Advanced One-Pot Synthesis of Indole Sulfoxides for High-Value Pharmaceutical Intermediates
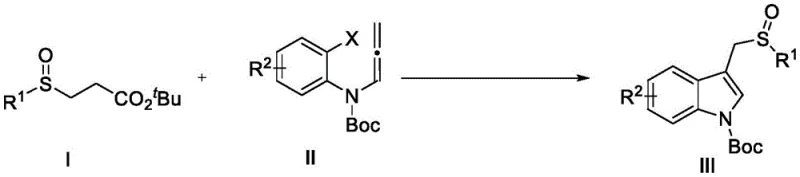
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those containing sulfoxide motifs which are prevalent in blockbuster drugs. Patent CN115181051A introduces a groundbreaking preparation method for sulfoxide compounds containing an indole skeleton, addressing critical bottlenecks in current synthetic methodologies. This innovation leverages a sophisticated palladium-catalyzed system to facilitate a one-pot cascade reaction between beta-sulfinyl tert-butyl esters and N-(o-iodophenyl) allenamides. The significance of this technology cannot be overstated for manufacturers aiming to produce high-purity pharmaceutical intermediates, as it offers a streamlined alternative to classical multi-step syntheses. By enabling the simultaneous construction of carbon-carbon and carbon-sulfur bonds, this process not only enhances atom economy but also drastically reduces the environmental footprint associated with traditional sulfoxide production. For R&D directors and procurement managers alike, understanding the nuances of this patent provides a strategic advantage in sourcing reliable pharmaceutical intermediate suppliers capable of delivering complex molecules with superior efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfoxide compounds has relied heavily on the direct oxidation of thioethers or nucleophilic substitution reactions involving organometallic reagents, both of which present significant challenges for large-scale manufacturing. Direct oxidation strategies often suffer from poor selectivity, leading to over-oxidation to sulfones or incomplete conversion to sulfoxides, thereby complicating downstream purification and reducing overall yield. Furthermore, methods utilizing sulfenic acid anion precursors for arylation, while effective for simple diaryl sulfoxides, frequently struggle when applied to heterocyclic systems like indoles due to compatibility issues with sensitive functional groups. Existing photocatalytic approaches for heterocyclic sulfoxides, although innovative, often require expensive specialized equipment and can result in substantial waste of raw materials like indole, making them economically unviable for cost reduction in pharmaceutical intermediate manufacturing. Additionally, Friedel-Crafts type reactions promoted by trimethylchlorosilane are severely limited in substrate scope, restricting the diversity of molecules that can be accessed and hindering the development of novel drug candidates.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN115181051A utilizes a robust palladium catalytic system to drive a continuous cascade cyclization and substitution reaction. This novel approach allows for the direct coupling of readily available starting materials, specifically beta-sulfinyl tert-butyl esters and N-(o-iodophenyl) allenamides, under remarkably mild conditions. The reaction proceeds efficiently at temperatures ranging from 50-100°C, with optimal results observed at 70°C, eliminating the need for cryogenic conditions or extreme thermal stress that can degrade sensitive intermediates. By employing a specific combination of palladium catalysts, such as tris(dibenzylideneacetone)dipalladium chloroform adduct, and specialized phosphine ligands like tris(2-furyl)phosphine, the process achieves high selectivity and yield without the formation of complex byproduct mixtures. This one-pot strategy not only simplifies the operational procedure but also significantly enhances the scalability of the process, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates where consistency and purity are paramount.
Mechanistic Insights into Palladium-Catalyzed Cascade Cyclization
The core of this technological breakthrough lies in its intricate catalytic cycle, which orchestrates the formation of multiple bonds in a single operational sequence. As illustrated in the mechanistic diagram, the reaction initiates with the oxidative addition of the palladium(0) species to the aryl iodide moiety of the allenamide substrate, generating a key organopalladium intermediate. This is followed by a migratory insertion of the allene functionality into the palladium-carbon bond, forming an allylic palladium complex that sets the stage for ring closure. Crucially, the beta-sulfinyl ester component is activated by the base, typically cesium carbonate, to generate a reactive sulfenic acid anion in situ. This anion then acts as a potent nucleophile, attacking the allylic palladium intermediate to forge the critical carbon-sulfur bond while simultaneously releasing the palladium catalyst back into the cycle via reductive elimination. This elegant mechanism ensures that both the indole skeleton and the sulfoxide functionality are constructed concurrently, minimizing the number of isolation steps and maximizing material throughput.
