Advanced Chiral Catalysis for Commercial Scale S-Nicotine Production
Introduction to Next-Generation Nicotine Synthesis
The global demand for high-purity nicotine, particularly the biologically active S-(-)-enantiomer, has driven significant innovation in organic synthesis methodologies. Traditional reliance on tobacco extraction is increasingly insufficient due to impurity profiles and supply chain volatility associated with agricultural sourcing. A pivotal advancement in this field is documented in patent CN113999201A, which outlines a robust synthetic preparation method for optical active S-(-)-nicotine. This technology represents a paradigm shift from extraction-based or racemic-resolution processes to a direct asymmetric synthesis strategy. By utilizing 3-pyridine acetonitrile and N-substituted pyrrolidones as key building blocks, the process achieves high stereocontrol without the inherent yield losses of resolution. For R&D directors and procurement specialists, understanding this pathway is critical for securing a reliable supply of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of nicotine has relied heavily on extraction from Nicotiana tabacum, a process fraught with challenges regarding consistency and purification. The extracted material often contains a complex matrix of alkaloids and plant-derived impurities that are difficult to separate, leading to variable quality that fails to meet stringent pharmaceutical standards. Alternatively, chemical synthesis routes described in prior art, such as those utilizing nicotinic acid ethyl ester and N-vinylpyrrolidone, typically generate racemic nicotine. To obtain the desired S-enantiomer, these methods require a subsequent resolution step using chiral resolving agents. This resolution process is inherently inefficient, theoretically capping the maximum yield at 50% for the desired isomer while generating an equivalent amount of unwanted R-enantiomer waste. Furthermore, the operational complexity of crystallization and recycling resolving agents adds significant cost and time burdens to the manufacturing timeline.
The Novel Approach
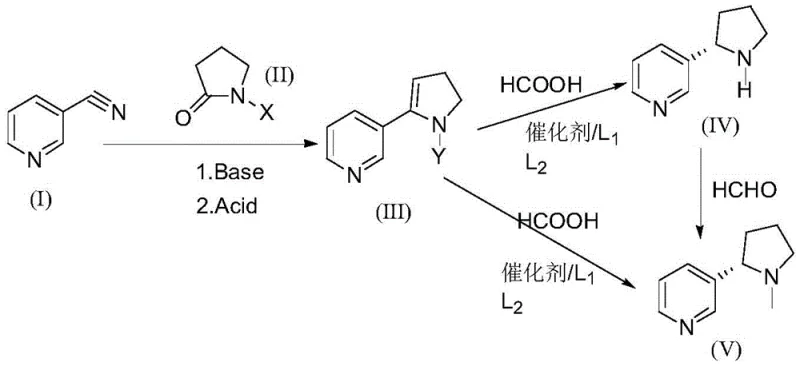
The methodology presented in the referenced patent overcomes these inefficiencies by employing a direct asymmetric catalytic reduction. Instead of creating a racemic mixture and separating it, the process constructs the chiral center directly during the reduction of a prochiral intermediate. The synthesis begins with the condensation of 3-pyridine acetonitrile and an N-substituted pyrrolidone to form a dihydropyrrole intermediate. This intermediate is then subjected to asymmetric transfer hydrogenation using a chiral ruthenium catalyst system. This approach eliminates the need for resolution, effectively doubling the theoretical yield compared to racemic routes and simplifying the downstream purification process. The streamlined nature of this synthesis not only enhances overall productivity but also aligns with green chemistry principles by reducing waste generation.
Mechanistic Insights into Asymmetric Transfer Hydrogenation
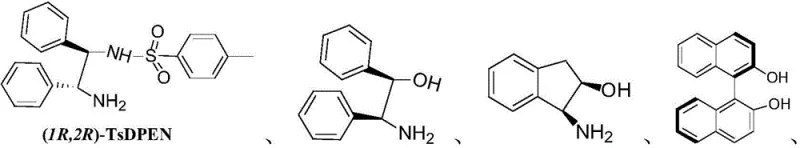
The core of this technological breakthrough lies in the enantioselective reduction of the 3-(N-Y-2,3-dihydropyrrolyl)pyridine intermediate. The reaction utilizes a formic acid-triethylamine azeotrope as the hydrogen source, facilitating a transfer hydrogenation mechanism mediated by a ruthenium complex. The stereochemical outcome is dictated by the chiral environment created by the ligand system surrounding the metal center. Specifically, the patent highlights the efficacy of chiral diamine ligands, such as (1R,2R)-TsDPEN, in conjunction with arene-ruthenium precursors. These ligands coordinate with the metal to form a rigid chiral pocket that directs the hydride transfer to a specific face of the imine or enamine double bond within the intermediate. This precise control ensures the formation of the S-configuration with high enantiomeric excess, as demonstrated by experimental data showing ee values exceeding 90%.
