Scalable Synthesis of 1,2,4-Triazolopyridine Derivatives via Mild Oxidative Cyclization for Commercial Production
Scalable Synthesis of 1,2,4-Triazolopyridine Derivatives via Mild Oxidative Cyclization for Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs) and functional materials. Patent CN111138429A introduces a transformative methodology for the synthesis of 1,2,4-triazolopyridine derivatives, utilizing 2-pyridylhydrazone derivatives and sodium hypochlorite as the primary oxidant. This innovation addresses long-standing challenges in heterocyclic chemistry by replacing hazardous reagents with a commodity chemical, thereby streamlining the production workflow. The process operates under remarkably mild conditions, typically ranging from 0°C to 100°C, and utilizes ethanol as a preferred green solvent, which significantly lowers the barrier for industrial adoption. For R&D directors and procurement managers alike, this patent represents a pivotal shift towards sustainable manufacturing, offering a pathway to high-purity intermediates with reduced operational complexity and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazolopyridine fused ring system has relied on methodologies that impose severe constraints on process safety, environmental compliance, and cost efficiency. Traditional routes often employ reagents such as Lawesson's reagent under microwave irradiation in acetic acid, or utilize highly corrosive and toxic phosphorylating agents like phosphorus oxychloride (POCl3) and phosphorus pentachloride. These conventional approaches generate substantial quantities of hazardous waste, including phosphorus and sulfur-containing byproducts, which necessitate expensive and energy-intensive waste treatment protocols. Furthermore, reactions mediated by transition metal carbonyls, such as Mo(CO)6, introduce the risk of heavy metal contamination, requiring rigorous and costly purification steps to meet stringent pharmaceutical purity specifications. The reliance on harsh conditions, such as high temperatures or strong acids, also limits the substrate scope, often leading to the decomposition of sensitive functional groups and resulting in lower overall yields and inconsistent batch-to-batch quality.
The Novel Approach
In stark contrast, the novel oxidative cyclization strategy disclosed in the patent leverages the readily available and inexpensive oxidant sodium hypochlorite to drive the ring-closing reaction efficiently. This method eliminates the need for toxic phosphorus or sulfur reagents and avoids the use of expensive transition metal catalysts, fundamentally simplifying the reaction matrix. By conducting the reaction in ethanol at ambient or slightly elevated temperatures, the process ensures excellent functional group tolerance, allowing for the successful synthesis of complex derivatives bearing sensitive moieties such as hydroxyl groups and extended conjugated systems. The operational simplicity is further enhanced by the fact that the byproducts are primarily inorganic salts and water, which facilitates straightforward work-up procedures. This approach not only aligns with green chemistry principles but also drastically reduces the total cost of ownership for manufacturing these valuable heterocyclic intermediates, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Sodium Hypochlorite-Mediated Oxidative Cyclization
The core of this technological breakthrough lies in the efficient oxidative cyclization mechanism where the 2-pyridylhydrazone derivative undergoes intramolecular bond formation facilitated by the electrophilic chlorine species generated from sodium hypochlorite. The reaction initiates with the activation of the hydrazone nitrogen, followed by a nucleophilic attack on the adjacent pyridine ring or an intermediate chlorinated species, ultimately leading to the formation of the triazole ring fused to the pyridine system. This mechanism is characterized by high atom economy, as the oxygen from the hypochlorite and the hydrogen from the substrate combine to form water, while the sodium and chloride ions remain as benign salt byproducts. The mildness of the oxidant prevents over-oxidation of sensitive substituents, a common pitfall in harsher oxidative protocols, thereby preserving the integrity of complex molecular architectures required for advanced applications such as fluorescent probes and bio-active agents.
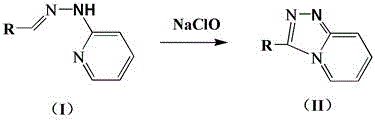
Impurity control is inherently superior in this system due to the selectivity of sodium hypochlorite under the optimized conditions. Unlike radical-based oxidations that can lead to non-specific degradation of the organic framework, this ionic pathway proceeds with high specificity towards the desired cyclization. The patent data indicates that by controlling the molar ratio of sodium hypochlorite to substrate at an optimal 1:4 ratio, side reactions are minimized, and the formation of chlorinated byproducts on the aromatic rings is suppressed. This results in a cleaner crude reaction profile, which significantly reduces the burden on downstream purification processes such as column chromatography or recrystallization. For quality assurance teams, this translates to a more consistent impurity profile and a higher probability of meeting strict regulatory limits for genotoxic impurities and residual solvents in the final API intermediate.
