Scalable Synthesis of Tetramethyl Spiroindane Bisoxazoline Ligands for Industrial Asymmetric Catalysis
Scalable Synthesis of Tetramethyl Spiroindane Bisoxazoline Ligands for Industrial Asymmetric Catalysis
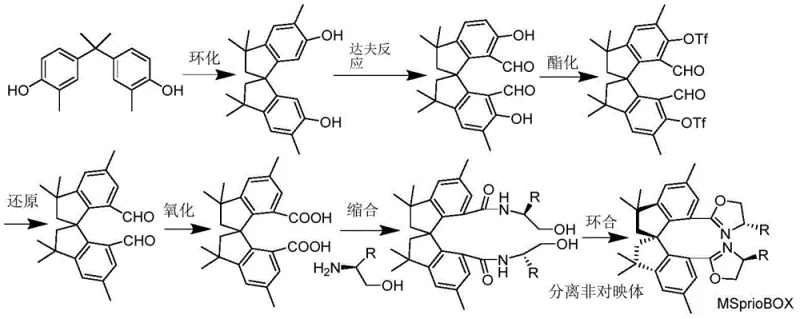
The landscape of asymmetric catalysis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal advancement in this field is detailed in patent CN108794420B, which introduces a novel class of bisoxazoline ligand compounds based on a tetramethyl spiroindane skeleton. Unlike traditional ligands that rely on complex and expensive starting materials, this innovation leverages the economic potential of industrial bisphenol derivatives. The patent outlines a robust synthetic pathway that transforms cheap, commercially abundant tetramethyl spiroindane diphenol into high-performance chiral ligands, known as MSpiroBOX. This technological breakthrough addresses critical pain points in catalyst design, specifically focusing on structural rigidity, stability, and cost-efficiency, making it an attractive option for reliable chiral ligand suppliers aiming to serve the global fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiro-based bisoxazoline ligands, such as the well-known SpiroBOX, has been hindered by significant economic and logistical barriers. The conventional route typically begins with m-methoxybenzaldehyde, a raw material that is not only more expensive but also requires a lengthy synthetic sequence to construct the spiro backbone. Literature indicates that obtaining the corresponding SpiroBOX from m-methoxybenzaldehyde necessitates at least 11 synthetic steps, coupled with a dedicated chiral resolution step. This extended reaction sequence inherently accumulates impurities, lowers overall yield, and drastically increases the cost of goods sold (COGS). Furthermore, the presence of active arylmethylene groups in some traditional spiro frameworks can lead to stability issues under harsh reaction conditions, limiting their utility in diverse catalytic applications. For procurement managers, these factors translate into supply chain volatility and inflated pricing for the final chiral catalysts.
The Novel Approach
The methodology disclosed in CN108794420B represents a paradigm shift by utilizing tetramethyl spiroindane diphenol (MSPINOL) as the foundational building block. This starting material can be produced directly and in high yield from industrial bisphenol series products via simple acid catalysis, a process that is already established on a multi-ton scale globally. By adopting this feedstock, the total number of synthetic steps to reach the final bisoxazoline ligand is reduced to approximately seven, with most post-reaction treatments being straightforward and scalable. The tetramethyl spiroindane framework offers enhanced rigidity and stability due to the absence of reactive arylmethylene moieties, ensuring consistent performance in metal-catalyzed asymmetric reactions. This streamlined approach not only simplifies the manufacturing process but also opens the door for cost reduction in asymmetric catalysis manufacturing, allowing producers to offer high-purity ligands at a fraction of the traditional cost.
Mechanistic Insights into the Synthetic Pathway
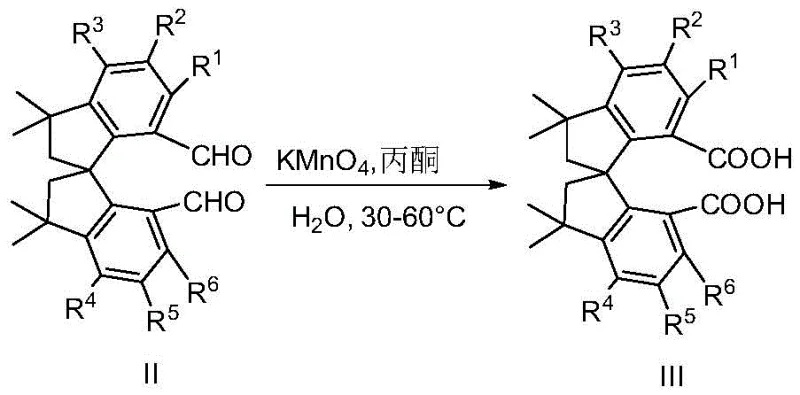
The core of this innovation lies in the efficient functionalization of the spiroindane core. The synthesis begins with a Duff reaction, where tetramethyl spiroindane diphenol is treated with hexamethylenetetramine (HMTA) in trifluoroacetic acid (TFA) to introduce formyl groups at the ortho positions relative to the hydroxyl groups. This electrophilic aromatic substitution is highly regioselective, ensuring the correct positioning for subsequent ring closure. Following formylation, the intermediate dialdehyde undergoes oxidation, typically using potassium permanganate in an acetone-water system, to yield the corresponding dicarboxylic acid. This oxidation step is critical as it activates the molecule for amide bond formation. The resulting diacid is then converted into an acyl chloride or directly condensed with chiral aminoethanol derivatives. The final cyclization to form the oxazoline rings is achieved using methanesulfonyl chloride (MsCl) in the presence of a base like triethylamine and a catalyst such as DMAP. This intramolecular dehydration creates the rigid bisoxazoline structure essential for inducing chirality in catalytic cycles.
