Revolutionizing Fluorinated Intermediate Production via Green Electrochemical Coupling Technology
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to incorporate fluorine-containing functional groups, particularly the trifluoromethylthio (SCF3) moiety, due to its profound impact on lipophilicity and metabolic stability. Patent CN111235594A introduces a groundbreaking electrochemical synthesis method that addresses the longstanding challenges associated with constructing carbon-sulfur bonds in these high-value intermediates. This technology leverages constant current electrolysis to couple aryl thiophenols with sodium trifluoromethanesulfinate, bypassing the need for toxic reagents or precious metal catalysts. By utilizing electricity as a clean reagent, this process aligns perfectly with modern green chemistry principles while delivering high-purity products essential for downstream drug development. The innovation represents a significant leap forward for manufacturers aiming to streamline their supply chains for fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for aryl trifluoromethylthio compounds have historically relied on the nucleophilic substitution of aryl halides with trifluoromethyl mercaptans or similar sulfur sources. These legacy methods suffer from severe drawbacks, including the requirement for elevated temperatures and the use of expensive, often hazardous reagents that complicate waste management. Furthermore, the reactivity of electron-rich aromatic substituents is frequently low in these thermal processes, leading to poor yields and necessitating extensive purification steps that drive up production costs. The reliance on transition metal catalysts in some modern variations introduces another layer of complexity, as residual metal impurities must be rigorously removed to meet stringent regulatory standards for active pharmaceutical ingredients. These cumulative inefficiencies create bottlenecks in both research and commercial manufacturing, limiting the practical utility of existing technologies for large-scale applications.
The Novel Approach
In stark contrast, the electrochemical methodology described in the patent utilizes a direct oxidative coupling strategy that operates under remarkably mild conditions. By employing an undivided cell with inexpensive graphite electrodes, the process facilitates the generation of reactive intermediates at room temperature without external oxidants. This approach not only simplifies the reaction setup but also drastically reduces the environmental footprint by eliminating heavy metal waste streams. The use of sodium trifluoromethanesulfinate as a stable and readily available sulfur source further enhances the safety profile and economic viability of the synthesis. This novel pathway effectively overcomes the electronic limitations of traditional methods, allowing for the efficient functionalization of a wide range of substrates with excellent chemoselectivity.
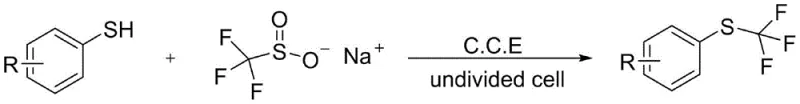
Mechanistic Insights into Electrochemical C-S Bond Formation
The core of this technology lies in the anodic oxidation of the aryl thiophenol substrate, which generates a thiyl radical or cationic species capable of reacting with the trifluoromethanesulfinate anion. Under constant current electrolysis, the graphite anode facilitates the single-electron transfer process that activates the sulfur center, while the cathode balances the charge without interfering with the productive coupling cycle. The presence of a supporting electrolyte, such as tetrabutylammonium tetrafluoroborate, ensures sufficient conductivity in the organic solvent medium, typically acetonitrile, to maintain a steady reaction rate. This electrochemical activation avoids the high-energy barriers associated with thermal homolysis, allowing the reaction to proceed smoothly at 25°C. The mechanism is inherently clean, as the only byproducts are typically hydrogen gas at the cathode and protonated species that are easily managed during workup.
From an impurity control perspective, the absence of transition metals means there is no risk of metal-catalyzed side reactions such as homocoupling of the aryl rings or decomposition of sensitive functional groups. The mild oxidative potential applied is selective for the thiol group, leaving other oxidizable moieties like ethers or esters intact, which is crucial for complex molecule synthesis. Additionally, the use of an undivided cell simplifies the reactor design and minimizes the formation of byproducts that might arise from compartmentalized redox events. This high level of selectivity translates directly into cleaner crude reaction mixtures, reducing the burden on downstream purification processes and improving overall material throughput. The robustness of the graphite electrodes also ensures consistent performance over extended operation times, which is vital for reproducible manufacturing.
