Revolutionizing Monosulfonyl Piperazine Production: A Direct One-Step Industrial Strategy
Introduction to Advanced Monosulfonyl Piperazine Synthesis
The pharmaceutical industry constantly seeks more efficient pathways for constructing critical heterocyclic scaffolds, and patent CN102060811A represents a significant breakthrough in the manufacturing of monosulfonyl piperazine derivatives. This intellectual property discloses a novel, direct synthetic methodology that bypasses the cumbersome multi-step protocols traditionally employed in medicinal chemistry laboratories. By utilizing a controlled low-temperature reaction between substituted sulfonic acid chlorides and a stoichiometric excess of piperazine in an organic solvent, this process achieves yields ranging from 66% to 79% with remarkable operational simplicity. The strategic elimination of protection and deprotection sequences not only accelerates the timeline from raw material to final intermediate but also fundamentally alters the economic landscape of producing these vital building blocks for antimalarial, antibacterial, and anticancer agents.
For global procurement teams and R&D directors, the implications of this technology extend far beyond mere academic interest; it offers a tangible pathway to robust supply chain resilience. The ability to synthesize high-purity monosulfonyl piperazine in a single operational step reduces the cumulative exposure to potential impurities that often accumulate during lengthy synthetic sequences. Furthermore, the mild reaction conditions, specifically maintaining temperatures below 5°C, ensure that thermal degradation of sensitive functional groups is minimized, thereby preserving the integrity of complex molecular architectures. This patent provides the foundational chemistry necessary for reliable monosulfonyl piperazine supplier networks to scale production efficiently while adhering to stringent quality standards required by regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of monosubstituted piperazines has been plagued by the necessity of orthogonal protection strategies to prevent disubstitution, a challenge that significantly inflates both cost and complexity. The conventional route, as documented in various literature sources and prior patents, typically involves a tedious three-step sequence that begins with the protection of one nitrogen atom of the piperazine ring. This initial step requires the use of expensive reagents such as di-tert-butyl dicarbonate (Boc2O) to install a tert-butoxycarbonyl group, creating a protected intermediate that is bulky and requires careful handling.
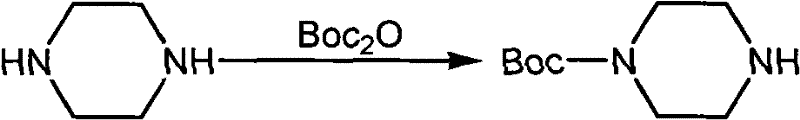
Following the protection step, the mono-protected piperazine must undergo sulfonylation with the desired sulfonyl chloride, a reaction that often requires additional bases and extended reaction times to reach completion. The final and perhaps most problematic stage involves the removal of the protecting group, which typically necessitates the use of strong acids or bases under potentially harsh conditions that can compromise the stability of the newly formed sulfonamide bond or other sensitive moieties within the molecule. This multi-step approach inherently increases the risk of yield loss at every stage, generates substantial chemical waste, and demands rigorous purification between each transformation, making it economically unviable for large-scale industrial applications.
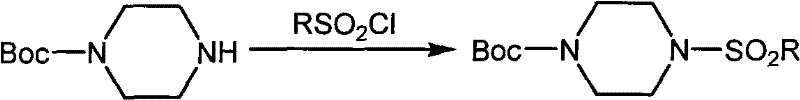

The Novel Approach
In stark contrast to the legacy methods, the innovative process described in CN102060811A achieves the same structural outcome through a direct, one-pot transformation that elegantly sidesteps the need for temporary protecting groups. By leveraging the differential nucleophilicity and the acid-scavenging capability of piperazine itself, this method allows for the selective formation of the monosulfonamide without the risk of significant disubstitution, provided the stoichiometry and temperature are strictly controlled. The reaction is conducted in common organic solvents like dichloromethane at low temperatures, ensuring that the kinetics favor the formation of the desired mono-substituted product while suppressing side reactions.
This streamlined approach drastically simplifies the downstream processing requirements, as the primary byproduct, piperazine hydrochloride, precipitates out of the organic solution as a solid that can be easily removed by simple filtration. This physical separation technique eliminates the need for complex aqueous workups or chromatographic purifications that are often required to remove soluble salts or organic byproducts in traditional methods. Consequently, the novel approach offers a superior profile for cost reduction in pharmaceutical intermediate manufacturing by reducing solvent consumption, minimizing waste disposal costs, and shortening the overall production cycle time from days to hours.
Mechanistic Insights into Direct Low-Temperature Sulfonylation
The success of this direct sulfonylation strategy relies heavily on the precise manipulation of reaction stoichiometry and thermodynamic control to achieve high selectivity. Mechanistically, the process utilizes two equivalents of piperazine for every one equivalent of sulfonyl chloride; the first equivalent acts as the nucleophile, attacking the sulfur center of the sulfonyl chloride to form the sulfonamide bond and release a molecule of hydrogen chloride. The second equivalent of piperazine immediately reacts with this liberated HCl to form piperazine hydrochloride, effectively acting as an intrinsic base that drives the equilibrium forward and prevents the protonation of the remaining free piperazine, which would otherwise deactivate it as a nucleophile.
