Scalable Synthesis of Dabigatran Etexilate via Solid-State Intermediate Isolation
Scalable Synthesis of Dabigatran Etexilate via Solid-State Intermediate Isolation
The pharmaceutical industry continuously seeks robust manufacturing routes for critical anticoagulant therapies, specifically targeting the efficient production of thrombin inhibitors like Dabigatran and its prodrug, Dabigatran Etexilate. Patent CN102985416A introduces a transformative methodology that addresses long-standing bottlenecks in the synthesis of these complex molecules by focusing on the solid-state isolation of key intermediates. Unlike traditional approaches that rely heavily on chromatographic purification and excessive reagent usage, this novel process leverages the crystallization of imido-ester and amidine intermediates to achieve superior purity and operational simplicity. By shifting the paradigm from liquid-phase processing to controlled solid-state separation, manufacturers can significantly enhance the reproducibility and safety profile of the synthesis. This technical advancement is particularly relevant for reliable pharmaceutical intermediate suppliers aiming to secure supply chains for high-volume API production without compromising on quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
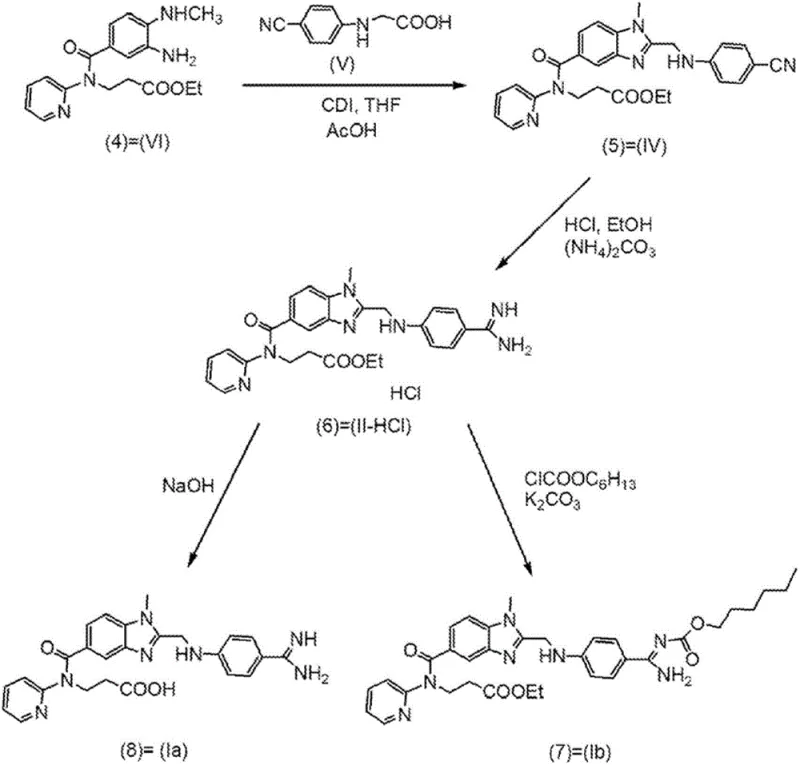
Historically, the synthesis of Dabigatran Etexilate, as described in earlier literature such as WO98/37075, has been plagued by significant downstream processing challenges that hinder industrial scalability. A primary drawback of these conventional routes is the reliance on chromatographic purification to isolate critical intermediates, specifically the conversion of the nitrile precursor to the amidine species. Chromatography is inherently batch-limited, solvent-intensive, and difficult to scale beyond pilot plant levels, creating a major bottleneck for commercial scale-up of complex pharmaceutical intermediates. Furthermore, prior art methods often necessitate the use of massive stoichiometric excesses of ammonia or volatile ammonium salts—sometimes up to 11 equivalents—to drive the amidination reaction to completion. This excessive reagent usage results in the formation of substantial quantities of ammonium chloride byproducts, which are notoriously difficult to remove completely and can contaminate the final product, thereby complicating the subsequent acylation steps required to generate the etexilate prodrug.