How to Synthesize Aryl Trifluoromethylthio Compounds Efficiently
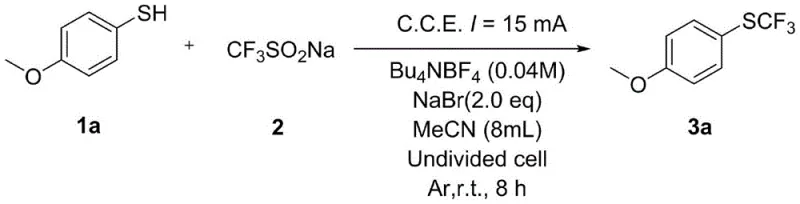
To implement this synthesis effectively, operators must carefully control the electrolysis parameters to ensure optimal conversion and selectivity. The process begins with dissolving the aryl thiophenol and sodium trifluoromethanesulfinate in a suitable solvent system containing the necessary supporting electrolyte. A constant current is then applied across the graphite electrodes, driving the reaction to completion over a period of 8 to 12 hours at ambient temperature. Detailed standardized synthesis steps see the guide below.
- Dissolve aryl thiophenol and sodium trifluoromethanesulfinate in acetonitrile with a supporting electrolyte like tetrabutylammonium tetrafluoroborate.
- Perform constant current electrolysis in an undivided cell using graphite electrodes at room temperature for 8 to 12 hours.
- Quench the reaction with water, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this electrochemical platform offers transformative benefits that extend far beyond simple yield improvements. By shifting away from precious metal catalysis and hazardous oxidants, the process fundamentally alters the cost structure of producing fluorinated intermediates. The elimination of expensive catalysts removes a significant variable cost component, while the simplified workup procedures reduce solvent consumption and labor hours. This leads to substantial cost savings in API manufacturing, making high-value fluorinated building blocks more accessible for early-stage drug development and commercial production alike. The reliance on commodity chemicals like graphite and common salts further insulates the supply chain from volatility in the precious metals market.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts such as palladium or copper eliminates the need for costly scavenging resins and specialized filtration equipment, resulting in significant operational expenditure reductions. Furthermore, the ability to run reactions at room temperature drastically lowers energy consumption compared to thermal processes requiring heating or cooling cycles. The use of inexpensive graphite electrodes instead of platinum or other noble metals ensures that capital expenditure for reactor hardware remains low. These factors combine to create a highly economical process that improves the margin profile for complex fluorinated intermediates without compromising quality.
- Enhanced Supply Chain Reliability: Sourcing stable and safe reagents like sodium trifluoromethanesulfinate is far more reliable than managing volatile thiols or toxic halogenated precursors. The simplified reaction conditions reduce the risk of batch failures due to thermal runaway or catalyst deactivation, ensuring consistent delivery schedules for downstream customers. Additionally, the robustness of the electrochemical setup allows for flexible production scaling, enabling manufacturers to respond quickly to fluctuating demand without requalifying complex catalytic systems. This stability is critical for maintaining continuity in the supply of key pharmaceutical intermediates.
- Scalability and Environmental Compliance: The undivided cell configuration is inherently scalable, allowing for easy transition from laboratory benchtop units to industrial flow reactors or large batch tanks. The green nature of the process, characterized by the absence of heavy metal waste and reduced solvent usage, simplifies regulatory compliance and waste disposal logistics. This environmental advantage aligns with increasingly strict global regulations on chemical manufacturing, future-proofing the production facility against tightening emission standards. The ability to scale efficiently ensures that commercial quantities can be produced with the same high purity and selectivity observed in small-scale trials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this electrochemical synthesis technology. These answers are derived directly from the patent specifications and practical implementation data to provide clarity for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this electrochemical method over traditional nucleophilic substitution?
A: This method eliminates the need for expensive metal catalysts and harsh oxidants, operating under mild room temperature conditions which significantly reduces energy consumption and safety risks associated with high-temperature processes.
Q: Is this process suitable for large-scale manufacturing of fluorinated intermediates?
A: Yes, the use of undivided cells and inexpensive graphite electrodes makes the process highly scalable and cost-effective for commercial production, avoiding the complexities of transition metal removal.
Q: What is the substrate scope for this trifluoromethylthiolation reaction?
A: The method demonstrates broad compatibility with various functional groups including electron-donating methoxy groups, electron-withdrawing esters and ketones, and heterocyclic systems, ensuring versatility for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Trifluoromethylthio Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced fluorination technologies in modern drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this electrochemical coupling can be successfully translated to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aryl trifluoromethylthio compounds meets the exacting standards required by global pharmaceutical clients. Our commitment to quality and technical excellence makes us the ideal partner for sourcing these critical intermediates.
We invite you to contact our technical procurement team to discuss how this green synthesis route can optimize your supply chain. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your project. We are ready to provide specific COA data and route feasibility assessments to support your next development milestone, ensuring a seamless transition from concept to commercial supply.
