Revolutionizing Indole Functionalization: A Scalable Visible Light Photocatalytic Strategy for High-Purity Intermediates
Revolutionizing Indole Functionalization: A Scalable Visible Light Photocatalytic Strategy for High-Purity Intermediates
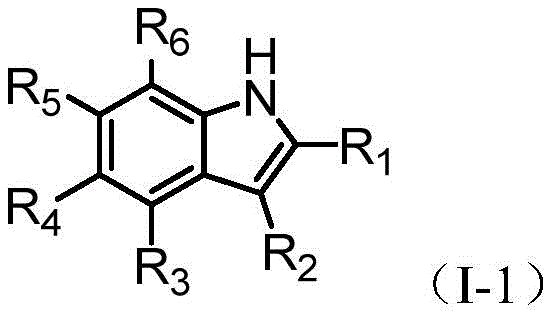
The synthesis of heteroaryl indoles represents a critical frontier in modern medicinal chemistry, serving as a foundational scaffold for numerous bioactive molecules ranging from kinase inhibitors to agrochemical agents. Patent CN113754631B introduces a transformative methodology that addresses the longstanding challenge of site-selective functionalization on the indole ring. Traditionally, achieving precise control over whether a heteroaryl group attaches to the nitrogen (N-1) or the carbon (C-3/C-2) positions has required complex protecting group strategies or expensive transition metal catalysts. This new technology leverages visible light photocatalysis to drive a radical-radical coupling mechanism, enabling direct, efficient, and highly regioselective synthesis under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift away from harsh thermal conditions toward sustainable photochemical processes that maintain high functional group tolerance.
This breakthrough is particularly significant for the development of reliable pharmaceutical intermediate supplier networks, as it simplifies the synthetic route to complex scaffolds. The core innovation lies in the ability to utilize simple indole derivatives and cyano-substituted heteroarenes, such as isoquinolines and quinolines, to generate valuable building blocks without the need for pre-functionalized halides. By operating at ambient temperatures and utilizing visible light, the process minimizes energy consumption and thermal degradation risks, ensuring higher purity profiles for the final active pharmaceutical ingredients (APIs). The versatility of this approach allows for the late-stage modification of bioactive molecules, providing a powerful tool for medicinal chemists to rapidly explore structure-activity relationships (SAR) in drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the arylation of indoles has been dominated by transition metal-catalyzed cross-coupling reactions, typically involving palladium or copper complexes. While effective in many contexts, these traditional methods suffer from inherent limitations that complicate large-scale manufacturing. Firstly, they often require the use of aryl halides or boronic acids, which adds synthetic steps and cost to the precursor preparation. More critically, controlling regioselectivity on the indole nucleus is notoriously difficult; the electronic similarity between the N-1 and C-3 positions often leads to inseparable mixtures of isomers, necessitating costly and yield-reducing purification steps. Furthermore, the reliance on heavy metal catalysts introduces significant regulatory burdens regarding residual metal limits in final drug substances, requiring additional scavenging steps that increase both processing time and waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in CN113754631B utilizes a visible-light-driven radical coupling strategy that bypasses the need for transition metals entirely. By employing organic photocatalysts and specific additives, this novel approach achieves exceptional control over the reaction outcome. As demonstrated in the patent examples, simply changing the additive system allows chemists to switch the selectivity exclusively between the N-1 and C-3 positions. For instance, the use of pyridine directs the coupling to the nitrogen atom, yielding N-heteroaryl indoles, whereas a combination of DABCO and a phosphate additive shifts the reactivity to the C-3 carbon position. This level of tunability is unprecedented in metal-free systems and eliminates the formation of isomeric mixtures.
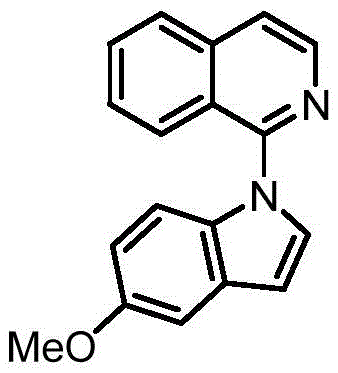
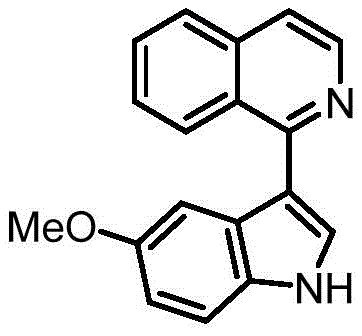
The practical implications of this selectivity control are profound for cost reduction in pharmaceutical intermediate manufacturing. The ability to access either isomer on demand from the same starting materials streamlines inventory management and reduces the need for multiple distinct synthetic routes. Moreover, the reaction proceeds efficiently at room temperature using standard LED light sources, removing the need for specialized high-pressure reactors or cryogenic cooling systems. This simplicity translates directly into lower capital expenditure (CAPEX) and operational expenditure (OPEX) for production facilities, making it an attractive option for both pilot-scale development and commercial tonnage production.
Mechanistic Insights into Visible Light Photocatalytic Radical Coupling
The success of this heteroarylation method relies on a sophisticated interplay between the photocatalyst, the substrate, and the additive. The reaction initiates when the organic photocatalyst, such as 4DPAIPN, absorbs visible light photons to reach an excited state. In this high-energy state, the catalyst facilitates a single-electron transfer (SET) process, generating radical intermediates from the cyano-substituted heteroarene. These radicals are highly reactive species that seek to couple with the electron-rich indole ring. The presence of the cyano group on the heteroarene is crucial, as it stabilizes the radical intermediate and lowers the reduction potential, making the activation energetically feasible under mild visible light irradiation.
