Advanced Synthesis of Renieramycin G Intermediates for Commercial Scale Pharmaceutical Production
Advanced Synthesis of Renieramycin G Intermediates for Commercial Scale Pharmaceutical Production
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing complex marine alkaloids with potent anticancer properties. Patent CN103709101A introduces a groundbreaking methodology for the preparation of a novel class of bis-tetrahydroisoquinoline compounds, designated as general formula (I), which serve as critical advanced intermediates. These compounds are pivotal in the efficient asymmetric synthesis of (-)-renieramycin G and its analogues, addressing the historical challenges of low natural abundance and complex total synthesis. By leveraging a highly efficient Pictet-Spengler cyclization reaction, this technology enables the convergence of two distinct fragments derived from affordable L-tyrosine precursors. This innovation represents a significant leap forward for reliable pharmaceutical intermediate suppliers aiming to secure the supply chain for next-generation oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of renieramycin-type alkaloids has been plagued by excessive step counts, reliance on expensive chiral pool materials, and harsh reaction conditions that hinder scalability. As illustrated in previous literature, such as the work by Williams et al. and Liu et al., traditional routes often involve the coupling of two fully functionalized tetra-substituted phenylalanine derivatives followed by difficult ring-closing steps to form the pentacyclic skeleton. 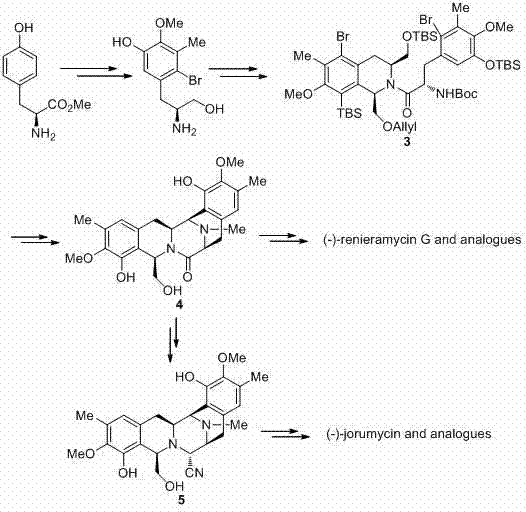 . These legacy methods frequently suffer from unsatisfactory yields in key cyclization steps and require the use of costly reagents or transition metals that complicate downstream purification. Furthermore, the necessity to prepare multiple complex synthetic fragments prior to the final assembly increases the overall lead time and operational expenditure, making cost reduction in pharmaceutical intermediate manufacturing a persistent challenge for procurement teams.
. These legacy methods frequently suffer from unsatisfactory yields in key cyclization steps and require the use of costly reagents or transition metals that complicate downstream purification. Furthermore, the necessity to prepare multiple complex synthetic fragments prior to the final assembly increases the overall lead time and operational expenditure, making cost reduction in pharmaceutical intermediate manufacturing a persistent challenge for procurement teams.
The Novel Approach
In stark contrast, the methodology disclosed in the present patent streamlines the construction of the bis-tetrahydroisoquinoline core through a convergent strategy that drastically simplifies the synthetic landscape. The novel approach utilizes a one-step Pictet-Spengler cyclization between an aldehyde segment containing a tetrahydroisoquinoline unit and a trisubstituted phenylalanine ester segment. This reaction is catalyzed by mild acids such as acetic acid in the presence of water-absorbing agents like 4Å molecular sieves, avoiding the need for harsh Lewis acids or precious metal catalysts. The ability to derive both coupling segments efficiently from cheap L-tyrosine methyl ester ensures a substantial reduction in raw material costs. This flexible and versatile route not only improves the total yield significantly but also offers a modular platform for generating diverse analogues, thereby enhancing the value proposition for research and development departments exploring structure-activity relationships.
Mechanistic Insights into Pictet-Spengler Cyclization
The core of this technological advancement lies in the precise execution of the Pictet-Spengler cyclization, a powerful tool for constructing nitrogen-containing heterocycles. In this specific application, the reaction involves the condensation of the amine functionality of the phenylalanine derivative with the aldehyde group of the tetrahydroisoquinoline fragment to form an iminium ion intermediate. 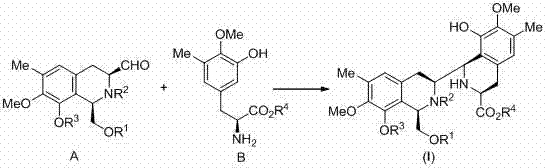 . Under the optimized conditions utilizing a mixed solvent system of dichloromethane and trifluoroethanol, the electron-rich aromatic ring undergoes an intramolecular electrophilic substitution to close the new ring system with high stereocontrol. The use of acetic acid as a catalyst provides sufficient protonation to drive the equilibrium towards the iminium species without promoting decomposition of sensitive functional groups, while the molecular sieves effectively remove water to push the reaction to completion. This mechanistic elegance ensures high purity of the resulting bis-tetrahydroisoquinoline scaffold, minimizing the formation of diastereomeric impurities that are notoriously difficult to separate in later stages.
. Under the optimized conditions utilizing a mixed solvent system of dichloromethane and trifluoroethanol, the electron-rich aromatic ring undergoes an intramolecular electrophilic substitution to close the new ring system with high stereocontrol. The use of acetic acid as a catalyst provides sufficient protonation to drive the equilibrium towards the iminium species without promoting decomposition of sensitive functional groups, while the molecular sieves effectively remove water to push the reaction to completion. This mechanistic elegance ensures high purity of the resulting bis-tetrahydroisoquinoline scaffold, minimizing the formation of diastereomeric impurities that are notoriously difficult to separate in later stages.
