Advanced NBS Bromination Strategy for High-Purity 2-Substituted-4,6-Dibromophenol Manufacturing
Introduction to Novel Bromination Technology
The synthesis of halogenated phenolic intermediates remains a cornerstone in the development of advanced functional materials and pharmaceutical agents. Specifically, the preparation of 2-substituted-4,6-dibromophenol derivatives has gained significant traction due to their utility as precursors for phosphite antioxidants and flame retardants. Patent CN116283507A introduces a robust and environmentally conscious methodology for producing these valuable intermediates. Unlike traditional methods that often rely on hazardous elemental bromine or harsh Lewis acids, this innovation employs N-bromosuccinimide (NBS) as a controlled brominating source. The core of this technology lies in its ability to achieve exceptional regioselectivity, targeting the 4 and 6 positions of the phenolic ring with high precision. 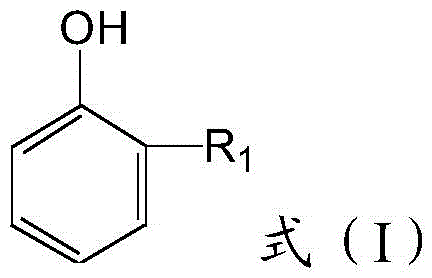 As illustrated in the general structure, the versatility of the R1 group allows for the synthesis of a broad library of derivatives, including alkyl, cycloalkyl, and alkoxy-substituted variants. This flexibility is critical for R&D teams seeking to tailor steric and electronic properties for specific downstream applications, such as Suzuki coupling reactions or coordination complex formation.
As illustrated in the general structure, the versatility of the R1 group allows for the synthesis of a broad library of derivatives, including alkyl, cycloalkyl, and alkoxy-substituted variants. This flexibility is critical for R&D teams seeking to tailor steric and electronic properties for specific downstream applications, such as Suzuki coupling reactions or coordination complex formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct bromination of phenols using molecular bromine (Br2) has been plagued by significant challenges regarding selectivity and safety. The high reactivity of elemental bromine often leads to uncontrolled polybromination, resulting in complex mixtures of mono-, di-, and tri-brominated species that are difficult to separate. Furthermore, the generation of stoichiometric amounts of hydrogen bromide (HBr) gas necessitates specialized scrubbing equipment and corrosion-resistant reactors, driving up capital expenditure. Traditional purification often requires column chromatography or multiple recrystallizations, which are impractical for ton-scale manufacturing. The presence of trace metal catalysts in some older protocols also introduces contamination risks, particularly unacceptable for pharmaceutical intermediate supply chains where heavy metal limits are stringent. These factors collectively contribute to high production costs, extended lead times, and substantial environmental burdens due to hazardous waste generation.
The Novel Approach
The methodology disclosed in CN116283507A represents a paradigm shift towards greener and more efficient synthesis. By utilizing NBS in a polar solvent system, the reaction kinetics are moderated, allowing for precise control over the degree of bromination. The process eliminates the need for handling volatile and corrosive elemental bromine, thereby enhancing operational safety. A key innovation is the workup procedure: instead of aqueous quenching which generates large volumes of saline wastewater, the process employs a solvent switch technique. The reaction mixture is concentrated and treated with a non-polar solvent, leveraging the poor solubility of the succinimide byproduct to effect its removal via simple filtration. This not only simplifies isolation but also facilitates solvent recovery, aligning with modern principles of atom economy and waste reduction. The result is a streamlined process capable of delivering high-purity products with minimal downstream processing.
Mechanistic Insights into NBS-Mediated Electrophilic Aromatic Substitution
The reaction mechanism proceeds via an electrophilic aromatic substitution pathway, driven by the activation of the phenolic ring. The hydroxyl group acts as a strong ortho/para director, activating the 4 and 6 positions for nucleophilic attack on the bromine species generated from NBS. In the polar solvent environment, NBS releases electrophilic bromine equivalents that selectively attack these activated sites. The stoichiometry is critical; as detailed in the patent, a molar ratio of phenol to NBS between 1:1.85 and 1:6.35 is required to drive the reaction to the dibromo stage. Insufficient NBS leads to the accumulation of mono-brominated intermediates, while excessive amounts do not significantly improve yield but may increase raw material costs. The choice of polar solvent, such as acetonitrile or DMF, stabilizes the transition state and ensures homogeneous reaction conditions, which is vital for consistent batch-to-batch reproducibility in a commercial setting.
