Revolutionizing Quinoline Production: A Metal-Free Synthetic Route For Scalable Pharmaceutical Intermediates
Revolutionizing Quinoline Production: A Metal-Free Synthetic Route For Scalable Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is undergoing a significant transformation with the introduction of greener, more efficient synthetic methodologies. Patent CN110724094A discloses a groundbreaking approach for the synthesis of quinoline compounds, a privileged scaffold ubiquitous in medicinal chemistry and material science. This technology leverages a novel [5+1] cyclization strategy between diazocarbonyl compounds and 2-vinylaniline derivatives, notably operating without the need for transition metal catalysts. 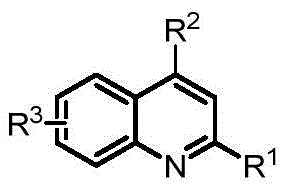 For R&D directors and procurement specialists alike, this represents a paradigm shift from traditional metal-catalyzed processes to a more sustainable, cost-effective, and operationally simple protocol. The ability to construct the quinoline core under metal-free conditions addresses critical pain points in API manufacturing, particularly regarding heavy metal residue limits and complex purification workflows. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative pathways is essential for maintaining competitive advantage in the global supply chain.
For R&D directors and procurement specialists alike, this represents a paradigm shift from traditional metal-catalyzed processes to a more sustainable, cost-effective, and operationally simple protocol. The ability to construct the quinoline core under metal-free conditions addresses critical pain points in API manufacturing, particularly regarding heavy metal residue limits and complex purification workflows. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative pathways is essential for maintaining competitive advantage in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinoline rings utilizing diazo compounds has relied heavily on transition metal catalysis. Conventional strategies often employ expensive and sensitive catalysts based on rhodium, copper, or other precious metals to facilitate carbene transfer and subsequent cyclization. These methods typically necessitate rigorous reaction conditions, including high temperatures reaching 150°C, to drive the transformation to completion. From a commercial manufacturing perspective, the reliance on transition metals introduces significant logistical and financial burdens. The cost of the catalysts themselves is substantial, but the hidden costs associated with their removal are even more critical. Pharmaceutical regulations impose strict limits on residual heavy metals in final drug substances, requiring dedicated scavenging steps, additional chromatography, or complex crystallization protocols to ensure compliance. Furthermore, metal catalysts can sometimes lead to unpredictable side reactions or decomposition of sensitive functional groups, limiting the substrate scope and overall yield of the process.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110724094A offers a streamlined alternative that eliminates the need for any transition metal catalyst. By utilizing strong acidic solvents such as hexafluoroisopropanol (HFIP) or trifluoroacetic acid (TFA), the reaction proceeds efficiently through a proton-promoted mechanism. 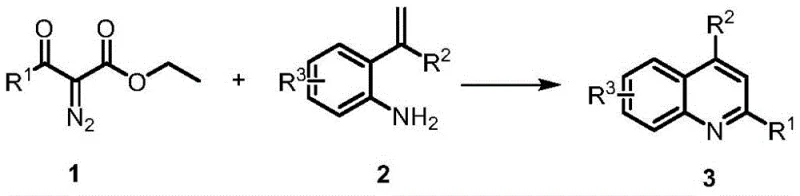 This metal-free approach not only drastically reduces the raw material costs by removing expensive catalysts from the bill of materials but also simplifies the downstream processing significantly. The reaction conditions are milder, typically operating between 80°C and 150°C, with many examples achieving high conversion at 120°C within just 3 hours. This reduction in thermal stress and chemical complexity enhances the robustness of the process, making it far more attractive for scale-up. The elimination of metal residues means that the purification workflow is less cumbersome, directly translating to higher throughput and reduced production timelines for high-purity pharmaceutical intermediates.
This metal-free approach not only drastically reduces the raw material costs by removing expensive catalysts from the bill of materials but also simplifies the downstream processing significantly. The reaction conditions are milder, typically operating between 80°C and 150°C, with many examples achieving high conversion at 120°C within just 3 hours. This reduction in thermal stress and chemical complexity enhances the robustness of the process, making it far more attractive for scale-up. The elimination of metal residues means that the purification workflow is less cumbersome, directly translating to higher throughput and reduced production timelines for high-purity pharmaceutical intermediates.
Mechanistic Insights into Metal-Free [5+1] Cyclization
The core of this innovation lies in the unique reactivity of diazocarbonyl compounds under strongly acidic conditions. In the absence of a metal catalyst to generate a metal-carbene species, the reaction is driven by the protonation of the diazo functionality or the activation of the vinyl group by the acidic solvent medium. The proposed mechanism involves the generation of a reactive carbene-like intermediate or an activated electrophilic species from the diazo compound, which subsequently undergoes nucleophilic attack by the amino group of the 2-vinylaniline. This initiates a cascade of intramolecular cyclization events, effectively stitching together the carbon and nitrogen atoms to form the fused quinoline ring system. The use of fluorinated alcohols like HFIP is particularly crucial, as these solvents possess high ionizing power and hydrogen-bond donating ability, which stabilizes charged intermediates and facilitates the cyclization without external metallic assistance. This mechanistic pathway ensures high atom economy and minimizes the formation of metal-associated byproducts.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for quality assurance teams. Traditional metal-catalyzed reactions often suffer from issues related to catalyst decomposition, leading to metal-containing impurities that are difficult to characterize and remove. By avoiding metals entirely, the impurity profile of the resulting quinoline compounds is significantly cleaner, consisting primarily of organic byproducts that are easier to separate via standard silica gel chromatography or recrystallization. The patent data demonstrates excellent tolerance for various electronic environments, with electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like halogens and esters, being well-tolerated on the aromatic rings. This broad substrate compatibility suggests that the mechanistic pathway is robust against steric and electronic variations, providing a versatile platform for synthesizing diverse libraries of quinoline derivatives for drug discovery programs.