Furthermore, the robustness of this cyclization mechanism allows for a wide tolerance of protecting groups, which is essential for the subsequent diversification of the molecule. The patent specifies that protecting groups such as benzyl, tert-butoxycarbonyl (Boc), and allyl can be employed on the nitrogen and oxygen atoms without interfering with the cyclization efficiency. This flexibility is crucial for impurity control, as it allows chemists to tailor the solubility and reactivity of the intermediates to suit specific purification protocols. By maintaining mild reaction temperatures ranging from -60°C to 120°C, the process avoids thermal degradation pathways that often plague the synthesis of complex alkaloids. Consequently, the resulting intermediate exhibits a clean impurity profile, reducing the burden on quality control laboratories and ensuring that the material meets the stringent purity specifications required for clinical trial applications.
How to Synthesize Renieramycin G Efficiently
The translation of this patented chemistry into a practical manufacturing protocol involves a sequence of well-defined transformations that convert the key intermediate into the final bioactive target. Starting from the bis-tetrahydroisoquinoline compound of general formula (I), the synthesis proceeds through a series of functional group manipulations including methylation, deprotection, and oxidation. The detailed standardized synthesis steps outlined below demonstrate the feasibility of scaling this route from gram-scale laboratory experiments to multi-kilogram production batches. Each step has been optimized to maximize yield and minimize waste, reflecting a deep understanding of process chemistry principles. For a comprehensive guide on the specific reagents and conditions required for each transformation, please refer to the structured protocol provided in the subsequent section.
- Preparation of Aldehyde Segment A from L-tyrosine derivatives using Swern oxidation conditions.
- Execution of Pictet-Spengler cyclization between Segment A and Phenylalanine Segment B using acetic acid and molecular sieves.
- Conversion of the resulting intermediate to (-)-renieramycin G through methylation, deprotection, and oxidation steps.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The shift from multi-step, linear syntheses to a convergent approach centered on a high-yielding cyclization step fundamentally alters the cost structure of producing these high-value intermediates. By eliminating the need for expensive transition metal catalysts and reducing the number of isolation and purification steps, the process inherently lowers the consumption of solvents and consumables. This efficiency translates into significant cost savings in pharmaceutical intermediate manufacturing, allowing companies to allocate resources more effectively towards clinical development and market expansion. Moreover, the reliance on commodity chemicals like L-tyrosine as the primary chiral source mitigates the risk of supply disruptions associated with specialized or proprietary starting materials.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available L-tyrosine derivatives as the foundational building blocks, which are far cheaper than the complex fragments required in traditional syntheses. The elimination of costly reagents and the reduction in overall step count mean that the direct material costs are drastically simplified, leading to a more competitive pricing structure for the final active pharmaceutical ingredient. Additionally, the high yields reported in the patent examples, such as the 92% yield in the methylation step and 98% in the deallylation step, ensure that material throughput is maximized, further driving down the cost per kilogram of the produced intermediate.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term drug development projects, and this synthesis route addresses that by utilizing raw materials that are abundant in the global chemical market. Unlike routes dependent on rare natural extracts or custom-synthesized chiral auxiliaries, the L-tyrosine-based strategy ensures a consistent and reliable supply of starting materials regardless of geopolitical or seasonal fluctuations. The robustness of the reaction conditions also means that the manufacturing process is less susceptible to batch-to-batch variability, ensuring that delivery schedules can be met with high confidence. This reliability is essential for maintaining the continuity of clinical trials and preventing costly delays in the drug approval timeline.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the mild reaction conditions and the absence of heavy metal catalysts make this process highly amenable to scale-up in standard chemical reactors. The use of common solvents like dichloromethane and acetonitrile simplifies waste management and solvent recovery operations, aligning with modern green chemistry principles and regulatory requirements. The ability to perform the key cyclization at ambient or moderately elevated temperatures reduces energy consumption compared to cryogenic or high-pressure processes. These factors collectively lower the barrier for commercial scale-up of complex pharmaceutical intermediates, enabling manufacturers to rapidly increase production capacity to meet growing market demand without significant capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The clarity provided here aims to eliminate ambiguity regarding the process capabilities and the quality of the resulting intermediates.
Q: What is the key advantage of this new synthesis route for Renieramycin G?
A: The primary advantage is the significant reduction in synthetic steps and the use of inexpensive L-tyrosine as a starting material, leading to a total yield improvement up to 15.8% compared to conventional methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions ranging from -60°C to 120°C and avoids expensive transition metal catalysts in the key cyclization step, making it highly scalable and cost-effective for industrial manufacturing.
Q: Can this intermediate be used for other alkaloids besides Renieramycin G?
A: Absolutely, the general formula (I) intermediate is versatile and can be chemically modified to synthesize various renieramycin-type alkaloids and analogues, including those with C21 amide or aminonitrile structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Renieramycin G Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in securing the future supply of potent anticancer agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Renieramycin G intermediate delivered meets the highest international standards. We are committed to leveraging our technical expertise to optimize this Pictet-Spengler based route for maximum efficiency and yield, providing you with a secure and high-quality supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your drug development timeline and enhance your competitive edge in the oncology market.