Controlling impurity profiles is paramount for high-value intermediates. The primary side reactions involve incomplete bromination, yielding 2-substituted-4-bromophenol or 2-substituted-6-bromophenol. 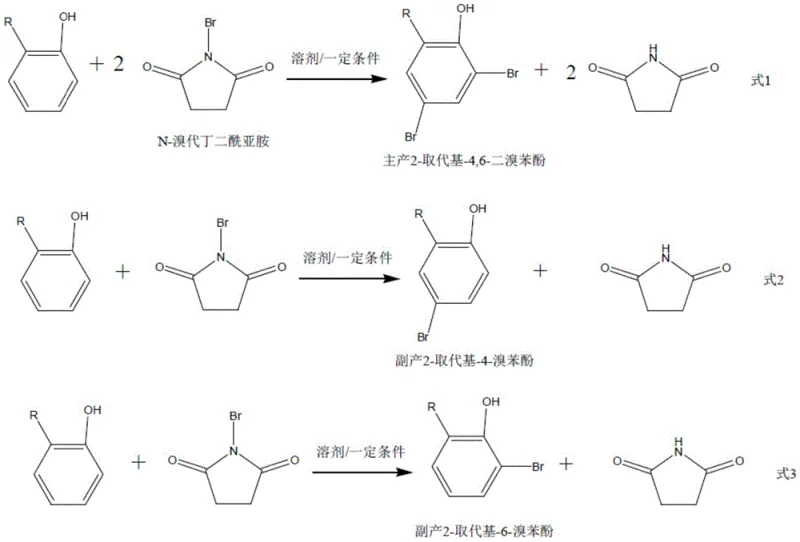 As depicted in the reaction scheme, the main reaction (Formula 1) consumes two equivalents of NBS to form the target dibromide, whereas side reactions (Formula 2 and 3) consume only one equivalent. The patent data indicates that optimizing the NBS ratio and reaction temperature (preferably -15°C to 60°C) suppresses these mono-bromo impurities, achieving selectivity exceeding 99%. Furthermore, the succinimide byproduct, formed in equimolar amounts to the bromine atoms transferred, is effectively sequestered during the non-polar solvent wash. This dual strategy of kinetic control during reaction and thermodynamic control during workup ensures a clean impurity profile, reducing the burden on analytical QC and ensuring the material meets rigorous specifications for sensitive applications like antioxidant synthesis.
As depicted in the reaction scheme, the main reaction (Formula 1) consumes two equivalents of NBS to form the target dibromide, whereas side reactions (Formula 2 and 3) consume only one equivalent. The patent data indicates that optimizing the NBS ratio and reaction temperature (preferably -15°C to 60°C) suppresses these mono-bromo impurities, achieving selectivity exceeding 99%. Furthermore, the succinimide byproduct, formed in equimolar amounts to the bromine atoms transferred, is effectively sequestered during the non-polar solvent wash. This dual strategy of kinetic control during reaction and thermodynamic control during workup ensures a clean impurity profile, reducing the burden on analytical QC and ensuring the material meets rigorous specifications for sensitive applications like antioxidant synthesis.
How to Synthesize 2-Substituted-4,6-Dibromophenol Efficiently
Implementing this synthesis route requires careful attention to solvent selection and phase separation dynamics. The process begins with the dissolution of the 2-substituted phenol substrate in a suitable polar solvent, followed by the controlled addition of NBS. Maintaining the temperature within the optimal range is essential to balance reaction rate and selectivity. Following the reaction period, typically 3 to 24 hours, the polar solvent is removed under reduced pressure. The resulting concentrate is then subjected to a crystallization step using a non-polar hydrocarbon solvent. This step is the linchpin of the purification strategy, as it precipitates the succinimide and other polar impurities while keeping the lipophilic dibromophenol product in solution.