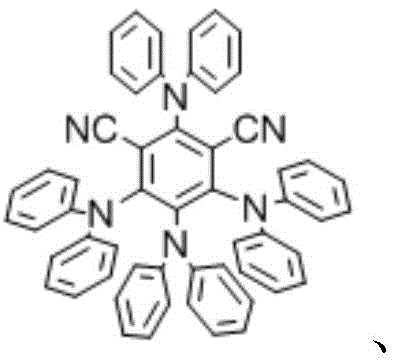
The role of the additive in dictating regioselectivity is a key mechanistic feature that distinguishes this technology. When pyridine is used, it likely acts as a base to deprotonate the indole nitrogen, increasing its nucleophilicity and favoring attack at the N-1 position by the electrophilic radical species. Conversely, when DABCO and phosphates are employed, the mechanism shifts to favor radical addition at the C-3 position, possibly through hydrogen atom transfer (HAT) pathways or by modulating the oxidation potential of the indole. Understanding these subtle mechanistic nuances allows process chemists to fine-tune reaction conditions for specific substrates, ensuring robust impurity control. This deep mechanistic understanding is vital for scaling up the process, as it provides a rational basis for troubleshooting any deviations in selectivity or yield during technology transfer.
How to Synthesize Heteroaryl Indoles Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting is straightforward and requires minimal specialized equipment beyond a visible light source. The general procedure involves dissolving the indole substrate and the cyano-heteroarene in a mixture of organic solvents such as THF and ethyl acetate. The photocatalyst is added in catalytic amounts (typically 0.5-5 mol%), along with the stoichiometric amount of the chosen additive to dictate the desired regioselectivity. After degassing the solution to remove oxygen, the mixture is irradiated with blue LEDs at room temperature. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures for various substrates, are outlined below.
- Prepare Solution A by dissolving the indole compound, cyano-substituted heteroarene, photocatalyst (e.g., 4DPAIPN), and specific additive in an organic solvent mixture.
- Degas the solution with inert gas (Argon or Nitrogen) to remove oxygen which may quench the excited state of the photocatalyst.
- Irradiate the mixture with visible light (LEDs, 380nm-780nm) at room temperature for 2-36 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light photocatalytic technology offers compelling strategic advantages beyond mere technical novelty. The elimination of transition metal catalysts such as palladium or iridium removes a significant cost driver from the bill of materials. These precious metals are subject to volatile market pricing and supply chain constraints; replacing them with abundant organic molecules or inexpensive metal complexes drastically stabilizes raw material costs. Furthermore, the absence of heavy metals simplifies the purification workflow, reducing the consumption of silica gel and scavenging resins, which contributes to substantial cost savings in downstream processing and waste disposal.
- Cost Reduction in Manufacturing: The transition from thermal transition-metal catalysis to ambient photocatalysis fundamentally alters the cost structure of indole functionalization. By removing the requirement for expensive ligands and precious metal salts, the direct material cost is significantly lowered. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, leading to lower utility costs. The simplified workup, devoid of complex metal removal steps, shortens the overall cycle time per batch, thereby increasing facility throughput and asset utilization without requiring new capital investment.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like indoles, isoquinolines, and organic photocatalysts mitigates the risk of supply disruptions often associated with specialized organometallic reagents. The robustness of the reaction conditions—operating effectively at room temperature and atmospheric pressure—means that the process is less sensitive to minor fluctuations in utility supplies or equipment performance. This reliability ensures consistent delivery schedules for high-purity intermediates, allowing downstream API manufacturers to maintain leaner inventory levels and respond more agilely to market demand changes.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles. The use of visible light as the energy source is inherently safer than high-temperature thermal processes, reducing the risk of thermal runaways. The solvent systems employed are standard and easily recyclable. As regulatory pressures regarding heavy metal residues in pharmaceuticals tighten globally, this metal-free approach future-proofs the supply chain against evolving compliance standards, ensuring long-term viability for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic heteroarylation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of the method's capabilities and limitations for potential partners and licensees.
Q: How does this method control regioselectivity between N-1 and C-3 positions?
A: The method achieves switchable regioselectivity through the strategic selection of additives. Using pyridine favors N-1 substitution, while a combination of DABCO and a phosphate additive directs the reaction to the C-3 position.
Q: What are the advantages of using organic photocatalysts over transition metals?
A: Organic photocatalysts like 4DPAIPN eliminate the need for expensive and toxic transition metals (Pd, Ir), significantly reducing raw material costs and simplifying downstream purification by removing heavy metal clearance steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates at room temperature under visible light using commercially available LEDs and common solvents, making it highly scalable and safe for commercial manufacturing without requiring high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heteroaryl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in accelerating drug discovery and development. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic methodologies like CN113754631B can be successfully translated into robust industrial processes. We are committed to delivering high-purity heteroaryl indoles that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify regioselectivity and impurity profiles.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this cutting-edge technology for your pipeline projects. Whether you require custom synthesis of specific isomers or large-scale supply of key intermediates, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis for your specific targets, and let us provide you with specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain.