How to Synthesize 2,4-Diphenylquinoline Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves charging a pressure-resistant vessel with the requisite diazocarbonyl compound and 2-vinylaniline derivative in a molar ratio ranging from 10.0:1.0 to 1.0:2.0, although a slight excess of the diazo component is often preferred to drive the reaction to completion. The concentration of the 2-vinylaniline substrate is typically maintained between 0.1 and 0.2 mol/L in the chosen solvent system. Following the addition of reagents, the vessel is purged with an inert gas such as argon to prevent oxidative degradation of the reactive intermediates. The mixture is then heated to the target temperature, typically around 120°C, and stirred for a period of 2 to 24 hours depending on the specific substrate reactivity. Upon completion, the reaction mixture is worked up via liquid-liquid extraction using ethyl acetate and aqueous sodium bicarbonate, followed by drying and concentration. The crude product is then purified by silica gel column chromatography to afford the target quinoline compound in high purity.
- Prepare the reaction mixture by dissolving the diazocarbonyl compound and 2-vinylaniline derivative in a solvent system comprising hexafluoroisopropanol or trifluoroacetic acid.
- Purge the reaction vessel with inert gas such as argon to ensure an oxygen-free environment, then seal the pressure-resistant tube.
- Heat the mixture to a temperature between 80°C and 150°C for 2 to 24 hours, followed by extraction and silica gel chromatography for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route presents compelling economic and operational benefits. The most immediate impact is seen in the reduction of manufacturing costs. By eliminating the requirement for precious metal catalysts, the direct material costs are lowered, and the supply chain becomes less vulnerable to fluctuations in the prices of rare earth metals. Furthermore, the simplified workup procedure reduces the consumption of specialized scavenging resins and solvents associated with metal removal, contributing to substantial cost savings in the overall production budget. The operational simplicity also means that the process can be executed in standard glass-lined or stainless steel reactors without the need for specialized equipment to handle air-sensitive metal catalysts, thereby enhancing the flexibility of existing manufacturing facilities.
- Cost Reduction in Manufacturing: The exclusion of transition metal catalysts removes a significant cost driver from the production equation. Precious metals like rhodium or palladium are not only expensive to purchase but also represent a sunk cost as they are consumed or lost during the process. Additionally, the regulatory requirement to reduce metal residues to ppm levels often necessitates expensive purification steps. By adopting this metal-free protocol, manufacturers can bypass these costly remediation steps entirely. The use of commercially available and relatively inexpensive solvents like trifluoroacetic acid further optimizes the cost structure. This streamlined approach allows for a more competitive pricing model for the final quinoline intermediates, providing a strategic advantage in tender negotiations for API contracts.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials. Diazocarbonyl compounds and 2-vinylanilines are accessible from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which do not require stringent exclusion of moisture or oxygen beyond standard inert gas purging, simplifies logistics and storage requirements. This reliability ensures consistent delivery schedules and reduces the likelihood of production delays caused by catalyst shortages or sensitivity issues. For long-term projects, having a synthesis route that is less dependent on specialized reagents guarantees a more stable supply of critical pharmaceutical intermediates, safeguarding the continuity of drug development pipelines.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden challenges, particularly with exothermic metal-catalyzed reactions. This metal-free method exhibits excellent scalability due to its thermal stability and lack of sensitive catalytic species. The reaction can be safely scaled from 100 kgs to 100 MT annual commercial production with minimal engineering changes. Moreover, the environmental footprint is significantly reduced. The absence of heavy metals eliminates the generation of hazardous metal-contaminated waste streams, simplifying waste treatment and disposal compliance. This aligns with modern green chemistry principles and corporate sustainability goals, making the process more attractive to environmentally conscious partners and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel quinoline synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN110724094A, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of this quinoline synthesis method over traditional routes?
A: The primary advantage is the complete elimination of transition metal catalysts. Traditional methods often require expensive metals like rhodium or copper and harsh conditions up to 150°C, whereas this method operates under metal-free conditions using acidic promoters, significantly reducing impurity profiles and downstream purification costs.
Q: What types of substituents are tolerated in this synthetic protocol?
A: The protocol demonstrates excellent substrate scope, tolerating a wide range of functional groups including alkyl, alkoxy, thioether, ester, halogens (fluorine, chlorine, bromine), and fused aromatic systems like naphthyl groups on both the aniline and diazo components.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its operational simplicity. It avoids sensitive metal catalysts that require strict removal protocols for pharmaceutical grades, uses readily available starting materials, and employs robust reaction conditions that are easily adaptable to industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Compounds Supplier
The technological advancements detailed in patent CN110724094A underscore the potential for more efficient and sustainable production of high-value heterocyclic intermediates. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such chemical innovations. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs in place to guarantee that every batch of quinoline compounds meets the highest international standards required for pharmaceutical applications.
We invite you to explore how this metal-free synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us collaborate to bring your next-generation therapeutics to market faster and more efficiently.