Impurity control is another critical aspect of this mechanism. The use of mild reaction conditions, typically between 20°C and 55°C, minimizes thermal degradation and side reactions that often plague high-temperature syntheses. The choice of solvent systems, such as acetonitrile or toluene mixtures, further optimizes the solubility of intermediates and catalysts, ensuring homogeneous reaction kinetics. By avoiding harsh reducing agents like lithium aluminum hydride or sodium borohydride in the chiral step, the process reduces the risk of over-reduction or non-selective background reactions. The result is a crude product with high HPLC purity, often surpassing 99% after simple workup and distillation, which significantly reduces the burden on final purification steps and ensures a clean impurity profile for regulatory submission.
How to Synthesize S-(-)-Nicotine Efficiently
Implementing this synthesis requires careful attention to the preparation of the dihydropyrrole precursor and the optimization of the catalytic reduction parameters. The initial condensation step involves the use of strong bases like sodium hydride to deprotonate the pyrrolidone, followed by nucleophilic attack on the nitrile. Once the intermediate is isolated, the focus shifts to the chiral reduction where catalyst loading and ligand ratio are paramount. The patent provides specific embodiments detailing the use of p-cymene ruthenium dichloride dimers paired with chiral ligands to achieve optimal results. For a comprehensive guide on executing this protocol with precision, please refer to the standardized operating procedures outlined below.
- Condense 3-pyridine acetonitrile with N-methyl-2-pyrrolidone using a strong base like sodium hydride in toluene at 80°C to form the dihydropyrrole intermediate.
- Perform asymmetric transfer hydrogenation on the intermediate using a ruthenium catalyst and chiral ligand (e.g., TsDPEN) in a formic acid/triethylamine system at 25-30°C.
- Purify the resulting S-(-)-nicotine via vacuum distillation to achieve high optical purity and chemical purity suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative benefits for supply chain stability and cost management in the fine chemical sector. The elimination of the resolution step is the primary driver of economic value, as it removes the need for expensive chiral resolving agents and the associated loss of 50% of the synthesized material. This improvement in atom economy translates directly into lower raw material consumption per kilogram of final product. Additionally, the starting materials, such as 3-pyridine acetonitrile and N-methyl-2-pyrrolidone, are commodity chemicals with robust global supply chains, mitigating the risk of raw material shortages that often affect botanical extracts. The ability to source stable, synthetic precursors ensures consistent pricing and availability, which is crucial for long-term production planning.
- Cost Reduction in Manufacturing: The process significantly lowers manufacturing costs by streamlining the synthetic sequence. By bypassing the resolution stage, manufacturers save on the costs of resolving agents, additional solvents for recrystallization, and the energy required for multiple heating and cooling cycles. The high yield of the asymmetric reduction step means that less starting material is needed to produce the same amount of active ingredient. Furthermore, the use of transfer hydrogenation with formic acid avoids the need for high-pressure hydrogenation equipment, reducing capital expenditure on specialized reactors and enhancing operational safety. These factors combine to create a leaner, more cost-effective production model.
- Enhanced Supply Chain Reliability: Reliance on agricultural sources for nicotine introduces variability due to weather, pests, and geopolitical factors affecting tobacco farming. This synthetic alternative decouples production from agricultural cycles, offering a consistent, year-round manufacturing capability. The use of standard organic solvents and commercially available catalysts ensures that the supply chain is resilient to disruptions. For procurement managers, this means the ability to secure long-term contracts with fixed specifications, reducing the volatility associated with commodity pricing of natural extracts. The synthetic route provides a dependable source of high-purity material that can scale with market demand without the lead times associated with crop harvesting.
- Scalability and Environmental Compliance: The reaction conditions described are highly amenable to scale-up, operating at near-ambient temperatures and atmospheric pressure. This simplicity facilitates the transition from laboratory benchtop to multi-ton commercial production without significant re-engineering of the process. From an environmental standpoint, the reduction in waste is substantial; avoiding the mother liquors from resolution steps and minimizing solvent usage contributes to a lower E-factor. The process generates fewer hazardous byproducts compared to traditional metal hydride reductions, simplifying waste treatment and disposal. This alignment with environmental regulations reduces compliance costs and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific supply chain needs. The answers reflect the practical realities of scaling chiral catalysis in an industrial setting.
Q: How does this synthetic route improve upon traditional nicotine extraction?
A: Traditional extraction from tobacco leaves often yields products with difficult-to-remove impurities. This synthetic method uses defined chemical starting materials to produce high-purity S-(-)-nicotine with controlled stereochemistry, eliminating biological variability.
Q: What is the advantage of this method over racemic synthesis followed by resolution?
A: Conventional methods often produce racemic nicotine requiring a resolution step, which theoretically limits yield to 50% and adds processing complexity. This patent describes a direct asymmetric synthesis that avoids the resolution step, significantly improving atom economy and overall process efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent highlights mild reaction conditions (20-55°C for the reduction step) and the use of commercially available solvents like toluene and acetonitrile. The avoidance of complex resolution steps makes the workflow more amenable to continuous or large-batch manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(-)-Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic routes like the one described in CN113999201A for securing the future of pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of S-(-)-nicotine meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of chiral catalysis, delivering products with consistent optical purity and chemical quality.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this synthetic route for your specific volume requirements. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to build a more efficient and reliable supply network for high-value chiral intermediates.