From an impurity control perspective, maintaining the reaction temperature below 5°C is critical for suppressing the formation of the disulfonylated byproduct, which is the primary competitor in this system. At higher temperatures, the kinetic energy of the molecules increases, potentially overcoming the activation barrier for the second sulfonylation event on the already substituted nitrogen or reacting with the second equivalent of piperazine before it can act as a base. By keeping the system cold, the reaction remains under kinetic control where the formation of the mono-sulfonamide is favored, and the precipitation of the piperazine hydrochloride salt further shifts the equilibrium by removing the acid byproduct from the solution phase, ensuring high purity of the final isolated material.
How to Synthesize Monosulfonyl Piperazine Efficiently
The practical execution of this synthesis requires careful attention to addition rates and thermal management to ensure safety and reproducibility on a commercial scale. The protocol dictates dissolving the piperazine in a suitable solvent like dichloromethane and cooling the mixture significantly before introducing the electrophile, a step that is crucial for managing the exothermic nature of the sulfonylation reaction. Detailed standardized operating procedures regarding the specific dropwise addition rates and stirring efficiencies are essential for maintaining the narrow temperature window required for optimal selectivity.
- Dissolve two equivalents of piperazine in an organic solvent such as dichloromethane and cool the mixture to 0°C using an ice-water bath.
- Slowly add a solution of substituted sulfonyl chloride dropwise while maintaining the reaction temperature below 5°C to control exothermicity.
- Filter off the solid piperazine hydrochloride byproduct, recover the solvent under reduced pressure, and wash the residue with ether to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-step synthesis technology translates directly into enhanced operational efficiency and reduced vulnerability to raw material price fluctuations. By eliminating the need for specialized protecting group reagents like Boc anhydride and the subsequent deprotection agents, manufacturers can significantly simplify their raw material inventory and reduce the number of suppliers required to support production. This consolidation of the supply chain not only lowers administrative overhead but also mitigates the risk of production delays caused by the shortage of any single specialized reagent, thereby enhancing overall supply chain reliability for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic reduction in unit operations, as collapsing a three-step sequence into a single reaction vessel significantly lowers labor, energy, and equipment occupancy costs. Furthermore, the avoidance of expensive protecting group chemistry removes a major cost driver from the bill of materials, while the simplified purification process reduces the volume of solvents and consumables needed for isolation. These factors combine to create a substantially lower cost of goods sold, allowing for more competitive pricing strategies in the global market for fine chemical intermediates without compromising margin.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions, which utilize commodity chemicals like piperazine and common sulfonyl chlorides, ensures that raw material sourcing is robust and less susceptible to geopolitical or logistical disruptions. The ability to produce high-quality intermediates with fewer processing steps means that lead times can be drastically shortened, enabling faster response to customer demand spikes or urgent clinical trial material requirements. This agility is a critical competitive advantage in the fast-paced pharmaceutical sector where time-to-market is often the deciding factor for commercial success.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the reduction in chemical steps inherently reduces the total waste load generated per kilogram of product, aligning with modern green chemistry principles and easing the burden on waste treatment facilities. The process generates a solid salt byproduct that is easily separated and potentially recyclable, minimizing the discharge of hazardous liquid waste streams. This cleaner profile facilitates easier regulatory compliance and permits smoother scale-up from pilot plant to multi-ton commercial production, ensuring long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this direct sulfonylation process for industrial applications. Understanding these nuances is essential for process chemists and engineers looking to adapt this laboratory-scale innovation to large-scale manufacturing environments while maintaining strict quality control standards.
Q: Why is a 2:1 molar ratio of piperazine to sulfonyl chloride required?
A: One equivalent of piperazine acts as the nucleophile to form the sulfonamide bond, while the second equivalent serves as an acid scavenger to neutralize the HCl generated during the reaction, forming a filterable piperazine hydrochloride salt.
Q: What are the advantages of this method over the traditional Boc-protection route?
A: This method eliminates the need for expensive protecting groups like Boc2O and avoids the harsh acidic or basic conditions required for deprotection, resulting in a shorter synthetic route, milder conditions, and easier purification.
Q: Which solvents are suitable for this low-temperature sulfonylation?
A: The patent specifies several compatible organic solvents including acetonitrile, acetone, chloroform, and tetrahydrofuran, with dichloromethane being the preferred choice for optimal solubility and reaction control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monosulfonyl Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN102060811A and have integrated these advanced methodologies into our state-of-the-art production facilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-step process are fully realized in terms of yield, purity, and cost-efficiency. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of monosulfonyl piperazine meets the exacting standards required for API synthesis.
We invite global partners to collaborate with us to leverage this technology for their specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, as well as obtain specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain and accelerate your path to market with our reliable, high-quality pharmaceutical intermediates.