The Novel Approach
The methodology disclosed in CN102985416A offers a decisive break from these inefficiencies by introducing a strategy centered on the isolation of intermediates in solid form, specifically the imido-ester hydrochloride (III-HCl) and the amidine hydrochloride (II-HCl). By converting the nitrile intermediate (IV) into the imido-ester salt (III-HCl) and isolating it as a solid, the process effectively creates a purification checkpoint that removes impurities before the critical amidination step. This solid-state isolation allows for the use of significantly reduced amounts of ammonia or ammonium salts, typically between 3 to 5 equivalents, compared to the excessive loads seen in older patents. The reduction in ammonium salt generation not only simplifies the workup procedure but also minimizes the risk of ammonium chloride contamination in the final active pharmaceutical ingredient. This approach facilitates cost reduction in pharmaceutical intermediate manufacturing by eliminating the need for expensive and time-consuming chromatographic columns, replacing them with standard filtration and crystallization unit operations that are easily adaptable to multi-ton production scales.
Mechanistic Insights into Solid-State Intermediate Purification
The core mechanistic advantage of this process lies in the controlled hydrolysis and salt formation of the imido-ester intermediate. When the nitrile compound (IV) is treated with hydrochloric acid in an alcoholic solvent like ethanol, it forms the imido-ester hydrochloride (III-HCl). The patent highlights that isolating this species in a crystalline or amorphous solid form is crucial because it acts as a 'chemical filter.' By precipitating the imido-ester, unreacted starting materials and side products remain in the mother liquor, ensuring that only the desired intermediate proceeds to the next stage. This is particularly important because the subsequent reaction with ammonia to form the amidine (II) is sensitive to impurities. If the imido-ester is not purified, residual acids or organic impurities can consume the ammonia reagent or lead to the formation of difficult-to-remove byproducts. The ability to characterize specific polymorphic forms of the amidine intermediate (such as Type I and Type V) via X-ray powder diffraction further ensures batch-to-batch consistency, a critical parameter for regulatory compliance in high-purity pharmaceutical intermediate supply.
Furthermore, the mechanism of amidination is optimized by minimizing the presence of ammonium chloride during the reaction. In conventional liquid-phase reactions, the accumulation of ammonium chloride can shift equilibria or cause solubility issues that trap the product in solution, necessitating complex extraction procedures. By starting with a purified solid imido-ester and using a controlled amount of ammonia source, such as ammonium carbonate or ammonia in ethanol, the reaction generates a manageable amount of salt that can be easily separated by filtration or solvent exchange. The use of anti-solvents like toluene allows for the precise precipitation of the amidine hydrochloride (II-HCl) with high chemical purity, often exceeding 99% without the need for further chromatographic refinement. This level of control over the solid-state chemistry ensures that the final acylation step to produce Dabigatran Etexilate proceeds with high efficiency, as the reactive amidine group is not deactivated by salt contaminants.
How to Synthesize Dabigatran Etexilate Efficiently
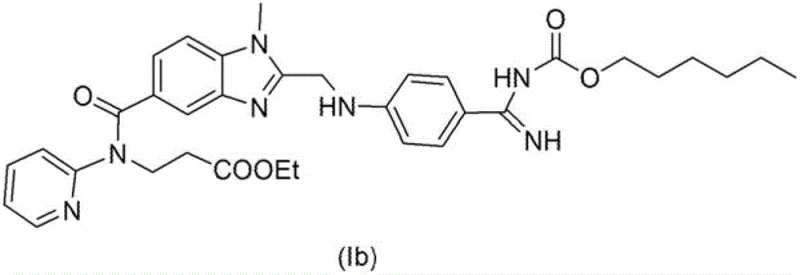
The synthesis of Dabigatran Etexilate via this improved route involves a sequence of well-defined unit operations that prioritize solid handling over liquid chromatography. The process begins with the preparation of the nitrile intermediate, which is then converted to the imido-ester hydrochloride in an alcoholic medium. Following isolation, the solid imido-ester is reacted with a controlled equivalent of ammonia to generate the amidine intermediate, which is subsequently crystallized and dried. The final step involves the acylation of the purified amidine with hexyl chloroformate to yield the target prodrug. This streamlined workflow is designed to maximize yield while minimizing waste generation, making it an ideal candidate for green chemistry initiatives in the fine chemical sector. For detailed operational parameters, temperature controls, and specific solvent ratios, please refer to the standardized synthesis guide below.