- React 2-substituted phenol with N-bromosuccinimide (NBS) in a polar solvent like acetonitrile or THF at temperatures between -15°C and 60°C.
- Evaporate the reaction mixture under reduced pressure to concentrate, then add a non-polar solvent such as hexane to precipitate succinimide impurities.
- Filter the solution to remove solids, then distill off the non-polar solvent under vacuum to isolate the high-purity 2-substituted-4,6-dibromophenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NBS-based protocol offers tangible strategic benefits beyond mere technical feasibility. The shift away from elemental bromine mitigates significant regulatory and safety compliance costs associated with storing and transporting hazardous Class 8 corrosive materials. This simplifies logistics and reduces insurance premiums, contributing to overall cost reduction in fine chemical manufacturing. Moreover, the ability to recover and recycle both the polar reaction solvent and the non-polar crystallization solvent drastically lowers raw material consumption. The process generates minimal aqueous waste, avoiding the high costs of wastewater treatment and disposal. These efficiencies translate into a more stable pricing structure and reduced vulnerability to fluctuations in waste management fees.
- Cost Reduction in Manufacturing: The elimination of expensive metal catalysts and the use of readily available NBS significantly lower the bill of materials. The simplified workup procedure, which avoids complex extraction and chromatography, reduces labor hours and energy consumption per kilogram of product. By integrating solvent recovery loops, the facility can achieve substantial cost savings on organic solvents, which often constitute a major portion of variable production costs. The high conversion rates reported (>99%) mean that raw material wastage is minimized, further enhancing the economic viability of the process for large-volume orders.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like NBS, acetonitrile, and hexane ensures a robust supply chain less prone to disruption compared to specialized reagents. The mild reaction conditions (near ambient temperature and pressure) allow for production in standard glass-lined or stainless steel reactors without the need for exotic metallurgy, increasing the number of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis. This flexibility reduces lead time for high-purity intermediates by expanding the available manufacturing capacity and preventing bottlenecks associated with specialized equipment requirements.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated from gram to multi-kilogram scales in the patent examples. The absence of toxic HBr gas evolution simplifies reactor venting systems and aligns with increasingly stringent environmental, health, and safety (EHS) regulations. The "green" nature of the process, characterized by low three-waste emissions and solvent recyclability, supports corporate sustainability goals. This makes the supply of these intermediates more resilient against future regulatory tightening, ensuring long-term continuity of supply for downstream customers in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-substituted-4,6-dibromophenols. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing supply chains and R&D pipelines.
Q: What is the primary advantage of using NBS over elemental bromine for this synthesis?
A: N-bromosuccinimide (NBS) acts as a mild brominating agent that significantly reduces safety hazards associated with handling elemental bromine. It allows for better control over regioselectivity, minimizing over-bromination and reducing the formation of toxic HBr byproducts, which simplifies downstream waste treatment.
Q: How does the process remove succinimide byproducts effectively?
A: The process utilizes a solubility differential strategy. After the reaction in a polar solvent, the mixture is concentrated and treated with a non-polar solvent (e.g., hexane or petroleum ether). Succinimide is poorly soluble in these non-polar media and precipitates out, allowing it to be easily filtered off before the final product is isolated by solvent removal.
Q: Can this method be scaled for industrial production of flame retardant intermediates?
A: Yes, the patent explicitly highlights suitability for large-scale industrial production. The reaction operates at mild temperatures (-15°C to 60°C) and atmospheric or low pressure, utilizing commercially available solvents that can be recovered and recycled, making it economically viable for manufacturing flame retardant precursors and polymer additives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted-4,6-Dibromophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent literature are faithfully reproduced at an industrial scale. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the exacting standards required for pharmaceutical and specialty chemical applications. Our commitment to quality assurance guarantees that the impurity profiles, particularly regarding mono-brominated species and residual succinimide, are tightly controlled.
We invite you to collaborate with us to leverage this advanced bromination technology for your specific project needs. Whether you require custom synthesis of novel derivatives or reliable supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can enhance the efficiency and profitability of your supply chain.
