Advanced Synthesis Of Imidazole Aromatic Alcohol Derivatives For High Potency Antifungal Drug Manufacturing
The pharmaceutical industry is constantly seeking novel chemical entities that can overcome the growing challenge of antifungal resistance, and patent CN1307163C presents a significant advancement in this domain by disclosing a robust method for producing imidazole aromatic alcohol derivatives. This specific intellectual property outlines a sophisticated synthetic pathway that transforms readily available starting materials into highly potent antifungal agents, specifically targeting deep mycoses which pose severe risks to immunocompromised patients. The technical breakthrough lies in the strategic combination of the Schiemann reaction for fluorination and optimized phase transfer catalysis for alkylation, resulting in compounds that demonstrate superior in vitro activity compared to legacy drugs like econazole and miconazole. By leveraging this patented methodology, manufacturers can access a new class of chemical structures that offer enhanced therapeutic indices and broader spectra of activity against pathogenic strains such as Candida albicans. The detailed procedural controls described within the patent ensure that the final nitrate salts possess the requisite purity and stability needed for rigorous clinical development and eventual commercialization in the global healthcare market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for azole-based antifungal intermediates often suffer from significant inefficiencies related to regioselectivity and harsh reaction conditions that can degrade sensitive functional groups. Many conventional processes rely on direct fluorination techniques that lack precision, leading to complex mixtures of isomers that require energy-intensive and costly purification steps to isolate the desired active pharmaceutical ingredient. Furthermore, older alkylation strategies frequently necessitate the use of strong, anhydrous bases and high temperatures, which can promote side reactions such as elimination or polymerization, thereby drastically reducing the overall yield of the target molecule. The reliance on stoichiometric amounts of expensive reagents and the generation of substantial hazardous waste streams also present major economic and environmental hurdles for large-scale production facilities aiming for sustainability. These legacy methods often struggle to consistently achieve the high purity standards required by modern regulatory bodies, leading to batch failures and supply chain disruptions that impact the availability of critical medications.
The Novel Approach
In stark contrast, the methodology described in patent CN1307163C utilizes a highly controlled sequence beginning with the Schiemann reaction to install fluorine atoms with exceptional precision on the aromatic ring system. This approach allows for the construction of the 2,4-difluoro phenyl core under mild thermal decomposition conditions, avoiding the chaotic reactivity associated with direct halogen exchange methods. Subsequent steps employ phase transfer catalysis to drive both N-alkylation and O-alkylation reactions efficiently in biphasic media, which simplifies the workup procedure and minimizes the formation of byproducts. The strategic ordering of reactions ensures that the reactive chloro-acetyl group is reduced and alkylated in a manner that preserves the integrity of the imidazole ring, which is crucial for the biological activity of the final drug substance. 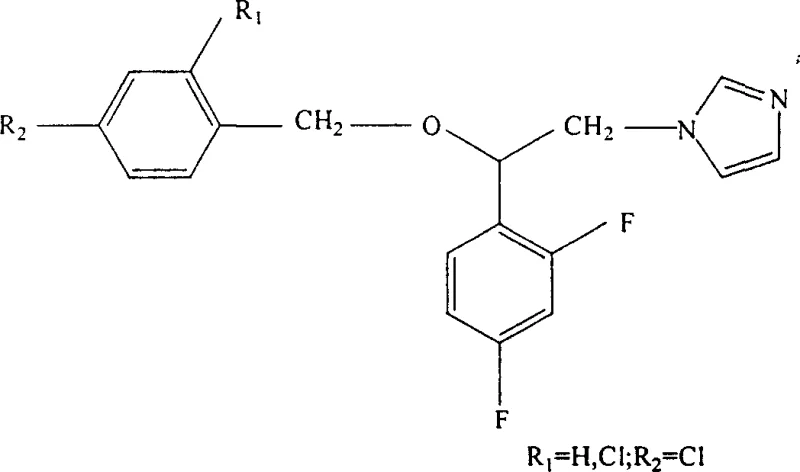 This novel pathway not only streamlines the manufacturing process but also delivers a final product with demonstrated higher antifungal potency, providing a clear competitive advantage for developers of next-generation antimicrobial therapies.
