Revolutionizing Formamide Production: A High-Efficiency Copper-Mediated Route for Pharmaceutical Intermediates
Revolutionizing Formamide Production: A High-Efficiency Copper-Mediated Route for Pharmaceutical Intermediates
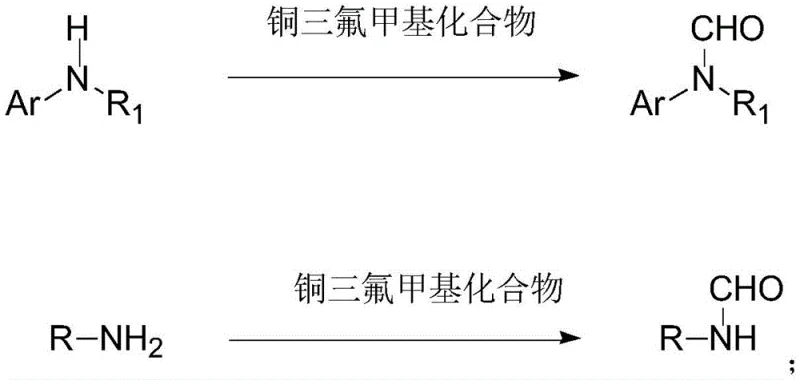
In the rapidly evolving landscape of fine chemical synthesis, the demand for efficient, safe, and scalable methods to produce nitrogen-containing heterocycles and amides is paramount. A groundbreaking development detailed in Chinese Patent CN116396180A introduces a transformative approach to synthesizing formamide compounds, a critical class of intermediates widely utilized in the manufacture of active pharmaceutical ingredients (APIs), agrochemicals, and functional materials. This novel methodology leverages a specific high-valent copper trifluoromethyl complex to achieve direct formylation of amines, bypassing the hazardous and cumbersome requirements of traditional carbonylation techniques. For R&D directors and procurement strategists alike, this technology represents a significant leap forward in process chemistry, offering a pathway to cost reduction in API manufacturing while simultaneously addressing critical supply chain vulnerabilities associated with hazardous reagent handling.
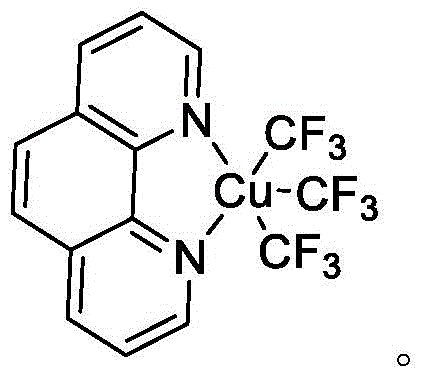
The core innovation lies in the utilization of a well-defined copper(III) species, specifically (Phen)CuIII(CF3)3, which acts as a unique reagent to convert readily available amine substrates into valuable formamide derivatives. Unlike conventional routes that often rely on toxic carbon monoxide gas under high pressure or expensive stoichiometric formylating agents, this protocol operates under relatively mild thermal conditions in a simple one-pot setup. The implications for industrial scalability are profound, as the method eliminates the need for specialized high-pressure reactors and complex gas handling systems, thereby lowering the barrier to entry for commercial scale-up of complex organic syntheses. As we delve deeper into the technical specifics, it becomes clear that this patent offers a robust solution for producing high-purity pharmaceutical intermediates with exceptional atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of formamides has been plagued by significant operational and safety challenges that hinder efficient large-scale production. Traditional methods typically involve the use of carbon monoxide (CO) as a C1 source in the presence of transition metal catalysts, a process known as carbonylation. While effective in theory, the practical application of CO carbonylation requires rigorous safety protocols due to the extreme toxicity of carbon monoxide, necessitating expensive containment infrastructure and continuous monitoring systems that drive up capital expenditure. Furthermore, alternative stoichiometric approaches using formic acid or formate salts often suffer from poor atom economy, generating substantial amounts of salt waste that complicate downstream purification and increase environmental disposal costs. These legacy processes frequently require harsh reaction conditions, such as strong acids or elevated temperatures, which can lead to the degradation of sensitive functional groups on complex amine substrates, resulting in lower yields and difficult-to-remove impurity profiles that compromise the quality of the final pharmaceutical intermediate.
The Novel Approach
In stark contrast to these legacy issues, the method disclosed in Patent CN116396180A utilizes a pre-formed copper trifluoromethyl complex to effect the transformation under remarkably benign conditions. This innovative route allows for the direct conversion of both primary and secondary amines into their corresponding formamides without the need for gaseous reagents or extreme pressures. The reaction proceeds efficiently in common organic solvents such as 1,4-dioxane or acetonitrile at temperatures around 100°C, making it compatible with standard glass-lined steel reactors found in most multipurpose chemical plants. By employing this specific copper complex, the process achieves high selectivity, minimizing the formation of side products and simplifying the purification workflow. This shift from gas-phase carbonylation to a solution-phase metal-mediated transformation not only enhances operator safety but also streamlines the manufacturing process, positioning it as an ideal candidate for reducing lead time for high-purity intermediates in a commercial setting.
