Advanced Electrochemical Manufacturing of High-Purity 5-Aminopyrazole-4-Thiocyanate Derivatives
Advanced Electrochemical Manufacturing of High-Purity 5-Aminopyrazole-4-Thiocyanate Derivatives
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, driven by the urgent need to reduce environmental footprints in pharmaceutical and agrochemical manufacturing. A pivotal advancement in this domain is detailed in Chinese Patent CN115074760A, which discloses a novel electrochemical synthesis method for 5-aminopyrazole-4-thiocyanate compounds. This technology represents a significant leap forward in heterocyclic chemistry, specifically addressing the challenges associated with functionalizing the pyrazole core. Pyrazole derivatives are ubiquitous in medicinal chemistry, serving as critical scaffolds for antifungal agents, anti-inflammatory drugs, and kinase inhibitors. The specific subclass of 5-aminopyrazole-4-thiocyanates has garnered attention due to its potent biological activity, particularly against dermatophytes like Trichophyton rubrum. However, traditional synthetic routes have been plagued by the use of harsh oxidants and poor atom economy. The patented electrochemical approach offers a transformative solution, utilizing electricity as a clean reagent to drive the thiocyanation reaction with exceptional efficiency and selectivity.
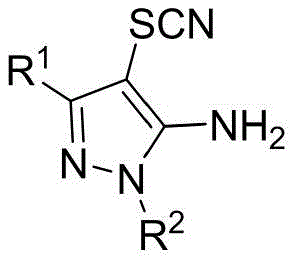
This innovation is not merely an academic curiosity but a robust industrial process capable of delivering high-purity intermediates essential for drug development. By leveraging the principles of organic electrosynthesis, the method circumvents the need for stoichiometric chemical oxidants, thereby reducing the generation of hazardous waste streams. For R&D directors and process chemists, this translates to a cleaner reaction profile and simplified work-up procedures. The ability to install a thiocyanate group directly onto the C4 position of the pyrazole ring under mild conditions opens new avenues for the rapid diversification of chemical libraries. As we delve deeper into the technical specifics, it becomes evident that this electrochemical protocol is poised to become a standard operation for the production of high-value nitrogen-containing heterocycles in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-aminopyrazole-4-thiocyanate derivatives has relied heavily on chemical oxidation systems that are inherently inefficient and environmentally burdensome. A notable precedent in the literature, reported by the Choudhury group in 2020, utilized hydrogen peroxide as the terminal oxidant to promote the thiocyanation of the C(sp2)-H bond. While effective in achieving the desired transformation, this method suffers from severe drawbacks that hinder its industrial applicability. Specifically, the protocol requires a massive excess of hydrogen peroxide, often up to 8 equivalents relative to the substrate. Such a high loading of oxidant not only escalates the raw material costs but also creates a complex reaction mixture filled with decomposition by-products and water. From a process safety perspective, handling large quantities of concentrated peroxide in the presence of organic solvents poses significant risks. Furthermore, the low atom economy of this oxidative system results in substantial waste generation, necessitating extensive downstream purification to remove residual oxidants and salts. For procurement managers, these factors translate into higher disposal costs and longer cycle times, making the conventional route economically unattractive for large-scale manufacturing.
The Novel Approach
In stark contrast, the electrochemical synthesis method described in patent CN115074760A revolutionizes this transformation by replacing chemical oxidants with electrical energy. This approach operates in an undivided cell, where the anodic oxidation of thiocyanate salts generates the active thiocyanating species in situ. The reaction proceeds under remarkably mild conditions, typically at room temperature, using simple and commercially available reagents such as potassium thiocyanate and acetic acid. The elimination of external chemical oxidants drastically simplifies the reaction matrix, leading to a cleaner crude product profile. This "green" methodology aligns perfectly with the principles of sustainable chemistry, offering high atom economy since the electrons serve as the traceless reagent. The versatility of this system is demonstrated by its compatibility with a wide range of substrates, including those with varying steric and electronic properties at the N1 and C3 positions of the pyrazole ring.