This novel pathway not only streamlines the manufacturing process but also delivers a final product with demonstrated higher antifungal potency, providing a clear competitive advantage for developers of next-generation antimicrobial therapies.
Mechanistic Insights into Schiemann Fluorination and Phase Transfer Catalysis
The core of this synthesis relies on the mechanistic elegance of the Schiemann reaction, where m-phenylenediamine is diazotized in the presence of tetrafluoroboric acid to form a stable diazonium tetrafluoroborate salt intermediate. Upon thermal decomposition, this salt releases nitrogen gas and boron trifluoride, facilitating the nucleophilic attack of the fluoride ion onto the aromatic ring to yield m-difluorobenzene with high fidelity. This mechanism is superior to other fluorination methods because the tetrafluoroborate anion acts as a safe and effective source of nucleophilic fluoride, preventing the formation of poly-fluorinated impurities that are common in electrochemical or direct fluorination processes. The subsequent Friedel-Crafts acylation utilizes anhydrous aluminum trichloride to activate the chloroacetyl chloride, ensuring that the acyl group attaches exclusively at the position ortho to one of the fluorine atoms due to electronic directing effects. This level of mechanistic control is essential for maintaining the structural integrity required for the molecule to effectively bind to the lanosterol 14α-demethylase enzyme in fungal cells.
Impurity control is further enhanced during the alkylation stages through the precise application of phase transfer catalysts such as tetrabutylammonium bromide or polyethylene glycols. These catalysts function by solubilizing the hydroxide anions in the organic phase, allowing them to deprotonate the alcohol or imidazole nitrogen without requiring extreme conditions that could degrade the substrate. The use of quaternary ammonium salts creates a microenvironment where the nucleophile is highly reactive yet shielded from competing side reactions, significantly reducing the formation of dialkylated byproducts or ether cleavage products. Additionally, the patent specifies careful pH control during the final salt formation with nitric acid, which ensures that only the basic imidazole nitrogen is protonated, leaving other functional groups intact and preventing the co-precipitation of acidic impurities. This rigorous attention to mechanistic detail throughout the synthesis ensures a clean impurity profile that facilitates easier downstream processing and meets the stringent specifications demanded by global pharmacopoeias.
How to Synthesize Imidazole Aromatic Alcohol Derivatives Efficiently
The synthesis of these high-value antifungal intermediates requires strict adherence to the temperature and molar ratio parameters defined in the patent to ensure optimal conversion and selectivity at every stage. Operators must begin by carefully controlling the diazotization temperature between 0°C and -15°C to prevent premature decomposition of the diazonium salt, which is critical for safety and yield. Following the thermal decomposition to generate the difluoroarene, the subsequent acylation and reduction steps must be monitored closely to avoid over-reduction or hydrolysis of the chloro-ketone intermediate. The final alkylation steps benefit significantly from the use of specific phase transfer catalysts like TBAB or PEG400, which should be added in precise catalytic amounts to drive the reaction to completion without complicating the isolation of the product. Detailed standardized synthesis steps see the guide below.
- Perform Schiemann reaction on m-phenylenediamine with tetrafluoroboric acid and sodium nitrite at low temperature to form m-difluorobenzene.
- Conduct Friedel-Crafts acylation with chloroacetyl chloride followed by reduction to obtain the chloro-alcohol intermediate.
- Execute sequential N-alkylation with imidazole and O-alkylation with benzyl chloride using phase transfer catalysts, finishing with nitric acid salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits primarily driven by the simplification of the manufacturing workflow and the use of commodity-grade starting materials. The reliance on m-phenylenediamine and common halogenating agents means that the supply chain is not vulnerable to the bottlenecks often associated with exotic or highly regulated specialty reagents. Furthermore, the high yields reported in the patent examples suggest that less raw material is wasted per kilogram of finished product, directly translating to improved material efficiency and lower variable costs per unit. The robustness of the phase transfer catalysis steps also implies that the process is more forgiving of minor variations in scale-up conditions, reducing the risk of batch failures during technology transfer from pilot plant to commercial production. These factors combine to create a more resilient and cost-effective supply chain for critical antifungal intermediates.
