Advancing N-Formamide Production: A Breakthrough in Mild Copper-Catalyzed Oxidation for Commercial Scale
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for greener, more efficient pathways to critical building blocks. A significant advancement in this domain is detailed in patent CN114591233A, which discloses a novel method for preparing N-formamide derivatives through the catalytic oxidation of N-heterocyclic compounds. This technology represents a paradigm shift from traditional, energy-intensive protocols to a streamlined, one-pot catalytic system that operates under remarkably mild conditions. By leveraging a copper-based catalytic cycle coupled with hydrogen peroxide as a benign oxidant, this invention addresses long-standing challenges in selectivity and operational safety. For R&D directors and procurement strategists alike, understanding the nuances of this patent is crucial, as it offers a viable route for the commercial scale-up of complex pharmaceutical intermediates while adhering to stringent environmental standards. The ability to synthesize these versatile motifs at room temperature without requiring high-pressure equipment fundamentally alters the economic and logistical feasibility of large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-formamides has been plagued by significant technical and safety hurdles that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Traditional methodologies often rely on the use of toxic carbon monoxide gas under high pressure, posing severe safety risks and requiring specialized, expensive reactor infrastructure that many facilities lack. Alternatively, classical formylation reagents such as formic acid or formyl chloride frequently necessitate harsh acidic or basic conditions, elevated temperatures, and prolonged reaction times, which can lead to the degradation of sensitive functional groups on the substrate. Furthermore, non-selective oxidation systems often result in the formation of undesirable byproducts, including dimers, oligomers, or ring-opened species, thereby complicating downstream purification and drastically reducing overall atom economy. These inefficiencies not only inflate the cost of goods sold but also generate substantial volumes of hazardous waste, creating a bottleneck for supply chain sustainability and regulatory compliance in modern chemical plants.
The Novel Approach
In stark contrast to these archaic techniques, the methodology outlined in CN114591233A introduces a highly selective and operationally simple catalytic oxidative coupling strategy. This innovative approach utilizes methanol not merely as a solvent but as the carbonyl source, activated by a copper(II) nitrate catalyst and hydrogen peroxide oxidant. The reaction proceeds smoothly at ambient temperature (25°C) under an inert argon atmosphere, effectively mitigating the thermal stress on sensitive N-heterocyclic substrates. By employing a mixed solvent system of methanol and acetonitrile, the protocol ensures optimal solubility and reaction kinetics, allowing for the direct conversion of diverse amines and heterocycles into their corresponding N-formamides. This eliminates the need for pre-functionalization steps or dangerous gaseous reagents, thereby simplifying the workflow and enhancing the safety profile of the manufacturing process. The robustness of this system is evidenced by its compatibility with a wide array of substrates, ranging from tetrahydroquinolines to substituted anilines, making it a versatile tool for reducing lead time for high-purity pharmaceutical intermediates.
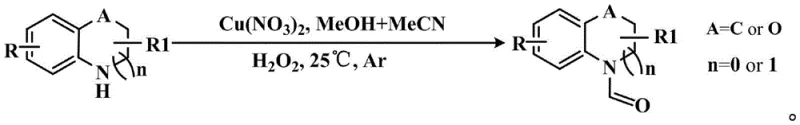
Mechanistic Insights into Cu(NO3)2-Catalyzed Oxidative Coupling
To fully appreciate the technical merit of this invention, one must delve into the mechanistic intricacies of the copper-catalyzed oxidative cycle. The process initiates with the coordination of the N-heterocyclic substrate to the copper(II) center, which acts as a Lewis acid to activate the nitrogen lone pair. Simultaneously, hydrogen peroxide interacts with the copper species to generate reactive oxygen intermediates, likely involving high-valent copper-oxo species or radical pathways that facilitate the selective oxidation of methanol. Unlike indiscriminate oxidants that might attack the electron-rich aromatic rings of the heterocycle, this catalytic system is finely tuned to target the alpha-C-H bonds of the methanol solvent. The resulting formaldehyde or hemiaminal intermediate then undergoes a condensation reaction with the amine substrate, followed by a final dehydrogenation step mediated by the copper catalyst to yield the stable N-formamide product. This precise control over the oxidation state prevents the over-oxidation of the substrate to imines or nitriles, ensuring high chemoselectivity even in the presence of other oxidizable functional groups like halides or alkyl substituents.
From an impurity control perspective, the mechanism offers distinct advantages for maintaining product purity, a critical metric for any reliable pharmaceutical intermediate supplier. The mild reaction conditions suppress the formation of thermal degradation products and polymerization side reactions that are common in high-temperature processes. Moreover, the use of hydrogen peroxide as the terminal oxidant ensures that the only stoichiometric byproduct is water, which is easily removed during the concentration phase. The absence of heavy metal contaminants from exotic catalysts simplifies the purification workflow; standard silica gel column chromatography using petroleum ether and ethyl acetate mixtures is sufficient to isolate the target compound with high purity. This streamlined purification process minimizes product loss and reduces the consumption of organic solvents, aligning with green chemistry principles. For process chemists, understanding this mechanism allows for the rational optimization of reaction parameters, such as the molar ratio of oxidant to substrate (typically 2 mmol H2O2 to 0.5 mmol substrate), to maximize yield while minimizing residual impurities.
