Advanced Organocatalytic Synthesis of Spiro-Oxindole Butenolides for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds that serve as privileged structures in drug discovery. Patent CN110698490B, titled "Synthetic method of spiro-epoxy indole-cyclopentenobutenoic acid lactone compound," introduces a significant breakthrough in the construction of spiro-oxindole-cyclopentenobutenolide frameworks. These molecular architectures are highly valued for their potent biological activities, including anticancer and antibacterial properties, yet their synthesis has historically been constrained by narrow substrate scope and harsh reaction conditions. This patent discloses a novel organocatalytic approach that utilizes beta-halogenated cycloalkenyl carboxaldehydes and N-substituted isatins to efficiently construct these challenging spiro-centers. By leveraging a thiazole salt-mediated cascade reaction, the technology offers a streamlined pathway to access derivatives that were previously difficult or impossible to synthesize, positioning it as a critical asset for the development of next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
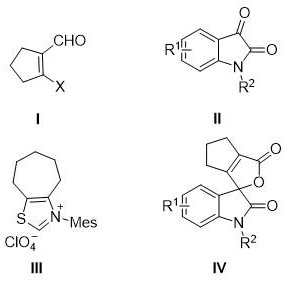
The Limitations of Conventional Methods
Historically, the synthesis of spiro-oxindole butenolides has been plagued by significant structural limitations that hindered the exploration of structure-activity relationships (SAR) in medicinal chemistry programs. Prior literature and existing patents, such as CN103788102A, were largely restricted to the preparation of 3-aryl substituted butenolide derivatives. This restriction meant that chemists could not easily introduce aliphatic or cyclic substituents at the C3 position of the butenolide ring, severely limiting the chemical diversity available for optimization. Furthermore, many traditional methods relied on multi-step sequences involving unstable intermediates or required stoichiometric amounts of expensive reagents, leading to poor atom economy and excessive waste generation. The inability to tolerate diverse functional groups on the isatin nitrogen or the cycloalkenyl ring often resulted in low yields and complicated purification processes, making these routes unsuitable for the commercial scale-up of complex pharmaceutical intermediates required for late-stage drug development.
The Novel Approach
The methodology described in CN110698490B fundamentally overcomes these barriers by enabling the direct assembly of 2,3-ring substituted butenolide derivatives through a convergent annulation strategy. As illustrated in the reaction scheme below, the process couples readily available beta-halogenated cycloalkenyl carboxaldehydes with N-substituted isatins in a single pot. This approach dramatically widens the substrate adaptability range, allowing for the incorporation of various N-substituents such as alkyl, allyl, propargyl, and aryl groups, as well as halogenated variants on the aromatic ring. The use of a thiazole salt catalyst facilitates a mild and efficient cyclization that tolerates a broad spectrum of functional groups without the need for protective group chemistry. This versatility not only accelerates the synthesis of novel analogues for biological screening but also provides a scalable route for the production of high-purity API intermediates, addressing a critical gap in the current supply chain for specialized heterocyclic compounds.
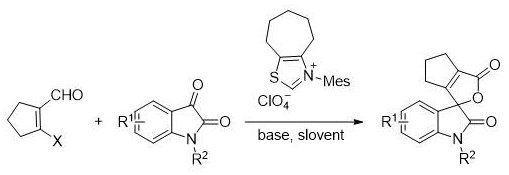
Mechanistic Insights into Thiazole Salt-Catalyzed Annulation
The core of this synthetic innovation lies in the organocatalytic activation of the aldehyde substrate by the thiazole salt, which likely functions as a precursor to an N-heterocyclic carbene (NHC) species upon deprotonation by the base. In this catalytic cycle, the generated carbene nucleophilically attacks the carbonyl carbon of the beta-halogenated cycloalkenyl carboxaldehyde to form a Breslow intermediate or a related enolate equivalent. This activated species then undergoes a conjugate addition or nucleophilic attack on the electrophilic C3 position of the N-substituted isatin. The subsequent intramolecular cyclization and elimination steps lead to the formation of the spiro-oxindole-cyclopentenobutenolide skeleton with high stereocontrol. The presence of the halogen atom on the cycloalkenyl ring plays a crucial role in facilitating the final elimination or substitution step that aromatizes or stabilizes the butenolide ring system. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters, as the interplay between the base strength, solvent polarity, and catalyst loading directly influences the rate of carbene formation and the stability of the key intermediates.
From an impurity control perspective, the mechanistic pathway offers distinct advantages over transition-metal catalyzed alternatives. Since the reaction proceeds via organocatalysis, there is no risk of heavy metal contamination, which is a stringent requirement for pharmaceutical ingredients. The primary byproducts are typically derived from the base or unreacted starting materials, which are easily removed during the workup procedure involving column chromatography. The patent specifies the use of inorganic bases like potassium carbonate or cesium carbonate, which are inexpensive and generate benign salt byproducts. Furthermore, the reaction conditions (50-120°C) are sufficiently mild to prevent the decomposition of sensitive functional groups, thereby minimizing the formation of degradation products or polymerization side-reactions. This clean reaction profile ensures that the final product meets the rigorous purity specifications demanded by regulatory agencies, reducing the burden on downstream purification processes and enhancing the overall efficiency of the manufacturing workflow.