- Cost Reduction in Manufacturing: The elimination of complex purification sequences and the high efficiency of the phase transfer catalyzed steps significantly reduce the consumption of solvents and energy utilities during production. By avoiding the need for cryogenic conditions in all but the initial diazotization step, the process lowers the capital expenditure required for specialized refrigeration equipment and reduces ongoing operational energy costs. The high selectivity of the reactions minimizes the generation of hazardous waste streams, thereby lowering the costs associated with waste treatment and environmental compliance disposal fees. Overall, the streamlined nature of the synthesis allows for a leaner manufacturing footprint with reduced labor hours per batch, driving down the total cost of goods sold.
- Enhanced Supply Chain Reliability: Since the synthesis begins with widely available industrial chemicals like m-phenylenediamine and chloroacetyl chloride, the risk of raw material shortages is significantly mitigated compared to routes relying on scarce fluorinated building blocks. The multi-step sequence is designed with robust intermediates that can be isolated and stored if necessary, providing flexibility in production scheduling and inventory management to buffer against demand fluctuations. The use of standard reactor materials and common catalysts ensures that the process can be easily replicated across multiple manufacturing sites, diversifying the supply base and reducing single-point failure risks. This reliability is crucial for maintaining continuous supply to pharmaceutical customers who require just-in-time delivery of key intermediates for their own formulation lines.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential as the exothermic reactions are well-managed through controlled addition rates and the use of standard cooling media, making it suitable for large-scale batch reactors. The reduction in solvent usage and the ability to recycle aqueous phases from the phase transfer steps contribute to a greener manufacturing profile that aligns with increasingly strict global environmental regulations. The final crystallization of the nitrate salt provides a high-purity solid form that is stable and easy to handle, package, and transport, minimizing the logistical complexities associated with liquid intermediates. These attributes make the technology highly attractive for contract development and manufacturing organizations looking to expand their portfolio of sustainable and scalable pharmaceutical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these imidazole derivatives based on the specific disclosures in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into their existing development pipelines or sourcing strategies. The answers provided reflect the specific experimental conditions and results validated within the intellectual property to ensure accuracy and relevance.
Q: What represents the key innovation in patent CN1307163C regarding antifungal intermediates?
A: The patent introduces a streamlined synthesis route for imidazole aromatic alcohol derivatives that achieves higher yields and superior antifungal activity compared to established drugs like econazole and miconazole, specifically through optimized phase transfer catalysis conditions.
Q: How does the Schiemann reaction contribute to the quality of the final pharmaceutical intermediate?
A: The Schiemann reaction allows for the precise introduction of fluorine atoms onto the aromatic ring under controlled low-temperature conditions, ensuring high regioselectivity and minimizing halogenated impurities that could complicate downstream purification.
Q: Why is phase transfer catalysis critical in the alkylation steps of this synthesis?
A: Phase transfer catalysts such as tetrabutylammonium bromide facilitate the reaction between organic substrates and inorganic bases in biphasic systems, significantly enhancing reaction rates and selectivity while allowing for milder reaction temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Aromatic Alcohol Derivatives Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of the Schiemann reaction and phase transfer catalysis steps described in patent CN1307163C with the utmost precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of imidazole aromatic alcohol derivatives meets the highest international standards for pharmaceutical intermediates. Our commitment to quality assurance ensures that the impurity profiles of our products are fully characterized and controlled, providing you with the confidence needed for regulatory filings.
We invite you to contact our technical procurement team to discuss how we can support your specific antifungal drug development programs with tailored solutions. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall project costs while maintaining superior quality. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions about your supply chain strategy. Partner with us to leverage our technical expertise and capacity to bring your next-generation antifungal therapies to market faster and more efficiently.
