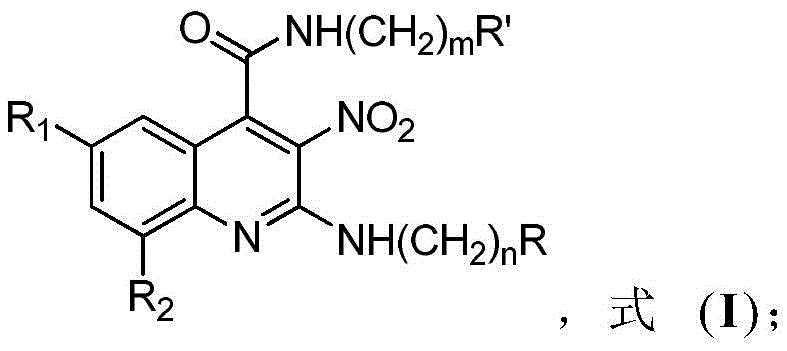
Advanced Metal-Free Synthesis of 3-Nitroquinoline Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking robust and scalable pathways to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN107522659B introduces a groundbreaking methodology for the preparation of 3-nitroquinoline derivatives, a class of nitrogen-containing heterocycles with profound implications in oncology, specifically for treating colorectal cancers which are often resistant to standard chemotherapy. This technology represents a significant leap forward by utilizing simple, commercially available raw materials—isatin compounds and 1,1-enediamine compounds—to construct the quinoline core in a single synthetic operation. The process is characterized by its operational simplicity, avoiding the stringent anhydrous conditions and toxic metal catalysts that typically plague traditional heterocycle synthesis. For R&D teams and procurement specialists alike, this patent offers a compelling route to high-purity intermediates that are critical for developing new anti-tumor agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline derivatives has relied upon classical named reactions such as the Skraup, Doebner-von Miller, or Friedlander syntheses. While these methods are well-documented in academic literature, they suffer from significant drawbacks when applied to modern industrial pharmaceutical manufacturing. These traditional routes often necessitate the use of strong oxidizing agents, high temperatures, and frequently require transition metal catalysts which introduce the risk of heavy metal contamination in the final Active Pharmaceutical Ingredient (API). Furthermore, these multi-step processes generally exhibit longer reaction times and lower overall yields, leading to increased production costs and complex waste streams. The presence of metal residues requires additional, costly purification steps to meet stringent regulatory limits, creating a bottleneck in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
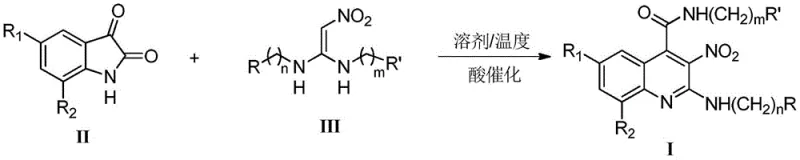
In stark contrast, the methodology disclosed in CN107522659B utilizes a direct addition-cyclization and rearrangement reaction between an isatin derivative and a 1,1-enediamine compound. This innovative strategy operates under mild conditions, typically ranging from 25°C to 120°C, and employs benign organic acids such as acetic acid or p-toluenesulfonic acid as catalysts instead of transition metals. The reaction proceeds efficiently in common aprotic solvents like toluene, xylene, or acetonitrile, achieving completion within 2 to 16 hours. This one-pot transformation not only drastically shortens the synthetic route but also ensures a cleaner reaction profile with yields reported between 75% and 95%. By eliminating the need for metal catalysis, this approach inherently reduces the impurity burden, facilitating easier downstream processing and significantly lowering the cost of goods sold for the final intermediate.
Mechanistic Insights into Organic Acid-Catalyzed Cyclization
The core of this technological advancement lies in the efficient construction of the quinoline ring system through a cascade sequence initiated by organic acid catalysis. The reaction begins with the activation of the carbonyl group on the isatin substrate by the proton donor, enhancing its electrophilicity towards the nucleophilic attack by the 1,1-enediamine. This initial addition is followed by a cyclization event that closes the pyridine ring, subsequently undergoing a rearrangement to establish the aromatic 3-nitroquinoline framework. The use of organic acids provides a controlled acidic environment that promotes this cascade without the aggressive side reactions often associated with mineral acids or Lewis acids. This mechanistic pathway is highly tolerant of various functional groups, allowing for the introduction of diverse substituents at the R1, R2, R, and R' positions, thereby enabling the rapid generation of a focused library of analogs for structure-activity relationship (SAR) studies.