How to Synthesize Spiro-Oxindole-Cyclopentenobutenolide Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly attractive for both laboratory-scale optimization and pilot plant production. The standard protocol involves dissolving the beta-halogenated cycloalkenyl carboxaldehyde and N-substituted isatin in a polar aprotic solvent such as tetrahydrofuran or dioxane under an inert nitrogen atmosphere. The thiazole salt catalyst and the inorganic base are then added to the mixture, which is subsequently heated to reflux temperatures ranging from 50°C to 120°C depending on the specific substrate reactivity. Reaction monitoring indicates that the transformation typically reaches completion within 12 hours, after which the mixture is cooled and concentrated. The crude product is purified via silica gel column chromatography using a petroleum ether and ethyl acetate gradient, yielding the target spiro-compound as a solid with high purity. For detailed standardized operating procedures and specific molar ratios optimized for different substrates, please refer to the technical guide below.
- Dissolve beta-halogenated cycloalkenyl carboxaldehyde and N-substituted isatin in a solvent such as tetrahydrofuran or dioxane under nitrogen protection.
- Add the thiazole salt catalyst and a base like potassium carbonate or cesium carbonate to the reaction mixture.
- Heat the reaction to 50-120°C, then cool, concentrate, and purify the product via column chromatography to obtain the target spiro-compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents compelling economic and logistical benefits that extend beyond mere chemical efficiency. The reliance on commodity chemicals such as isatins, cycloalkenyl aldehydes, and common inorganic bases ensures a stable and cost-effective supply of raw materials, mitigating the risks associated with sourcing exotic or proprietary reagents. The elimination of transition metal catalysts not only reduces the direct material costs but also significantly lowers the expenses related to waste disposal and environmental compliance, as there is no need for specialized heavy metal scavenging resins or complex aqueous workups. This streamlined process translates into substantial cost savings in fine chemical manufacturing, allowing for more competitive pricing of the final intermediates. Additionally, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring consistent batch-to-batch reproducibility which is essential for maintaining long-term supply contracts with pharmaceutical clients.
- Cost Reduction in Manufacturing: The organocatalytic nature of this synthesis eliminates the need for expensive palladium, rhodium, or iridium catalysts that are often required for similar C-C bond-forming reactions. By replacing these precious metals with affordable thiazole salts and inorganic bases, the direct material cost of the reaction is drastically reduced. Furthermore, the simplified post-treatment process, which avoids complex metal removal steps, reduces the consumption of auxiliary materials and shortens the overall production cycle time. This efficiency gain allows manufacturers to allocate resources more effectively, resulting in a lower cost of goods sold (COGS) and improved profit margins for high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The substrates used in this method, specifically N-substituted isatins and beta-halogenated aldehydes, are widely available from multiple global suppliers, reducing the dependency on single-source vendors. This diversification of the supply base enhances resilience against market fluctuations and geopolitical disruptions that can impact the availability of specialized reagents. Moreover, the use of common solvents like THF and dioxane, which are produced at massive scales globally, ensures that solvent shortages are unlikely to interrupt production schedules. This reliability is crucial for meeting the tight delivery timelines expected by multinational pharmaceutical companies, thereby strengthening the partnership between the chemical manufacturer and its downstream clients.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, as they do not require extreme pressures or cryogenic temperatures that pose engineering challenges in large-scale reactors. The mild thermal requirements (50-120°C) can be easily managed with standard heating systems, facilitating a smooth transition from gram-scale laboratory experiments to ton-scale commercial production. From an environmental standpoint, the absence of toxic heavy metals and the use of recyclable solvents align with green chemistry principles, simplifying the permitting process for new manufacturing facilities. This compliance with increasingly stringent environmental regulations future-proofs the production process, ensuring long-term operational continuity without the risk of shutdowns due to regulatory non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of spiro-oxindole-cyclopentenobutenolides based on the data provided in patent CN110698490B. These answers are derived directly from the experimental examples and technical disclosures within the patent document, ensuring accuracy and relevance for stakeholders evaluating this technology for potential licensing or procurement. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing pipelines and for anticipating any specific handling or storage requirements for the intermediates involved.
Q: What are the primary advantages of this synthesis method over conventional aryl-substituted routes?
A: Unlike prior art limited to 3-aryl substituted butenolides, this method enables the synthesis of 2,3-ring substituted derivatives, significantly expanding the chemical space available for drug discovery and allowing for greater structural diversity in pharmaceutical intermediates.
Q: Does this process require expensive transition metal catalysts?
A: No, the process utilizes an organocatalytic system based on thiazole salts and inorganic bases, eliminating the need for costly transition metals and simplifying the removal of heavy metal impurities, which is critical for API manufacturing compliance.
Q: What are the typical reaction conditions and yields reported in the patent?
A: The reaction operates under mild conditions (50-120°C) using common solvents like dioxane. Experimental examples demonstrate robust yields ranging from 61% to 92%, indicating a highly efficient and scalable process suitable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-Oxindole-Cyclopentenobutenolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced heterocyclic intermediates in the development of novel therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN110698490B can be successfully translated into industrial reality. We are committed to delivering high-purity spiro-oxindole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex organocatalytic reactions allows us to offer customized manufacturing solutions that optimize yield and minimize impurities, providing our partners with a competitive edge in the global pharmaceutical market.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cutting-edge synthesis technology for your next drug discovery program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your pipeline with reliable, high-quality chemical building blocks.