Mechanistic Insights into Copper-Mediated Formylation
The efficacy of this synthesis relies on the unique reactivity of the tris(trifluoromethyl)copper(III) complex stabilized by a phenanthroline ligand. While the precise mechanistic pathway for the conversion of the trifluoromethyl group to a formyl moiety involves complex organometallic steps, the overall process is driven by the high oxidation state of the copper center, which facilitates the activation of the amine substrate. The phenanthroline ligand plays a crucial role in stabilizing the Cu(III) species, preventing premature decomposition and ensuring that the reactive species remains available for the transformation throughout the reaction duration. For secondary amines, the reaction proceeds smoothly in the absence of additives, suggesting a direct nucleophilic attack or insertion mechanism that is highly tolerant of various electronic environments on the aromatic ring. This robustness is evidenced by the successful conversion of substrates containing electron-donating groups like methoxy and electron-withdrawing groups like chloro, demonstrating the versatility required for diverse drug discovery campaigns.
Regarding impurity control, the one-pot nature of the reaction significantly reduces the risk of cross-contamination that often occurs in multi-step sequences. The use of a defined molecular catalyst rather than a heterogeneous mixture helps in maintaining a consistent impurity profile, which is critical for regulatory compliance in pharmaceutical manufacturing. When primary amines are employed, the addition of a mild acid such as trifluoroacetic acid is necessary to facilitate the reaction, likely by protonating intermediate species or activating the amine for nucleophilic attack. This controlled acidity ensures that the reaction proceeds to completion without promoting hydrolysis or other degradation pathways that could compromise the integrity of the final product. The result is a clean reaction mixture that, after a simple workup involving dilution and filtration, yields the target formamide with high purity, often exceeding 90% isolated yield in optimized conditions, thereby meeting the stringent specifications demanded by reliable pharmaceutical intermediate suppliers.
How to Synthesize Formamide Compounds Efficiently
The practical implementation of this technology is straightforward, designed to be easily adopted by process chemists looking to optimize their synthetic routes. The protocol involves charging a reaction vessel with the copper complex under an inert atmosphere, followed by the addition of the amine substrate and solvent. The mixture is then heated to reflux, allowing the transformation to proceed to completion over a standard overnight period. This simplicity belies the sophistication of the underlying chemistry, offering a powerful tool for the rapid generation of formamide libraries. For detailed operational parameters, including specific molar ratios and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction vessel by adding the copper trifluoromethyl complex (1 equiv) and purging with inert gas to ensure an oxygen-free environment.
- Introduce the amine substrate (primary or secondary) and the appropriate solvent (1,4-dioxane for secondary amines, MeCN with acid for primary amines).
- Heat the mixture to 100°C for 12 hours, then cool, dilute with dichloromethane, and purify via silica gel chromatography to isolate the high-purity formamide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-mediated formylation technology offers compelling advantages that directly impact the bottom line and supply chain resilience. By eliminating the reliance on toxic carbon monoxide gas, manufacturers can drastically reduce the costs associated with safety compliance, specialized equipment maintenance, and hazardous waste disposal. This shift not only lowers the operational expenditure but also mitigates the risk of production stoppages due to safety incidents or regulatory inspections, ensuring a more consistent supply of critical intermediates. Furthermore, the use of inexpensive and widely available amine starting materials means that raw material costs are kept low, while the high yields reported in the patent examples suggest minimal material loss during processing. These factors combine to create a highly cost-effective manufacturing process that enhances the overall competitiveness of the final API or fine chemical product in the global market.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO infrastructure and the use of a simple one-pot procedure significantly lowers capital and operational expenditures. By avoiding complex gas handling systems and reducing the number of purification steps required due to high selectivity, the overall cost of goods sold (COGS) is substantially decreased. Additionally, the ability to run the reaction in standard solvents like 1,4-dioxane or acetonitrile avoids the need for exotic or expensive reagents, further contributing to economic efficiency.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents like the copper complex is inherently more reliable than managing the logistics of toxic gases, which are subject to strict transportation regulations and supply disruptions. The robustness of the reaction across a wide range of substrates ensures that the process can be easily adapted to different product lines without extensive re-optimization, providing flexibility in production planning. This reliability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The method's green chemistry credentials, characterized by high atom economy and the absence of heavy metal catalysts that require rigorous removal, align perfectly with modern environmental standards. The simplified workup procedure reduces solvent consumption and waste generation, making it easier to scale from kilogram to tonne quantities without encountering the bottlenecks typical of batch carbonylation processes. This scalability ensures that the technology can grow with demand, supporting long-term strategic partnerships.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the process capabilities and limitations. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for integrating this method into existing production workflows effectively.
Q: What are the key advantages of this copper-mediated formylation over traditional CO carbonylation?
A: Unlike traditional methods requiring toxic carbon monoxide gas and high-pressure equipment, this patent-described method operates at atmospheric pressure using a stable solid copper complex, significantly enhancing operational safety and reducing infrastructure costs.
Q: Does this method support a wide range of amine substrates?
A: Yes, the technology demonstrates excellent substrate scope, successfully converting both primary amines (with acid additives) and secondary amines (under neutral conditions) into formamides with high selectivity and yields up to 95%.
Q: Is this process suitable for large-scale industrial production?
A: The patent explicitly highlights the method's potential for large-scale industrial production due to its one-pot nature, simple operation, use of cheap and easy-to-obtain amine starting materials, and environmentally friendly profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formamide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the copper-mediated formylation described in Patent CN116396180A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this high-efficiency route are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of formamide intermediate meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and discuss route feasibility assessments that demonstrate how we can optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthetic strategy.