Moreover, the use of an undivided cell configuration reduces the capital expenditure required for specialized electrochemical equipment, making the technology accessible for both laboratory-scale optimization and pilot-plant operations. The reaction utilizes standard electrode materials, such as platinum sheets, which are durable and reusable, further enhancing the economic viability of the process. By shifting the driving force of the reaction from chemical potential to electrical potential, this novel approach effectively decouples the oxidation power from the reagent stoichiometry, allowing for precise control over the reaction kinetics. This level of control is crucial for maintaining the integrity of sensitive functional groups, such as the amino moiety, which might otherwise be susceptible to over-oxidation in harsh chemical environments.
Mechanistic Insights into Electrochemical C-H Thiocyanation
Understanding the mechanistic underpinnings of this electrochemical transformation is vital for R&D teams aiming to optimize the process for specific substrates. The reaction initiates at the anode surface, where the thiocyanate anion (SCN⁻) undergoes a single-electron oxidation to generate a thiocyanate radical (•SCN). This radical species is highly reactive and serves as the key electrophile in the subsequent C-H functionalization step. The 5-aminopyrazole substrate, activated by the electron-donating amino group and the heterocyclic nitrogen atoms, presents a nucleophilic character at the C4 position. The interaction between the electrophilic thiocyanate radical and the electron-rich C4 carbon leads to the formation of a radical cation intermediate. Subsequent deprotonation and rearomatization steps restore the aromaticity of the pyrazole ring, yielding the final 4-thiocyanato product. The presence of a weak acid, such as acetic acid, plays a dual role: it acts as a proton shuttle to facilitate the deprotonation step and helps maintain the conductivity of the electrolyte solution. The use of a mixed solvent system, typically comprising acetonitrile and water, ensures adequate solubility for both the organic substrate and the inorganic thiocyanate salt, promoting efficient mass transfer to the electrode interface.
From an impurity control perspective, the electrochemical method offers distinct advantages over chemical oxidation. In traditional methods, the presence of excess oxidant can lead to non-selective oxidation of the amino group or the pyrazole ring, resulting in complex impurity profiles that are difficult to separate. In the electrochemical regime, the oxidation potential can be finely tuned by adjusting the current density, ensuring that only the thiocyanate anion is oxidized while the substrate remains intact until the radical attack occurs. This selectivity minimizes the formation of over-oxidized by-products, such as sulfoxides or sulfones, which are common contaminants in sulfur chemistry. Furthermore, the absence of transition metal catalysts eliminates the risk of metal leaching, a critical quality attribute for pharmaceutical intermediates intended for clinical use. The rigorous control over reaction parameters allows for the consistent production of high-purity materials, reducing the burden on analytical quality control laboratories and accelerating the release of batches for downstream processing.
How to Synthesize 5-Aminopyrazole-4-Thiocyanate Efficiently
The practical implementation of this electrochemical synthesis is straightforward and amenable to standard laboratory setups, making it an attractive option for process development teams. The protocol involves a simple one-pot procedure where the substrate, thiocyanate source, and supporting electrolyte are combined in a reaction vessel equipped with electrodes. The reaction is driven by a constant current power supply, eliminating the need for complex potential control systems. Detailed operational parameters, including electrode spacing, stirring rates, and specific current densities, are critical for maximizing yield and reproducibility. The patent data provides a robust framework for scaling this reaction, highlighting the importance of optimizing the ratio of thiocyanate to substrate and the choice of acid promoter. For those looking to implement this technology, adhering to the standardized conditions outlined in the patent examples ensures the best outcomes.
- Prepare the electrolytic cell by adding thiocyanate salt, 5-aminopyrazole substrate, acid promoter, and solvent mixture into an undivided reaction vessel equipped with catalytic electrodes.
- Initiate the electrocatalytic reaction by applying a constant current at room temperature while stirring the mixture to ensure homogeneous mass transfer and efficient electron transfer at the electrode surface.