From an impurity control perspective, the absence of transition metals is a decisive advantage for pharmaceutical quality assurance. Metal-catalyzed reactions often leave behind trace amounts of palladium, copper, or iron, which are difficult to remove and can catalyze degradation pathways in the final drug product. By relying solely on organic acid catalysis, the impurity profile of the resulting 3-nitroquinoline derivatives is significantly simplified, primarily consisting of unreacted starting materials or minor regioisomers that are easily separated via standard silica gel chromatography. The purification protocol described involves extraction with ethyl acetate and saturated brine, followed by drying and column chromatography using petroleum ether and ethyl acetate mixtures. This straightforward workup ensures that the final product meets the rigorous purity specifications required for clinical-grade materials, reducing the analytical burden on QC laboratories.
How to Synthesize 3-Nitroquinoline Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for laboratory and pilot-scale production. The process involves charging the isatin and 1,1-enediamine reactants into a reactor with an appropriate aprotic solvent, followed by the addition of the organic acid catalyst. The mixture is then heated to reflux or maintained at a specific temperature until TLC monitoring indicates the complete consumption of the starting material. Detailed standardized synthesis steps see the guide below.
- Combine an isatin compound (Formula II) and a 1,1-enediamine compound (Formula III) in an aprotic solvent such as toluene or acetonitrile.
- Add an organic acid catalyst like acetic acid or trifluoroacetic acid and heat the mixture to a temperature between 25°C and 120°C for 2 to 16 hours.
- Upon completion, extract the reaction mixture with ethyl acetate, dry the organic phase, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The reliance on simple, commodity-grade starting materials such as isatin derivatives and readily accessible enediamines ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or proprietary reagents. The elimination of expensive transition metal catalysts not only reduces the direct material cost but also removes the necessity for specialized scavenging resins or complex filtration units typically required to meet residual metal specifications. This streamlining of the process translates directly into a more favorable cost structure, allowing for competitive pricing in the global market for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this metal-free methodology is substantial. By removing the requirement for precious metal catalysts, manufacturers avoid the high capital expenditure associated with catalyst recovery systems and the ongoing operational costs of purchasing these expensive reagents. Furthermore, the high yields achieved (75-95%) minimize raw material waste, ensuring that a greater proportion of input costs are converted into valuable product. The simplified purification process, which avoids complex metal removal steps, reduces solvent consumption and energy usage during workup, contributing to a leaner and more cost-efficient manufacturing operation that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for API production, and this method strengthens reliability by utilizing robust and widely available chemical building blocks. Isatin and its derivatives are produced on a large industrial scale, ensuring that raw material shortages are unlikely to disrupt production schedules. Additionally, the mild reaction conditions (25°C to 120°C) allow the process to be run in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. This flexibility enables multiple contract manufacturing organizations (CMOs) to potentially produce the intermediate, diversifying the supplier base and reducing dependency on a single source.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden challenges, but this one-pot cyclization is inherently scalable due to its thermal safety profile and lack of hazardous reagents. The use of common organic solvents like toluene and ethyl acetate facilitates easy solvent recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing site. The absence of heavy metals simplifies waste treatment protocols, as the effluent does not require specialized processing to remove toxic metal ions. This environmental compatibility ensures smoother regulatory approvals and long-term sustainability for large-scale commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 3-nitroquinoline derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees.
Q: What are the primary advantages of this synthesis method over classical quinoline production?
A: Unlike classical methods such as Skraup or Doebner-von Miller which often require harsh conditions and metal catalysts, this novel approach utilizes a one-step addition-cyclization rearrangement under mild organic acid catalysis, eliminating the need for expensive transition metals and simplifying purification.
Q: What yields can be expected from this process?
A: The patent data indicates that this method consistently achieves high yields ranging from 75% to 95%, depending on the specific substituents used on the isatin and enediamine starting materials.
Q: What is the biological application potential of these compounds?
A: These 3-nitroquinoline derivatives have demonstrated significant anti-tumor activity, particularly against human colon cancer cell lines such as HCT116 and HT29, showing superior potency compared to standard controls like cisplatin in specific configurations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitroquinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in organic synthesis allows us to optimize this specific acid-catalyzed cyclization route for maximum efficiency and yield, providing our clients with a reliable source of material for their drug development programs.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their pipeline projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and reduce overall development costs.