How to Synthesize N-Formamide Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to ensure reproducibility and safety. The patent describes a straightforward one-pot procedure where the N-heterocyclic compound is first dissolved in a mixture of methanol and acetonitrile within a pressure tube equipped with magnetic stirring. The reaction environment is rigorously controlled by purging with high-purity argon gas (99.999%) to exclude oxygen and moisture, which could interfere with the catalytic cycle. Following the addition of the copper(II) nitrate catalyst (12 mol%), the mixture is stirred briefly before the careful introduction of hydrogen peroxide. The reaction vessel is then sealed and maintained at room temperature for a period ranging from 13 to 26 hours, allowing the oxidative coupling to reach completion. Upon conclusion, the solvent is removed via rotary evaporation, and the crude residue is subjected to flash chromatography. Detailed standardized synthesis steps see the guide below.
- Mix the N-heterocyclic substrate with methanol and acetonitrile in a pressure tube under an argon atmosphere.
- Add copper(II) nitrate catalyst and stir, followed by the slow addition of hydrogen peroxide as the oxidant.
- Stir the reaction mixture at room temperature for 13 to 26 hours, then concentrate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed methodology offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The elimination of high-pressure reactors and toxic gas handling systems translates directly into reduced capital expenditure (CAPEX) and lower operational risk profiles. By operating at ambient temperature, the process drastically cuts energy consumption associated with heating and cooling cycles, contributing to significant utility cost savings over the lifecycle of the product. Furthermore, the reliance on commodity chemicals such as methanol, acetonitrile, and hydrogen peroxide ensures a stable and resilient supply chain, insulating manufacturers from the volatility often seen with specialized reagents. This robustness facilitates cost reduction in pharmaceutical intermediates manufacturing by streamlining inventory management and reducing the need for complex storage infrastructure. The simplicity of the workup procedure also accelerates batch turnover times, enhancing overall plant throughput and responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of earth-abundant copper salts rather than precious metals like palladium or rhodium, which are subject to extreme price volatility and supply constraints. The catalyst loading is relatively low (12 mol%), and the potential for catalyst recovery or recycling further enhances the cost-efficiency profile. Additionally, the avoidance of expensive protecting group strategies or multi-step sequences reduces the overall material intensity of the synthesis. By consolidating the formylation into a single oxidative step, manufacturers can eliminate intermediate isolation and drying stages, thereby saving on labor, solvent, and time costs. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, and this method bolsters reliability by utilizing reagents that are globally available in bulk quantities. Methanol and hydrogen peroxide are produced on a massive industrial scale, ensuring that raw material shortages are unlikely to disrupt production schedules. The mild reaction conditions also reduce wear and tear on processing equipment, extending asset life and minimizing unplanned maintenance downtime. For logistics planners, the non-hazardous nature of the reagents compared to alternatives like phosgene or carbon monoxide simplifies transportation and storage compliance, reducing regulatory burdens and insurance premiums. This stability allows for more accurate forecasting and long-term supply agreements, fostering stronger partnerships between chemical suppliers and their clients.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the tonne scale often introduces new challenges, but the inherent safety of this oxidative system facilitates a smoother scale-up trajectory. The exothermicity of the reaction is manageable at room temperature, reducing the risk of thermal runaway incidents in large reactors. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding waste disposal and emissions. The generation of water as the primary byproduct minimizes the load on wastewater treatment facilities, and the high atom economy ensures that a greater proportion of raw materials end up in the final product rather than as waste. This eco-friendly profile not only aids in meeting corporate sustainability goals but also streamlines the permitting process for new manufacturing lines, accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of what stakeholders can expect during technology transfer. Understanding these details is essential for assessing the fit of this methodology within existing production portfolios and for evaluating its potential to replace legacy synthetic routes. The answers reflect the consensus on the operational parameters and substrate scope defined by the inventors.
Q: What are the primary advantages of this copper-catalyzed method over traditional formylation?
A: This method operates at room temperature (25°C) using inexpensive hydrogen peroxide and copper nitrate, avoiding the high pressures and toxic carbon monoxide sources often required in conventional formylation processes.
Q: Which substrates are compatible with this oxidative coupling system?
A: The system demonstrates broad substrate scope, successfully converting tetrahydroquinolines, indoles, benzomorpholines, and various aniline derivatives into their corresponding N-formamides with moderate to excellent yields.
Q: How does this process impact environmental compliance in manufacturing?
A: By utilizing hydrogen peroxide, which generates water as the only byproduct, and eliminating the need for heavy metal scavengers or harsh chlorinating agents, the process significantly reduces hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic oxidation route described in CN114591233A for the production of high-value N-formamide intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative academic and patent discoveries into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory gram-scale to industrial tonne-scale is seamless and efficient. We are committed to delivering stringent purity specifications through our state-of-the-art rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch. Whether you require custom synthesis of complex tetrahydroquinoline derivatives or bulk supply of standard aniline-based formamides, our facility is equipped to meet your specific volume and quality requirements with precision.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains using this advanced chemistry. By leveraging our process development capabilities, we can conduct a Customized Cost-Saving Analysis tailored to your specific target molecules, identifying opportunities to reduce waste and improve yields further. We encourage you to contact our technical procurement team to request specific COA data for similar compounds and to discuss route feasibility assessments for your upcoming projects. Together, we can accelerate the development of next-generation therapeutics by ensuring a reliable, cost-effective, and sustainable supply of critical chemical building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
