Advanced Metal-Free Synthesis of 1,2,4-Benzotriazine Derivatives for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to access bioactive scaffolds. A significant breakthrough in this domain is detailed in patent CN114957145A, which discloses a novel preparation method for 1,2,4-benzotriazine derivatives. This class of compounds is renowned for its pervasive presence in medicinal chemistry, serving as a critical core structure for numerous therapeutic agents ranging from anticancer reagents like SN29751 to potent microbiocides and SGLT2 inhibitors. The traditional reliance on multi-step pre-assembly and harsh reaction conditions has long been a bottleneck for the rapid development of these valuable molecules. However, the methodology outlined in this patent introduces a paradigm shift by utilizing a direct [5+1] tandem cyclization strategy. This approach bypasses the need for complex precursor functionalization, offering a streamlined route that is both economically viable and environmentally benign. For R&D teams seeking to optimize their synthetic pipelines, this technology represents a substantial opportunity to enhance purity profiles while reducing the overall carbon footprint of the manufacturing process.
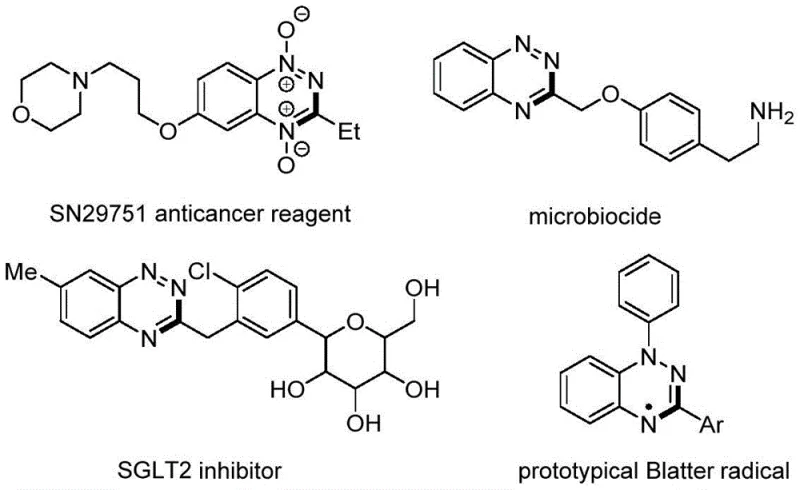
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-benzotriazine skeleton has been fraught with synthetic challenges that hinder efficient commercial production. Classical methods, such as the Bischler synthesis reported in the late 19th century, often necessitate the use of (2-nitrophenyl) acetyl hydrazide as a starting material, subjecting it to a rigorous sequence of reduction, nucleophilic addition, and oxidation steps. More modern approaches have attempted to address these inefficiencies through transition metal catalysis. For instance, copper-catalyzed domino reactions involving 2-haloanilines and hydrazides, or palladium-catalyzed couplings, have been explored. While these methods offer some improvements, they introduce significant drawbacks for industrial application. The primary concern is the reliance on expensive transition metals like palladium and copper, which not only inflate raw material costs but also create stringent regulatory hurdles regarding heavy metal residues in Active Pharmaceutical Ingredients (APIs). Furthermore, these conventional routes typically require the pre-assembly of specific functional groups, such as halogen atoms, on the open-chain molecular precursors before the final ring-closure can occur. This pre-functionalization adds extra synthetic steps, lowers overall atom economy, and generates additional waste streams, making the process less attractive for large-scale manufacturing where cost reduction in pharmaceutical intermediate manufacturing is a top priority.
The Novel Approach
In stark contrast to the legacy methodologies, the invention described in CN114957145A presents a revolutionary metal-free strategy that fundamentally simplifies the synthetic architecture. The core of this innovation lies in the direct reaction between 1-(trifluoromethanesulfonyl)-1H-benzotriazole compounds and sulfonium salts. This [5+1] cycloaddition tandem reaction proceeds without the need for any transition metal catalysts, effectively eliminating the risk of metal contamination and the associated purification costs. The reaction design is remarkably elegant; it allows for the direct formation of the 1,2,4-benzotriazine ring system from readily available starting materials without the tedious pre-assembly of functional groups. By leveraging the reactivity of sulfur ylides generated in situ, the process achieves high reaction yields under exceptionally mild conditions. This simplicity translates directly into operational efficiency, as it removes the need for specialized equipment required for handling air-sensitive catalysts or high-pressure hydrogenation. The result is a robust, green, and highly efficient synthetic route that provides a new avenue for the industrial production of 1,2,4-benzotriazine derivatives, addressing the critical pain points of cost, safety, and environmental compliance simultaneously.