How to Synthesize 1,2,4-Triazolopyridine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for laboratory and pilot-scale production, emphasizing the ease of handling and the robustness of the reaction conditions. The procedure typically involves dissolving the 2-pyridylhydrazone precursor in ethanol, followed by the controlled addition of the sodium hypochlorite solution. The reaction mixture is then stirred at room temperature or slightly heated for a duration of 1 to 24 hours, depending on the specific electronic nature of the substituents. Upon completion, the solvent is removed via distillation, and the residue is purified using standard chromatographic techniques. This streamlined workflow eliminates the need for inert atmosphere gloveboxes or specialized high-pressure reactors, making it accessible for a wide range of manufacturing facilities. The detailed standardized synthesis steps see the guide below.
- Dissolve the 2-pyridylhydrazone derivative substrate in ethanol solvent within a reaction vessel equipped with stirring.
- Add sodium hypochlorite solution slowly to the reaction mixture while maintaining the temperature between 0°C and 100°C, preferably at room temperature.
- Stir the reaction for 1 to 24 hours, monitor completion, remove solvent, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this sodium hypochlorite-mediated synthesis route offers profound advantages that directly impact the bottom line and operational resilience. The shift away from proprietary or hazardous reagents to commodity chemicals like bleach and ethanol decouples the production process from volatile supply chains associated with specialized catalysts or toxic reagents. This ensures a stable and continuous supply of raw materials, mitigating the risk of production stoppages due to reagent shortages. Furthermore, the simplified waste stream, consisting mainly of saline water and ethanol, dramatically lowers the costs associated with hazardous waste disposal and environmental compliance, contributing to substantial cost savings in the overall manufacturing lifecycle.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and toxic phosphorylating agents removes significant cost drivers from the bill of materials. Sodium hypochlorite is a globally available commodity chemical with a stable and low price point, which contrasts sharply with the fluctuating costs of organometallic reagents. Additionally, the reduced need for complex purification steps to remove heavy metals or phosphorus residues lowers the consumption of silica gel and solvents during the isolation phase. This cumulative effect leads to a significantly reduced cost of goods sold (COGS), enabling more competitive pricing for the final triazolopyridine intermediates in the global market.
- Enhanced Supply Chain Reliability: Utilizing ethanol as the primary solvent and sodium hypochlorite as the oxidant ensures that the critical inputs for this synthesis are readily available from multiple suppliers worldwide, reducing dependency on single-source vendors. The mild reaction conditions also mean that the process can be executed in standard glass-lined or stainless steel reactors without requiring exotic materials of construction that are resistant to strong acids or corrosive gases. This flexibility allows for easier technology transfer between different manufacturing sites and ensures that production schedules can be maintained even during regional supply disruptions, thereby enhancing the overall reliability of the supply chain for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The inherent safety of the process, characterized by the absence of pyrophoric reagents and the generation of non-toxic byproducts, makes it exceptionally well-suited for commercial scale-up of complex heterocyclic compounds. The simplified effluent profile facilitates easier wastewater treatment, helping manufacturers meet increasingly stringent environmental regulations without investing in costly abatement technologies. This alignment with green chemistry principles not only future-proofs the manufacturing process against regulatory changes but also enhances the corporate sustainability profile, which is becoming a critical factor in vendor selection for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, drawing directly from the experimental data and comparative studies presented in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for potential partners and technical stakeholders evaluating this technology for their own production pipelines. Understanding these nuances is essential for making informed decisions about process adoption and integration into existing manufacturing portfolios.
Q: What are the advantages of using sodium hypochlorite over traditional oxidants like Lawesson's reagent?
A: Sodium hypochlorite offers superior atom economy and generates benign byproducts (NaCl and water), whereas Lawesson's reagent produces toxic phosphorus and sulfur waste, requiring complex disposal protocols and increasing environmental compliance costs.
Q: Is this synthesis method suitable for large-scale manufacturing of fluorescent probes?
A: Yes, the method operates under mild conditions (0-100°C) in common solvents like ethanol, making it highly scalable for producing complex fluorophores such as coumarin and rhodamine-based triazolopyridine derivatives without requiring specialized high-pressure equipment.
Q: What is the typical molar ratio of oxidant to substrate for optimal yield?
A: According to patent data, the optimal molar ratio of the hydrazone substrate to sodium hypochlorite is 1:4, ensuring complete conversion while minimizing excess reagent usage and downstream purification burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazolopyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this oxidative cyclization technology to redefine the production standards for triazole-based heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, maintaining stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. We are committed to delivering high-purity 1,2,4-triazolopyridine derivatives that meet the exacting demands of the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this innovative synthetic route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our reference standards and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes quality, sustainability, and cost-efficiency, ensuring your projects remain competitive and compliant in a rapidly evolving market landscape.
