Scalable Copper-Catalyzed Synthesis of Polyfluoroalkyl Indolines and Tetrahydroisoquinolines for Commercial Production
Introduction to Advanced Fluorinated Scaffold Synthesis
The introduction of fluorinated alkyl groups into organic molecules is a critical strategy in modern medicinal chemistry and agrochemical development, as these moieties significantly enhance metabolic stability, lipophilicity, and bioavailability. Patent CN110003081B presents a groundbreaking methodology for the synthesis of polyfluoroalkyl-substituted indolines and tetrahydroisoquinolines, which are valuable structural cores found in numerous biologically active compounds. This technology addresses the longstanding challenge of functionalizing unactivated olefins through a tandem cyclization process that is both operationally simple and economically viable. By shifting away from precious metal catalysis, this invention offers a robust pathway for generating complex fluorinated heterocycles directly from acyclic precursors. For research and development teams seeking reliable pharmaceutical intermediates supplier partnerships, understanding this technological shift is essential for optimizing supply chains and reducing the cost of goods sold for fluorinated drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the construction of 3-polyfluoroalkyl indoline scaffolds predominantly relied on palladium-catalyzed systems, which present significant hurdles for industrial adoption. As illustrated in the prior art reaction scheme, traditional methods typically employ tetrakis(triphenylphosphine)palladium as the catalyst alongside expensive bidentate phosphine ligands such as Xantphos.  Furthermore, these legacy processes often necessitate the use of cesium carbonate as a base, which is not only costly but also highly hygroscopic, complicating storage and handling in large-scale manufacturing environments. The requirement for dry conditions and the sensitivity of the palladium system to impurities can lead to inconsistent batch quality and increased production downtime. Additionally, the use of 1,4-dioxane as a solvent raises environmental and safety concerns due to its toxicity profile, further limiting the sustainability of these conventional synthetic routes in a regulated commercial setting.
Furthermore, these legacy processes often necessitate the use of cesium carbonate as a base, which is not only costly but also highly hygroscopic, complicating storage and handling in large-scale manufacturing environments. The requirement for dry conditions and the sensitivity of the palladium system to impurities can lead to inconsistent batch quality and increased production downtime. Additionally, the use of 1,4-dioxane as a solvent raises environmental and safety concerns due to its toxicity profile, further limiting the sustainability of these conventional synthetic routes in a regulated commercial setting.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes inexpensive copper catalysis to drive the tandem cyclization of N-allylaniline compounds with polyfluoroalkyl halides. This method eliminates the need for costly palladium complexes and specialized phosphine ligands, replacing them with readily available copper powder or copper iodide. The reaction proceeds efficiently in polar aprotic solvents like dimethyl sulfoxide (DMSO) at temperatures ranging from 80°C to 130°C. This transition represents a paradigm shift in cost reduction in fine chemical manufacturing, as it removes the dependency on precious metals that are subject to volatile market pricing. The simplicity of the reaction setup, which does not require rigorous exclusion of moisture for the base component, streamlines the operational workflow and enhances the overall safety profile of the synthesis, making it an ideal candidate for high-volume production of high-purity polyfluoroalkyl indolines.
Mechanistic Insights into Copper-Catalyzed Radical Cyclization
The core of this technological advancement lies in the copper-mediated radical generation and subsequent intramolecular cyclization mechanism. The reaction initiates with the interaction between the copper catalyst and the polyfluoroalkyl halide (Rf-X), facilitating the homolytic cleavage of the carbon-halogen bond to generate a reactive polyfluoroalkyl radical species. This radical then adds regioselectively to the unactivated terminal olefin of the N-allylaniline substrate, forming a new carbon-carbon bond and a transient carbon-centered radical intermediate.  Following this addition, the intermediate undergoes an intramolecular radical cyclization onto the aromatic ring, effectively closing the five-membered indoline or six-membered tetrahydroisoquinoline ring system. The final step involves the restoration of aromaticity and the regeneration of the copper catalyst, completing the catalytic cycle. This mechanistic pathway is particularly advantageous because it tolerates a wide array of functional groups on the aromatic ring, including electron-withdrawing halogens and electron-donating alkyl groups, without interfering with the radical propagation steps. The high selectivity observed minimizes the formation of by-products, thereby simplifying downstream purification and ensuring consistent product quality.
Following this addition, the intermediate undergoes an intramolecular radical cyclization onto the aromatic ring, effectively closing the five-membered indoline or six-membered tetrahydroisoquinoline ring system. The final step involves the restoration of aromaticity and the regeneration of the copper catalyst, completing the catalytic cycle. This mechanistic pathway is particularly advantageous because it tolerates a wide array of functional groups on the aromatic ring, including electron-withdrawing halogens and electron-donating alkyl groups, without interfering with the radical propagation steps. The high selectivity observed minimizes the formation of by-products, thereby simplifying downstream purification and ensuring consistent product quality.
From an impurity control perspective, the radical nature of this transformation ensures that side reactions typical of ionic mechanisms, such as elimination or rearrangement, are largely suppressed. The use of copper as a single-electron transfer agent allows for precise control over the radical flux, preventing oligomerization of the olefin substrate which is a common issue in free-radical polymerizations. Furthermore, the compatibility of the reaction with various protecting groups on the nitrogen atom, such as acetyl, sulfonyl, or carbamate groups, provides chemists with the flexibility to integrate this step at different stages of a multi-step synthesis. This robustness is critical for maintaining high purity specifications in the final active pharmaceutical ingredient, as it reduces the burden on chromatographic separation processes and lowers the risk of genotoxic impurity carryover from heavy metal catalysts.