Impurity control is meticulously managed throughout this sequence. The use of robust reagents like HMTA and KMnO4 minimizes the formation of side products common in more delicate organometallic couplings. Additionally, the crystallization properties of the intermediates, such as the dicarboxylic acid, allow for effective purification via recrystallization or simple filtration, ensuring that the final ligand meets stringent purity specifications required for pharmaceutical applications. The ability to separate diastereomers at the final stage further enhances the optical purity of the product, a key metric for R&D directors evaluating ligand efficacy.
How to Synthesize Tetramethyl Spiroindane Bisoxazoline Efficiently
The synthesis of these advanced ligands is designed for operational simplicity and high throughput. The process initiates with the formylation of the spiro-diphenol backbone, followed by oxidation to the diacid, and concludes with a two-step condensation-cyclization sequence using chiral amino alcohols. This route avoids the need for cryogenic conditions or exotic catalysts in the early stages, making it highly accessible for standard chemical manufacturing facilities. The detailed standardized synthetic steps, including specific reagent ratios, temperature controls, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up.
- Start with tetramethyl spiroindane diphenol and perform a Duff reaction using HMTA and TFA to introduce formyl groups.
- Oxidize the resulting dialdehyde intermediate using potassium permanganate to generate the corresponding dicarboxylic acid.
- Condense the diacid with aminoethanol derivatives followed by cyclization using methanesulfonyl chloride to form the final bisoxazoline ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this tetramethyl spiroindane-based ligand technology offers compelling strategic benefits. The primary driver of value is the decoupling of ligand production from scarce or volatile specialty chemical markets. By anchoring the supply chain to commodity bisphenols, manufacturers can insulate themselves from price spikes associated with fine chemical precursors. This shift fundamentally alters the cost structure of chiral ligand production, moving it from a boutique, low-volume operation to a commodity-adjacent model capable of supporting large-scale demand.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like m-methoxybenzaldehyde and the reduction of synthetic steps from eleven to seven result in substantial cost savings. The removal of transition metal catalysts in the backbone construction phase further reduces the burden on downstream purification, eliminating the need for expensive heavy metal scavenging resins. This streamlined process logic translates directly into a lower cost per gram for the final ligand, enabling more competitive pricing for downstream API manufacturers without compromising margin.
- Enhanced Supply Chain Reliability: The reliance on industrial-grade bisphenols, which are produced in millions of tons annually for the polymer industry, ensures a stable and abundant raw material supply. Unlike niche intermediates that may have single-source suppliers, bisphenol derivatives are available from a global network of chemical producers. This diversity in sourcing significantly reduces lead time for high-purity ligand precursors and mitigates the risk of supply disruptions caused by capacity constraints at specialized facilities.
- Scalability and Environmental Compliance: The synthetic route employs standard unit operations such as reflux, filtration, and solvent extraction, which are easily transferable from laboratory to pilot and commercial scales. The avoidance of hazardous reagents in key steps and the generation of manageable waste streams simplify environmental compliance and waste treatment processes. This operational robustness facilitates the commercial scale-up of complex chiral ligands, allowing suppliers to rapidly respond to increasing market demand for asymmetric synthesis solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance and application scope.
Q: What is the primary advantage of the tetramethyl spiroindane skeleton over traditional SPINOL ligands?
A: The tetramethyl spiroindane skeleton lacks active arylmethylene groups, providing superior stability and rigidity. Furthermore, it is derived from inexpensive industrial bisphenols rather than costly m-methoxybenzaldehyde, significantly reducing raw material costs.
Q: Can this ligand synthesis be scaled for commercial production?
A: Yes, the synthesis utilizes robust reactions like the Duff reaction and standard oxidation protocols that are amenable to large-scale manufacturing. The starting materials are available in multi-ton quantities globally.
Q: What types of asymmetric reactions can these ligands catalyze?
A: These ligands are versatile and can complex with various metal salts (Fe, Cu, Rh, etc.) to catalyze asymmetric Friedel-Crafts alkylations, cyclopropanations, and Si-H insertion reactions with high enantioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetramethyl Spiroindane Bisoxazoline Ligand Supplier
The development of tetramethyl spiroindane-based bisoxazoline ligands marks a significant milestone in the accessibility of high-performance chiral catalysts. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chemistry for the global pharmaceutical and fine chemical industries. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of ligand delivered meets the exacting standards required for GMP manufacturing environments.
We invite forward-thinking organizations to collaborate with us to leverage this cost-effective and scalable technology. Whether you require custom synthesis of specific ligand variants or comprehensive process development services, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your project needs.
