Scalable Synthesis of Pyrazole-O-Glycoside Derivatives for Next-Generation Diabetes Therapeutics
The pharmaceutical landscape for type 2 diabetes treatment has been significantly reshaped by the advent of SGLT2 inhibitors, a class of drugs that function by inducing urinary glucose excretion. Central to the manufacturing of these life-saving therapeutics is the efficient synthesis of pyrazole-O-glycoside derivatives, as detailed in patent CN101218244A. This intellectual property outlines a robust methodology for preparing compounds of general formula (I), which serve as critical active pharmaceutical ingredient (API) intermediates. The disclosed process addresses longstanding challenges in carbohydrate chemistry, specifically focusing on achieving high chemical and diastereomeric purity while maintaining low technical costs. For R&D directors and procurement specialists, understanding the nuances of this synthetic route is essential for securing a reliable supply chain for next-generation antidiabetic medications. The technology leverages catalytic hydrogenation and phase transfer catalysis to ensure scalability and reproducibility.
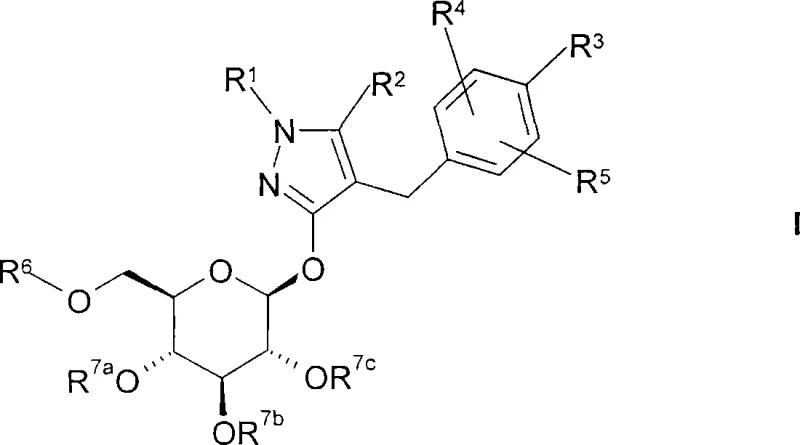
The synthesis of complex glycosides often faces hurdles related to anomeric selectivity and the stability of the aglycone moiety under acidic or basic conditions. Conventional methods frequently rely on harsh Lewis acids or mercury salts for glycosylation, which pose significant environmental and safety liabilities in a commercial setting. Furthermore, traditional routes to the pyrazole aglycone may involve multiple protection-deprotection sequences that erode overall yield and increase waste generation. In contrast, the novel approach described in the patent utilizes a streamlined pathway where the aglycone is constructed via a Mannich-type condensation followed by a selective catalytic hydrogenation. This strategy minimizes the number of isolation steps and avoids the use of toxic heavy metals in the final coupling stages, representing a substantial improvement over legacy methodologies.
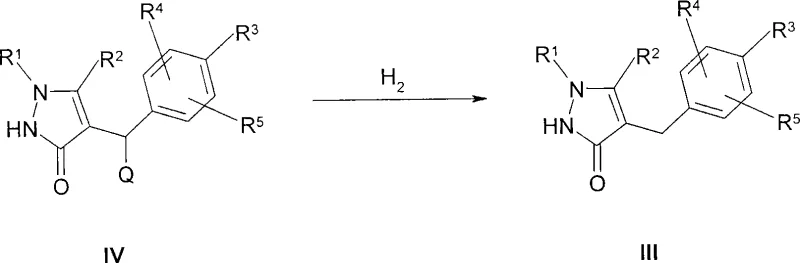
The core of this innovative synthesis lies in the transformation of the benzylic precursor (Formula IV) into the stable aglycone (Formula III) through catalytic hydrogenation. This step is critical as it establishes the carbon framework necessary for subsequent glycosylation without compromising the sensitive pyrazole ring. The reaction is typically conducted in solvents such as methanol, ethanol, or tetrahydrofuran, using transition metal catalysts like palladium on carbon or Raney nickel. The process parameters are tightly controlled, with hydrogen pressures ranging from 1 to 20 bar and temperatures between 20°C to 100°C. In certain embodiments, the presence of an acid additive, such as hydrochloric acid or acetic acid, facilitates the reduction, particularly when the leaving group Q is an alkoxy or amino functionality. This flexibility allows manufacturers to tune the reaction kinetics based on the specific substitution pattern of the aromatic ring.
Following the formation of the aglycone, the glycosylation step employs a phase transfer catalysis (PTC) system to couple the pyrazole with a protected glucose donor (Formula II). This method is superior to traditional Koenigs-Knorr conditions because it operates under milder basic conditions, reducing the risk of epimerization at the anomeric center. The reaction occurs in a biphasic mixture, typically involving an organic solvent like dichloromethane or toluene and an aqueous alkaline phase. Quaternary ammonium salts, such as tetrabutylammonium chloride, act as the phase transfer agent, shuttling the deprotonated pyrazole anion into the organic phase where it reacts with the glucosyl bromide. The pH of the aqueous phase is maintained above 10, often using potassium hydroxide, to ensure complete deprotonation of the aglycone. This precise control over the reaction environment is key to achieving the high beta-selectivity required for biological activity.