How to Synthesize Polyfluoroalkyl-Substituted Indolines Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The general procedure involves charging a pressure-resistant reactor with the N-allylaniline substrate, the copper catalyst, and the polyfluoroalkyl halide reagent in a suitable solvent system. The molar ratio of the substrate to the fluorinating agent is typically maintained between 1:1.5 and 1:3.5 to ensure complete conversion while minimizing excess reagent waste. 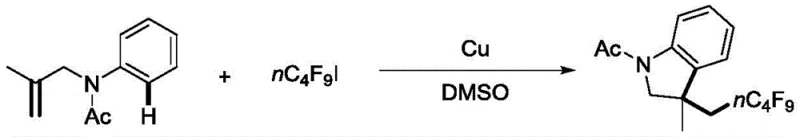 The reaction mixture is then heated to a temperature of approximately 120°C for a duration of 3 to 8 hours, depending on the specific reactivity of the substrates involved. Upon completion, the product is isolated through standard aqueous workup procedures involving extraction with ethyl acetate and drying over anhydrous sodium sulfate, followed by purification via column chromatography. This straightforward protocol underscores the practical utility of the method for generating diverse libraries of fluorinated heterocycles.
The reaction mixture is then heated to a temperature of approximately 120°C for a duration of 3 to 8 hours, depending on the specific reactivity of the substrates involved. Upon completion, the product is isolated through standard aqueous workup procedures involving extraction with ethyl acetate and drying over anhydrous sodium sulfate, followed by purification via column chromatography. This straightforward protocol underscores the practical utility of the method for generating diverse libraries of fluorinated heterocycles.
- Dissolve the N-allylaniline substrate and copper catalyst (Cu or CuI) in dimethyl sulfoxide (DMSO) solvent within a pressure-resistant vessel.
- Add the polyfluoroalkyl halide reagent (such as perfluoroiodobutane) to the reaction mixture maintaining a molar ratio between 1: 1.5 and 1:3.5 relative to the substrate.
- Heat the reaction mixture to a temperature range of 80-130°C, preferably 120°C, for a duration of 3 to 8 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. By replacing palladium and complex ligands with commodity-grade copper, manufacturers can achieve significant cost savings that directly improve the margin profile of the final product. Moreover, the elimination of moisture-sensitive bases like cesium carbonate simplifies inventory management and reduces the risk of batch failures due to reagent degradation, thereby enhancing supply chain reliability. The use of common solvents like DMSO further aligns with green chemistry initiatives and reduces the regulatory burden associated with solvent recovery and disposal.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive copper powder or salts results in a direct decrease in the bill of materials for each production batch. Since the reaction does not require specialized ligands or rigorous anhydrous conditions for the base, the operational overhead related to equipment maintenance and reagent handling is also significantly lowered. This economic efficiency allows for more competitive pricing strategies when sourcing these complex intermediates, making it easier to scale production without proportionally increasing capital expenditure on catalyst recovery systems.
- Enhanced Supply Chain Reliability: Copper and polyfluoroalkyl halides are widely available commodity chemicals with stable supply chains, unlike precious metals which can be subject to geopolitical volatility and supply constraints. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor variations in raw material quality or environmental factors. This reliability ensures consistent delivery timelines for downstream customers, which is critical for maintaining just-in-time manufacturing workflows in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which relies on standard liquid-liquid extraction rather than complex filtration or scavenging steps, facilitates easy scale-up from gram to ton quantities. The absence of toxic heavy metals like palladium in the final product simplifies the regulatory approval process for new drug applications, as residual metal limits are easier to meet. Additionally, the use of less hazardous solvents and the potential for solvent recycling contribute to a reduced environmental footprint, aligning with increasingly stringent global sustainability standards for chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the scope and limitations of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing platforms. We encourage stakeholders to review these points to fully appreciate the versatility and efficiency of the copper-catalyzed approach.
Q: Why is copper catalysis preferred over palladium for this synthesis?
A: Copper catalysis significantly reduces raw material costs compared to expensive palladium catalysts and phosphine ligands like Xantphos. Additionally, copper systems often tolerate a broader range of functional groups and simplify the workup process by avoiding moisture-sensitive bases.
Q: What types of functional groups are tolerated in this reaction?
A: The method demonstrates excellent functional group tolerance, accommodating substituents such as methyl, methoxy, halogens (chlorine, bromine), and phenyl groups on the aromatic ring without compromising the cyclization efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes inexpensive copper powder or salts and common solvents like DMSO. The reaction conditions are robust (80-130°C) and do not require stringent anhydrous conditions for bases, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyfluoroalkyl Indoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis technology for the production of high-value fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for clinical and commercial grade materials. We are committed to leveraging innovative methodologies like the one described in CN110003081B to deliver cost-effective and sustainable solutions for complex chemical challenges.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to obtain specific COA data and route feasibility assessments for new targets, our experts are ready to assist. Partner with us to secure a reliable supply of high-purity polyfluoroalkyl indolines and accelerate your development timelines with confidence.