- Provide the imido-ester intermediate (III-HCl) in solid form, typically obtained by reacting the nitrile precursor with HCl in ethanol.
- React the solid imido-ester with 3-5 equivalents of ammonia or ammonium carbonate to form the amidine intermediate (II-HCl).
- Separate the amidine intermediate (II-HCl) as a solid crystal, wash to remove ammonium salts, and convert to the final ester (Ib) via acylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solid-state isolation process represents a strategic opportunity to de-risk the supply of critical anticoagulant intermediates. The elimination of chromatography not only reduces direct manufacturing costs but also drastically shortens the production cycle time, allowing for faster turnaround on large orders. The reliance on standard filtration and crystallization equipment means that the process can be implemented in existing multipurpose chemical plants without the need for specialized chromatographic skids, thereby enhancing supply chain flexibility. Moreover, the stability of the isolated solid intermediates, such as the various polymorphic forms of the amidine hydrochloride, allows for safer storage and transportation, reducing the risk of degradation during logistics. This robustness translates directly into reducing lead time for high-purity pharmaceutical intermediates and ensures a more predictable delivery schedule for downstream API manufacturers.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the complete removal of chromatographic purification steps, which are notoriously expensive due to high solvent consumption and silica gel costs. By replacing these with crystallization and filtration, the process achieves substantial cost savings through reduced solvent purchase and disposal expenses. Additionally, the reduction in ammonia usage from excessive equivalents to a near-stoichiometric amount lowers raw material costs and decreases the load on waste treatment facilities. The qualitative improvement in process efficiency means that labor hours per kilogram of product are significantly reduced, contributing to a lower overall cost of goods sold (COGS) without compromising product quality.
- Enhanced Supply Chain Reliability: The ability to isolate intermediates as stable solids provides a buffer against supply chain disruptions. Unlike liquid intermediates that may require immediate processing or cold chain storage, solid intermediates like the imido-ester hydrochloride can be stockpiled and quality-tested before proceeding to the next step. This decoupling of process steps allows manufacturers to manage inventory more effectively and respond to fluctuating market demands with greater agility. The use of common, non-hazardous solvents like ethanol and toluene further ensures that raw material availability is not a constraint, securing the continuity of supply for long-term contracts.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent volume and the elimination of silica waste align with increasingly stringent global regulations on pharmaceutical manufacturing emissions. The process generates significantly less hazardous waste, simplifying compliance with environmental protection standards and reducing the carbon footprint of the manufacturing site. The scalability of filtration and crystallization units is well-understood in the chemical industry, allowing for seamless scale-up from pilot batches to commercial production capacities of hundreds of tons. This ease of scale-up ensures that the technology remains viable and cost-effective as market demand for thrombin inhibitors continues to grow globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding the nuances of intermediate stability and purification mechanisms is essential for evaluating the feasibility of technology transfer.
Q: Why is solid-state isolation of the imido-ester intermediate critical for scalability?
A: Isolating the imido-ester (III-HCl) as a solid allows for the removal of impurities and excess reagents before the amidination step. This prevents the accumulation of ammonium chloride residues, which are difficult to remove and can interfere with the subsequent acylation reaction required to form Dabigatran Etexilate.
Q: How does this process reduce solvent consumption compared to prior art?
A: By utilizing solid-state crystallization and filtration instead of chromatographic purification, the process significantly reduces the volume of solvents required. The ability to precipitate intermediates directly from the reaction mixture minimizes the need for large volumes of mobile phases associated with column chromatography.
Q: What polymorphic forms of the amidine intermediate are described?
A: The patent describes multiple crystalline forms (Types I, II, III, IV, and V) of the amidine hydrochloride intermediate (II-HCl). Type I and Type V are highlighted as particularly suitable for industrial operations due to their stability and favorable filtration properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of solid-state isolation described in CN102985416A can be fully realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to verify the polymorphic form and chemical purity of every batch. Our commitment to technical excellence ensures that we can deliver high-purity Dabigatran Etexilate intermediates that meet the exacting standards required for regulatory filing and commercial drug manufacture.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for thrombin inhibitor production. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, demonstrating how our advanced manufacturing capabilities can drive value and security for your organization.