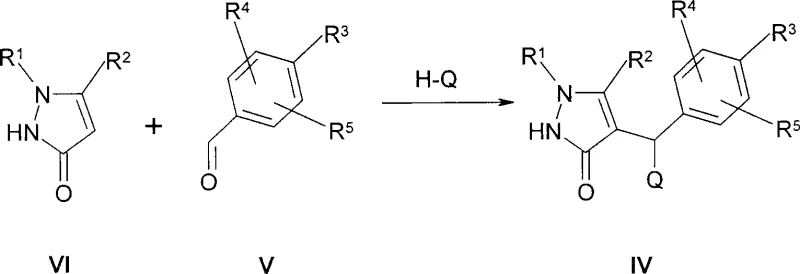
Mechanistically, the efficiency of this process is driven by the interplay between the electronic properties of the pyrazole ring and the steric environment of the glucose donor. The hydrogenation step effectively removes the benzylic heteroatom substituent (Q), which acts as a leaving group, generating a stable methylene linkage. This reduction is highly chemoselective, leaving other functional groups on the aromatic ring, such as fluorine or methoxy substituents, intact. In the glycosylation phase, the nucleophilicity of the pyrazole oxygen is enhanced by the base, allowing it to attack the anomeric carbon of the glucose bromide with inversion of configuration. The use of peracetylated glucose donors ensures that the hydroxyl groups are protected, preventing side reactions. The final deprotection step utilizes alkali metal alcoholates, such as sodium ethoxide, to cleave the ester protecting groups via transesterification, yielding the free hydroxyl groups essential for the drug's mechanism of action.
How to Synthesize Pyrazole-O-Glycoside Intermediates Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized reaction conditions outlined in the patent to ensure consistent quality and yield. The process begins with the preparation of the aglycone, followed by the coupling with the sugar moiety and final deprotection. Each step demands careful monitoring of temperature, pressure, and pH to prevent the formation of impurities that could complicate downstream purification. The following guide summarizes the critical operational parameters derived from the experimental examples, providing a roadmap for technical teams aiming to implement this chemistry. Detailed standardized synthesis steps are provided in the section below.
- Prepare the aglycone intermediate (Formula III) via catalytic hydrogenation of the benzylic precursor (Formula IV) using Pd/C or Raney Nickel under controlled pressure.
- Execute phase transfer glycosylation by reacting the aglycone with protected glucose bromide (Formula II) in a biphasic system using tetrabutylammonium salts.
- Perform final deprotection of the acetyl groups using alkali metal alcoholates in anhydrous alcohol to yield the target pyrazole-O-glycoside.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the methodology described in CN101218244A offers distinct advantages regarding raw material sourcing and process scalability. The starting materials, including substituted benzaldehydes and pyrazolones, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The reliance on catalytic hydrogenation, a well-established unit operation in the fine chemical industry, means that the process can be easily transferred to existing manufacturing facilities without requiring specialized equipment. This compatibility significantly reduces capital expenditure for new production lines. Furthermore, the avoidance of exotic reagents and the use of common solvents like ethanol and dichloromethane simplify waste management and regulatory compliance, leading to substantial cost savings in manufacturing overhead.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and toxic heavy metal promoters often used in traditional glycosylation, such as mercury or silver salts. By replacing these with inexpensive phase transfer catalysts and base metals like nickel or palladium, the direct material costs are significantly lowered. Additionally, the high selectivity of the hydrogenation step reduces the burden on purification processes, minimizing solvent consumption and waste disposal fees. The ability to perform the glycosylation in a biphasic system also simplifies work-up procedures, as the product can be easily extracted into the organic phase, reducing processing time and labor costs associated with isolation.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory approval and market supply. The use of stable intermediates, such as the peracetylated glucose bromide, allows for inventory buffering, decoupling the production of the sugar donor from the final coupling step. This modularity enhances supply chain resilience against disruptions in raw material availability. Moreover, the reaction conditions are forgiving enough to accommodate minor variations in feedstock quality without compromising the final product specification, ensuring a steady flow of intermediates to API manufacturing sites.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in examples ranging from gram to multi-hundred-gram scales with consistent results. The transition from laboratory to commercial scale is facilitated by the use of standard reactors capable of handling pressurized hydrogen and exothermic reactions safely. Environmentally, the process aligns with green chemistry principles by reducing the use of hazardous substances and generating less toxic waste streams. The final deprotection step produces acetate esters as byproducts, which are biodegradable and easier to treat than halogenated waste, supporting corporate sustainability goals and reducing environmental compliance costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of pyrazole-O-glycoside derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the feasibility and advantages of this specific synthetic route for stakeholders evaluating potential manufacturing partnerships. Understanding these details is crucial for making informed decisions about process adoption and supplier selection.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The hydrogenation of Formula IV to Formula III is optimally conducted at temperatures between 20°C to 100°C and hydrogen pressures ranging from 1 to 20 bar, often utilizing palladium or nickel catalysts.
Q: How is high stereochemical purity achieved in the glycosylation step?
A: High diastereomeric purity is maintained through the use of specific phase transfer catalysts like tetrabutylammonium chloride and precise pH control in the aqueous phase during the coupling reaction.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes standard industrial unit operations such as catalytic hydrogenation and liquid-liquid extraction, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole-O-Glycoside Supplier
The technological advancements detailed in CN101218244A represent a significant leap forward in the synthesis of antidiabetic intermediates, offering a pathway to high-purity products with improved economic efficiency. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative patent technologies into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch adheres to the highest quality standards.
We invite you to collaborate with us to leverage this advanced chemistry for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to our deep expertise in catalytic processes and glycosylation chemistry. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can optimize your supply chain for these critical diabetes therapeutics.