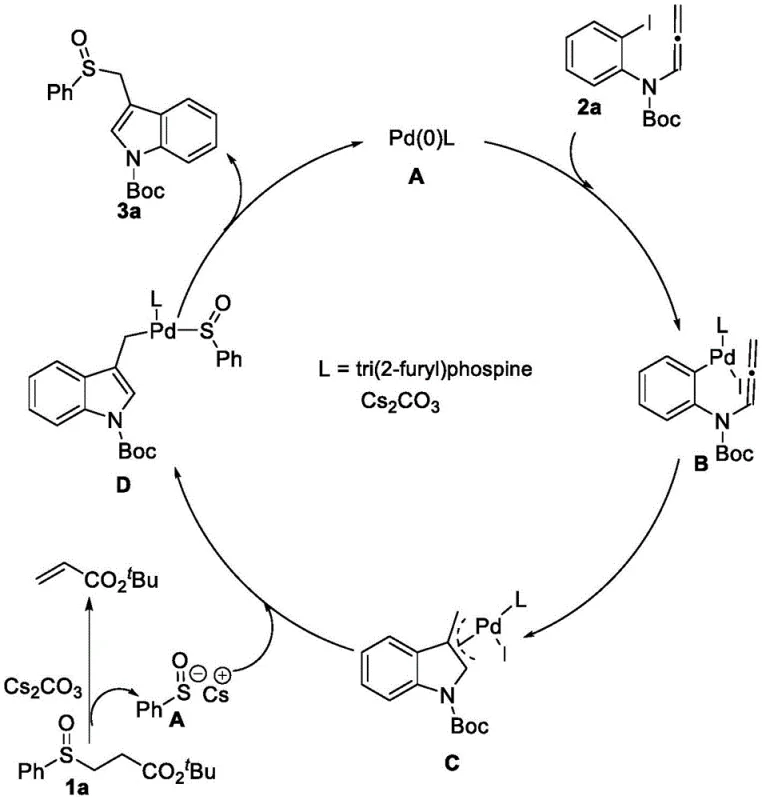
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses where intermediate isolation can introduce contaminants. The continuous nature of the catalytic cycle means that reactive intermediates are consumed as soon as they are formed, reducing the likelihood of side reactions such as polymerization or decomposition. The choice of ligand plays a pivotal role in stabilizing the palladium center and directing the regioselectivity of the insertion step, ensuring that the sulfoxide group is installed at the desired position on the indole ring. Furthermore, the use of mild bases like cesium carbonate helps maintain the integrity of the sulfoxide chirality, preventing racemization which is a common issue in harsher alkaline environments. For quality assurance teams, understanding this mechanism underscores the robustness of the process, as the defined pathway limits the generation of unknown impurities, thereby facilitating easier regulatory approval and stricter adherence to stringent purity specifications required for active pharmaceutical ingredients.
How to Synthesize Indole Sulfoxide Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The patent outlines a standardized protocol where the molar ratios of reactants and catalysts are optimized to ensure complete conversion while minimizing catalyst loading. Typically, the reaction is conducted in a sealed tube under an inert atmosphere to prevent oxidation of the sensitive palladium species and the sulfoxide product. The selection of solvent is also critical, with dry cyclohexane or toluene proving to be the most effective media for solubilizing the reactants and supporting the catalytic cycle. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the palladium catalyst, phosphine ligand, and base in a sealed tube under inert atmosphere.
- Add the beta-sulfinyl tert-butyl ester (Compound I) and N-(o-iodophenyl) allenamide (Compound II) along with the organic solvent.
- Heat the reaction mixture at 50-100°C for 8-50 hours, then purify the resulting indole sulfoxide via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation strategies. The ability to synthesize complex indole sulfoxides in a single pot eliminates the need for multiple reactor vessels and intermediate storage, which directly correlates to reduced capital expenditure and lower working capital requirements. Since the starting materials are commercially available or easily prepared via known literature methods, the supply chain is less vulnerable to bottlenecks associated with exotic reagents. This accessibility ensures a more reliable pharmaceutical intermediate supplier network, allowing manufacturers to secure raw materials with shorter lead times and greater price stability. Moreover, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure processes, contributing to lower utility costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis significantly lowers processing costs by removing the need for intermediate isolation and purification steps, which are often the most expensive parts of a chemical campaign. By avoiding the use of stoichiometric oxidants or harsh reagents that require specialized waste treatment, the process minimizes disposal costs and environmental compliance burdens. The high atom economy of the cascade reaction ensures that a larger proportion of the input mass ends up in the final product, reducing the overall cost of goods sold. Additionally, the potential for catalyst recycling or the use of low-loading palladium systems further drives down the expense associated with precious metal usage, making the process economically viable even for price-sensitive markets.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in substrate structure means that a single production line can be adapted to manufacture a wide library of derivatives without extensive retooling. This flexibility allows suppliers to respond quickly to changing market demands or custom synthesis requests from clients, enhancing the overall agility of the supply chain. The use of stable, shelf-stable reagents reduces the risk of raw material degradation during storage and transport, ensuring consistent quality upon arrival at the manufacturing site. Consequently, partners can rely on a steady flow of high-quality intermediates, reducing the risk of production delays caused by material shortages or quality failures.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the simplicity of the operation, which does not require specialized equipment beyond standard glass-lined or stainless steel reactors. The absence of hazardous byproducts and the use of relatively benign solvents simplify the waste management process, ensuring compliance with strict environmental regulations in major manufacturing hubs. The mild conditions also enhance safety profiles by reducing the risk of thermal runaways or pressure build-ups, creating a safer working environment for plant operators. This combination of scalability and safety makes the technology highly attractive for long-term commercial partnerships focused on sustainable growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the optimal reaction conditions for this indole sulfoxide synthesis?
A: According to the patent data, the optimal conditions involve using cesium carbonate as the base, tris(2-furyl)phosphine as the ligand, and heating at 70°C for 36 hours in dry cyclohexane or toluene.
Q: What is the substrate scope of this palladium-catalyzed method?
A: The method demonstrates a wide substrate expansion range, accommodating various R1 groups including phenyl, benzyl, alkyl chains, and heterocycles like thiophene and pyridine, as well as diverse substituents on the indole ring.
Q: How does this method compare to traditional sulfoxide synthesis?
A: Unlike traditional methods that often require harsh oxidation or multi-step sequences, this approach constructs C-C and C-S bonds simultaneously in a one-pot process under mild conditions, significantly simplifying the operational workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN115181051A for the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of indole sulfoxide meets the exacting standards required for downstream drug synthesis. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing our clients with a secure and dependable source of critical building blocks.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and target specifications. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how this technology can optimize your supply chain and reduce your overall manufacturing costs. Let us be your trusted partner in navigating the complexities of modern pharmaceutical synthesis.