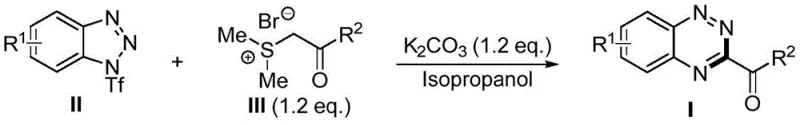
Mechanistic Insights into Sulfur Ylide-Mediated [5+1] Cyclization
To fully appreciate the technical superiority of this method, one must delve into the mechanistic intricacies that drive the transformation. The reaction initiates with the generation of a reactive sulfur ylide species from the sulfonium salt precursor, facilitated by the action of a mild acid binding agent such as potassium carbonate. This ylide then acts as a potent nucleophile, attacking the electron-deficient 1-(trifluoromethanesulfonyl)-1H-benzotriazole substrate. This nucleophilic attack triggers a ring-opening event in the benzotriazole moiety, creating a transient intermediate that is primed for cyclization. Subsequently, an intramolecular nucleophilic attack occurs where a nitrogen anion targets the alpha-carbon of the sulfur ylide fragment. This step is crucial as it leads to the expulsion of dimethyl sulfide and the closure of the new heterocyclic ring. The final stage involves the elimination of trifluoromethanesulfinic acid under basic conditions, resulting in the aromatization of the system to yield the stable 1,2,4-benzotriazine derivative. This cascade of events occurs seamlessly in a single pot, showcasing a high degree of chemoselectivity. The mechanism explains the broad substrate scope observed in the patent data, where various electron-withdrawing and electron-donating groups are well-tolerated. For R&D directors, understanding this mechanism is vital for troubleshooting and optimizing the process for specific analogues, ensuring that the electronic properties of substituents do not impede the critical ring-opening or cyclization steps.
Furthermore, the impurity profile of this reaction is inherently cleaner compared to metal-catalyzed alternatives. In traditional copper or palladium-catalyzed processes, side reactions such as homocoupling or incomplete dehalogenation can lead to difficult-to-remove impurities that compromise the purity specifications required for pharmaceutical grades. In this metal-free protocol, the primary byproducts are dimethyl sulfide and trifluoromethanesulfinic acid salts, which are generally easier to separate from the organic product during the workup phase. The use of potassium carbonate as the base ensures that the reaction medium remains sufficiently basic to drive the aromatization without being so harsh as to degrade sensitive functional groups like esters or nitriles that might be present on the substrate. This gentle yet effective control over the reaction environment minimizes the formation of polymeric byproducts or decomposition products. Consequently, the downstream purification process, often achieved via standard silica gel column chromatography as demonstrated in the examples, becomes more straightforward. This mechanistic clarity provides a solid foundation for scaling the process, as the reaction kinetics are predictable and the potential failure modes are well-understood, allowing for precise control over the quality of the high-purity pharmaceutical intermediates produced.
How to Synthesize 1,2,4-Benzotriazine Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-plant production. The standard procedure involves charging a reaction vessel with the 1-(trifluoromethanesulfonyl)-1H-benzotriazole substrate and the appropriate sulfonium salt in a molar ratio of approximately 1:1.2. Isopropanol serves as the preferred reaction solvent due to its ability to dissolve both organic reactants and the inorganic base while maintaining a green solvent profile. Potassium carbonate is added as the acid scavenger, typically in a slight excess to ensure complete conversion. The reaction is conducted under an air atmosphere at temperatures ranging from 20°C to 40°C, eliminating the need for cryogenic cooling or high-temperature heating. Monitoring the reaction progress is straightforward, with complete consumption of starting materials typically observed within 5 to 10 hours. Upon completion, the mixture is subjected to a simple workup involving concentration and purification via column chromatography. This operational simplicity significantly reduces the technical barrier for adoption, allowing procurement and supply chain teams to source the necessary raw materials without requiring specialized handling protocols for air-sensitive reagents. The detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below.
- Combine 1-(trifluoromethanesulfonyl)-1H-benzotriazole, sulfonium salt, and potassium carbonate in isopropanol under an air atmosphere.
- Stir the reaction mixture at room temperature (20-40°C) for approximately 8 hours until the starting material is fully consumed.
