Revolutionizing Agrochemical Intermediates: Scalable Synthesis of (4-Isopropoxy-2-Methyl) Phenyl Isopropyl Ketone
The global demand for high-performance fungicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN111548257A introduces a groundbreaking preparation method for (4-isopropoxy-2-methyl) phenyl isopropyl ketone, a pivotal precursor for the succinate dehydrogenase inhibitor (SDHI) fungicide Isofetamid. This technical disclosure represents a significant paradigm shift from traditional Friedel-Crafts acylation towards a more efficient cyano-Grignard strategy. By leveraging heterogeneous catalysis and avoiding toxic reagents, this methodology addresses long-standing challenges in purity and environmental safety. For R&D directors and procurement specialists, understanding this transition is crucial for securing a reliable agrochemical intermediate supplier capable of meeting stringent regulatory standards while optimizing production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the hydroxy-ketone precursor involved the Friedel-Crafts acylation of m-cresol with isobutyryl chloride, utilizing aluminum trichloride as a Lewis acid catalyst in carbon disulfide solvent. This legacy approach suffers from severe drawbacks, including poor regioselectivity which necessitates energy-intensive column chromatography for purification. Furthermore, the quenching of aluminum complexes generates massive volumes of corrosive acidic wastewater, creating significant disposal costs and environmental liabilities. The overall yield of this traditional pathway is often restricted to approximately 50%, rendering it economically inefficient for large-scale commercial operations.
The Novel Approach
In stark contrast, the patented methodology employs a three-step sequence starting with the cyanation of m-cresol using thiocyanate salts and a recoverable solid acid catalyst. This is followed by an etherification step to install the isopropoxy group, and finally, a Grignard addition to the nitrile functionality to construct the ketone moiety. This route completely bypasses the need for toxic cyanides like CuCN or KCN and eliminates the generation of acidic waste associated with Lewis acid quenching. The strategic redesign of the synthetic pathway results in a total yield exceeding 79% and simplifies the isolation process to basic filtration and distillation.
Mechanistic Insights into Cyano-Grignard Ketone Synthesis
The core innovation lies in the initial electrophilic aromatic substitution where m-cresol reacts with thiocyanate ions activated by a silica-supported chlorosulfonic acid catalyst. This heterogeneous system ensures high regioselectivity for the para-position relative to the hydroxyl group, minimizing ortho-isomer impurities that are difficult to separate. The use of a solid support allows the catalyst to be physically separated from the reaction mixture via simple filtration, enabling multiple reuse cycles without significant loss of activity. This mechanistic advantage directly translates to reduced raw material consumption and lower catalyst procurement costs over the lifecycle of the manufacturing campaign.
Subsequent transformation involves the nucleophilic attack of the phenoxide anion on haloisopropane, facilitated by phase transfer catalysts such as tetramethylammonium hydroxide. The final key step utilizes isopropyl magnesium halide to attack the nitrile carbon of the intermediate, forming an imine salt which is subsequently hydrolyzed to the desired ketone. This Grignard-based carbonyl formation is inherently cleaner than acylation, as it avoids the formation of stable aluminum-chelated byproducts. The entire sequence is depicted below, highlighting the structural evolution from the starting phenol to the final high-purity ketone.
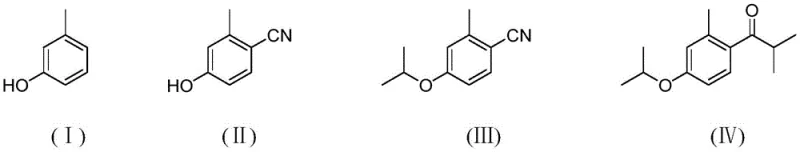
How to Synthesize (4-Isopropoxy-2-Methyl) Phenyl Isopropyl Ketone Efficiently
The operational protocol described in the patent outlines a robust procedure suitable for kilogram-to-tonne scale manufacturing. The process begins with the suspension of the solid acid catalyst in the reaction vessel, followed by the controlled addition of m-cresol and thiocyanate salts at elevated temperatures. After the cyanation is complete, the mixture is filtered to recover the catalyst, and the filtrate is concentrated to isolate the nitrile intermediate. This intermediate is then subjected to alkylation conditions before undergoing the final Grignard reaction under inert atmosphere. Detailed standardized synthesis steps are provided in the guide below.
- React m-cresol with thiocyanate salts using a silica-supported solid acid catalyst to form the nitrile intermediate (Product A).
- Perform nucleophilic substitution on Product A using haloisopropane and a phase transfer catalyst to generate the ether derivative (Product B).
- Treat Product B with isopropyl magnesium halide followed by acidic hydrolysis and vacuum distillation to isolate the final ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits. The elimination of column chromatography and the reduction of hazardous waste streams significantly lower the operational expenditure associated with waste treatment and solvent recovery. Furthermore, the use of readily available commodity chemicals like m-cresol and potassium thiocyanate ensures a stable supply chain不受 geopolitical constraints often associated with specialized acylating agents. The simplified workup procedures also reduce the batch cycle time, enhancing overall plant throughput and responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The implementation of a recyclable heterogeneous catalyst system drastically reduces the recurring cost of catalyst procurement compared to stoichiometric Lewis acids. Additionally, the avoidance of complex purification techniques like column chromatography lowers labor and solvent costs, leading to substantial overall cost savings in agrochemical manufacturing. The higher overall yield means less raw material is required per unit of finished product, further improving the cost basis.
- Enhanced Supply Chain Reliability: By relying on bulk commodity feedstocks rather than specialized acyl chlorides or toxic cyanides, the supply chain becomes more resilient to disruptions. The simplified process flow reduces the number of critical processing steps, minimizing the risk of batch failures and ensuring consistent delivery schedules. This reliability is essential for maintaining the continuity of downstream fungicide production lines.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as filtration, distillation, and liquid-liquid extraction. The significant reduction in acidic wastewater generation aligns with increasingly strict environmental regulations, reducing the risk of regulatory fines and facilitating easier permitting for capacity expansion. This makes the technology highly attractive for long-term commercial scale-up of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route improve environmental compliance compared to traditional methods?
A: The novel process eliminates the use of toxic carbon disulfide solvents and stoichiometric aluminum chloride, significantly reducing acidic wastewater generation and removing the need for complex column chromatography purification.
Q: What is the expected purity and yield for industrial scale production?
A: According to patent data, the optimized route achieves a total yield exceeding 79% based on m-cresol, with final product purity consistently maintained above 99% through simple distillation.
Q: Can the catalyst system be recycled to reduce operational costs?
A: Yes, the heterogeneous solid acid catalyst used in the initial cyanation step can be recovered via filtration and reused after solvent washing, providing substantial long-term cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4-Isopropoxy-2-Methyl) Phenyl Isopropyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating innovative patent technologies into commercially viable manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this cyano-Grignard route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (4-isopropoxy-2-methyl) phenyl isopropyl ketone meets the exacting standards required for modern SDHI fungicide synthesis.
We invite you to collaborate with us to optimize your supply chain for Isofetamid intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
