentyl glycol boron ester (2 eq) in a sealed reactor under inert atmosphere.
- Add DMF solvent and vinyl epoxy substrate to the mixture, then heat the reaction to 60°C and stir for 10 hours to ensure complete conversion.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, filter through silica, and purify the crude product via column chromatography to obtain the pure alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed technology offers transformative benefits for procurement and supply chain management. The elimination of expensive catalysts like Grubbs' reagent and the avoidance of hazardous reagents like n-BuLi directly translate to a leaner cost structure. By utilizing commodity chemicals such as cuprous chloride and common boronates, manufacturers can significantly reduce raw material expenditures. Moreover, the simplified one-step nature of the reaction reduces labor costs and minimizes the time required for batch processing, thereby enhancing overall throughput. This efficiency is critical for maintaining competitive pricing in the global market for pharmaceutical intermediates, allowing companies to offer more attractive quotes to their clients without compromising on quality.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with abundant copper salts drastically lowers the catalyst cost per kilogram of product. Additionally, the removal of protection and deprotection steps reduces the consumption of auxiliary reagents and solvents, leading to substantial savings in waste disposal and material handling. The mild reaction conditions also lower energy consumption compared to processes requiring deep freezing or high-pressure hydrogenation, contributing to a more sustainable and cost-effective production model.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as aryl boronic esters and vinyl epoxides mitigates the risk of supply disruptions often associated with specialized or custom-synthesized reagents. Since the reaction does not require air- or moisture-sensitive reagents that demand stringent storage conditions, logistics are simplified, and inventory management becomes more flexible. This robustness ensures a consistent supply of high-quality intermediates, which is essential for meeting the rigorous delivery schedules of multinational pharmaceutical clients.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and workup makes this process highly amenable to scale-up from gram to ton quantities. The absence of heavy metal contaminants like Ruthenium simplifies the purification process and ensures that the final product meets stringent regulatory limits for residual metals in drug substances. Furthermore, the reduced generation of chemical waste due to fewer synthetic steps aligns with green chemistry principles, helping manufacturers comply with increasingly strict environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is key for stakeholders evaluating its potential for their specific applications. The following questions address common inquiries regarding the reaction's scope, selectivity, and operational requirements. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of this copper-catalyzed method over traditional Wittig or Grubbs routes?
A: This method eliminates the need for cryogenic temperatures (-78°C) and strong bases like n-BuLi required in Wittig reactions, and avoids the expensive Ruthenium catalysts used in olefin metathesis. It operates at a mild 60°C with cheap copper salts, significantly reducing operational costs and safety risks.
Q: Does this synthesis method offer good functional group tolerance for complex drug molecules?
A: Yes, the protocol demonstrates excellent compatibility with sensitive functional groups including halides (Cl, Br, I), ethers, nitriles, and heterocycles like furan and pyridine, making it highly suitable for late-stage functionalization in pharmaceutical synthesis.
Q: Is the stereochemistry of the resulting alkene controlled in this reaction?
A: The reaction is highly stereoselective, exclusively producing the E-configured aryl-substituted homoallyl alcohol isomers without detectable Z-isomer byproducts, which simplifies downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl-Substituted Homoallyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the technology described in CN109678673B and is fully equipped to implement this copper-catalyzed methodology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are designed to handle sensitive organometallic reactions with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality standards for every batch produced.
We invite you to leverage our technical expertise to optimize your supply chain for aryl-substituted homoallyl alcohols and related intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and efficiency for your organization.