- Upon completion monitored by TLC, perform separation and purification via column chromatography or recrystallization to isolate the target 5-aminopyrazole-4-thiocyanate compound with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology presents a compelling value proposition centered on cost efficiency and supply reliability. The primary driver for cost reduction lies in the drastic simplification of the reagent profile. By eliminating the need for expensive and hazardous chemical oxidants like hydrogen peroxide or hypervalent iodine reagents, the raw material costs are significantly lowered. Additionally, the removal of transition metal catalysts negates the requirement for costly metal scavenging steps and the associated validation testing for residual metals, which is a mandatory regulatory requirement for API manufacturing. This streamlining of the process not only reduces direct material costs but also shortens the overall production cycle time, allowing for faster turnaround on customer orders. The use of commodity chemicals such as potassium thiocyanate and acetic acid ensures a stable and secure supply chain, insulating the manufacturing process from the volatility associated with specialty reagent markets.
- Cost Reduction in Manufacturing: The economic benefits of this electrochemical route are multifaceted, extending beyond simple reagent savings. The high atom economy of the reaction means that a larger proportion of the input mass is converted into the desired product, minimizing waste disposal fees which constitute a significant portion of operational expenditures in the fine chemical industry. The mild reaction conditions, operating effectively at room temperature, reduce the energy consumption associated with heating or cooling large reactor vessels. Furthermore, the simplified work-up procedure, often requiring only basic extraction and chromatography, reduces the consumption of solvents and silica gel. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), providing a competitive edge in the pricing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the electrochemical process. The reliance on electricity as the primary "reagent" decouples production capacity from the logistics of shipping hazardous oxidants, which are often subject to strict transportation regulations and storage limitations. The equipment required, such as undivided cells and DC power supplies, is widely available and easy to maintain, reducing the risk of production downtime due to equipment failure. Moreover, the tolerance of the reaction to various substrates allows for flexible manufacturing campaigns, enabling producers to switch between different pyrazole derivatives with minimal changeover time. This flexibility is crucial for meeting the dynamic demands of the pharmaceutical market, where quick adaptation to new drug candidates is essential.
- Scalability and Environmental Compliance: Scaling electrochemical reactions from the bench to the plant scale is increasingly feasible with modern flow chemistry technologies, although the patent demonstrates success in batch mode as well. The inherent safety of the process, characterized by the absence of exothermic oxidation events and hazardous reagents, facilitates easier regulatory approval for new manufacturing facilities. From an environmental standpoint, the reduction in chemical waste aligns with global sustainability goals and stringent environmental regulations, such as REACH in Europe. This compliance not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturer, a factor that is becoming increasingly important for multinational clients when selecting suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 5-aminopyrazole-4-thiocyanates. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances is key to evaluating the feasibility of integrating this technology into existing production workflows.
Q: What are the primary advantages of this electrochemical method over traditional oxidation?
A: Unlike traditional methods requiring stoichiometric amounts of hazardous chemical oxidants like hydrogen peroxide, this electrochemical approach uses electrons as the primary oxidant. This eliminates heavy metal waste, significantly improves atom economy, and simplifies downstream purification processes.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes standard undivided cells and commercially available electrode materials like platinum or carbon. The mild reaction conditions (room temperature) and absence of unstable oxidants make it highly suitable for scale-up in continuous flow or batch electrochemical reactors.
Q: What is the regioselectivity of the thiocyanation reaction?
A: The reaction exhibits excellent regioselectivity for the C4 position of the pyrazole ring. The electrochemical generation of thiocyanate radicals allows for precise electrophilic attack at the electron-rich C4 site, even in the presence of sensitive amino groups, minimizing isomeric by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aminopyrazole-4-Thiocyanate Supplier
The electrochemical synthesis of 5-aminopyrazole-4-thiocyanate compounds represents a frontier in green chemical manufacturing, offering a pathway to high-quality intermediates with minimal environmental impact. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging our deep expertise in organic electrosynthesis to deliver superior products to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical companies. We understand that the transition to new synthetic technologies requires trust and proven capability, and our track record in process optimization and scale-up serves as a testament to our commitment to excellence.
We invite you to explore the potential of this innovative synthesis route for your specific drug development projects. Whether you require custom synthesis of novel pyrazole derivatives or reliable supply of established intermediates, our technical team is ready to assist. Contact our Customized Cost-Saving Analysis department today to discuss how we can tailor this electrochemical process to your unique requirements. We encourage you to reach out to our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and value in your supply chain.