- Purify the crude product via silica gel column chromatography using a petroleum ether and ethyl acetate mixture to isolate the target 1,2,4-benzotriazine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift to this metal-free methodology offers compelling strategic advantages. The elimination of transition metal catalysts is not merely a technical improvement; it is a significant economic driver. Palladium and copper catalysts are subject to volatile market pricing and supply chain disruptions, and their removal from the process stabilizes the cost structure of the final intermediate. Moreover, the absence of heavy metals simplifies the regulatory filing process for new drug applications, as extensive testing for residual metals is no longer required, accelerating time-to-market for downstream API manufacturers. The use of inexpensive and widely available reagents like sulfonium salts and benzotriazole derivatives further contributes to substantial cost savings in pharmaceutical intermediate manufacturing. These raw materials are commodity chemicals with robust global supply chains, ensuring that production schedules are not held hostage by the scarcity of exotic reagents.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive transition metal catalysts. In conventional synthesis, the cost of palladium or copper salts, along with the specialized ligands often required to maintain their activity, can constitute a significant portion of the batch cost. By replacing these with cheap inorganic bases like potassium carbonate, the direct material cost is drastically reduced. Additionally, the simplified workup procedure eliminates the need for expensive metal scavenging resins or complex extraction protocols designed to lower metal residues to ppm levels. This streamlining of the purification process reduces solvent consumption and labor hours, leading to a leaner manufacturing operation. The high yields reported in the patent examples, often exceeding 80% for a wide range of substrates, mean that less raw material is wasted, further enhancing the overall process economics and maximizing the return on investment for every kilogram of product produced.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical in the pharmaceutical industry, and this synthesis route bolsters reliability by relying on commoditized feedstocks. The key starting materials, 1-(trifluoromethanesulfonyl)-1H-benzotriazoles and sulfonium salts, are structurally simple and can be sourced from multiple suppliers globally, reducing the risk of single-source dependency. The reaction conditions are equally forgiving; operating under an air atmosphere at near-ambient temperatures means that the process does not require specialized inert gas infrastructure or high-energy heating systems. This flexibility allows for production in a wider range of facilities, including those with limited utility capabilities, thereby expanding the potential manufacturing network. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates, as the risk of batch failure due to equipment malfunction or utility fluctuation is minimized. The robustness of the chemistry ensures consistent output, enabling reliable long-term supply agreements with downstream partners.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, this protocol aligns perfectly with sustainability goals. The use of isopropanol, a relatively benign solvent, replaces more hazardous polar aprotic solvents often used in metal-catalyzed cross-couplings. The absence of heavy metal waste simplifies effluent treatment, reducing the environmental burden and associated disposal costs. From a scalability perspective, the exothermic profile of the reaction is manageable, and the lack of gas evolution (other than trace amounts during workup) makes it safe for scale-up from grams to tons. The process avoids the use of toxic reagents like hydrazine derivatives in free form or hazardous oxidants, creating a safer working environment for plant operators. This inherent safety and environmental compatibility facilitate smoother regulatory approvals for commercial scale-up of complex pharmaceutical intermediates, ensuring that the manufacturing process remains compliant with increasingly stringent global environmental regulations while maintaining high throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this route for their own portfolios. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production lines or new product development pipelines.
Q: Does this synthesis method require transition metal catalysts?
A: No, this novel protocol is completely metal-free. It eliminates the need for expensive and toxic transition metals like palladium or copper, thereby avoiding heavy metal residue issues in the final pharmaceutical intermediates.
Q: What is the substrate scope for this reaction?
A: The method demonstrates excellent functional group tolerance. It successfully accommodates various substituents on the benzotriazole ring, including methyl, methoxy, fluoro, chloro, bromo, and nitro groups, as well as diverse sulfonium salt precursors.
Q: Are the reaction conditions suitable for industrial scale-up?
A: Yes, the reaction operates under mild conditions (20-40°C) in an air atmosphere using green solvents like isopropanol. These factors significantly simplify process engineering and enhance safety for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Benzotriazine Derivatives Supplier
The technological potential of this metal-free [5+1] cycloaddition route is immense, offering a clear path to high-quality, cost-effective 1,2,4-benzotriazine derivatives. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this innovative chemistry from the laboratory bench to full-scale commercial reality. As a seasoned CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate we deliver adheres to the highest industry standards. We understand the critical nature of supply chain continuity in the pharmaceutical sector and are committed to being a dependable extension of your manufacturing capabilities.
We invite you to explore how this advanced synthesis method can optimize your specific project requirements. Whether you are looking to reduce costs in API manufacturing or secure a stable supply of complex heterocyclic building blocks, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your quality and timeline expectations. Let us collaborate to bring your next generation of therapeutics to market faster and more efficiently.
