Advanced Chiral Bidentate Ligands for Efficient Indole Alkylation and Commercial Scale-Up
Advanced Chiral Bidentate Ligands for Efficient Indole Alkylation and Commercial Scale-Up
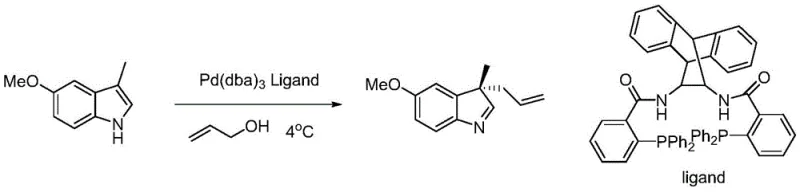
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing quaternary carbon stereocenters, particularly within the privileged indole scaffold. Patent CN110845547A, published in early 2020, introduces a groundbreaking class of chiral bidentate ligands based on a ferrocene backbone that addresses long-standing challenges in asymmetric catalysis. This technology enables the efficient synthesis of C-3 substituted indole compounds, which are critical intermediates for a wide array of bioactive molecules. By leveraging a novel coordination geometry between the ferrocene-derived ligand and palladium metal centers, this invention achieves high enantioselectivity under remarkably mild conditions. For R&D directors and process chemists, this represents a significant leap forward in reliable pharma intermediates supplier capabilities, offering a pathway to complex molecular architectures that was previously hindered by harsh reaction requirements and inconsistent stereocontrol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric synthesis of indole derivatives possessing quaternary chiral centers has been fraught with significant technical hurdles. Prominent methodologies, such as the Mukaiyama Aldol reaction utilized by Overman et al., while effective in introducing chiral aldehydes, suffer from poor atom economy and severe substrate limitations. Furthermore, these legacy processes often mandate cryogenic conditions to achieve acceptable levels of stereochemical induction, imposing heavy energy costs and engineering constraints on large-scale manufacturing. Similarly, the palladium-catalyzed alkylation strategies developed by Trost et al., which employ trialkylboranes, exhibit a critical vulnerability: their enantioselectivity is highly temperature-dependent. As illustrated in prior art representations, raising the reaction temperature even slightly causes a precipitous drop in ee values, forcing manufacturers to operate at inefficiently low temperatures that slow down throughput and complicate heat transfer in large reactors.
The Novel Approach
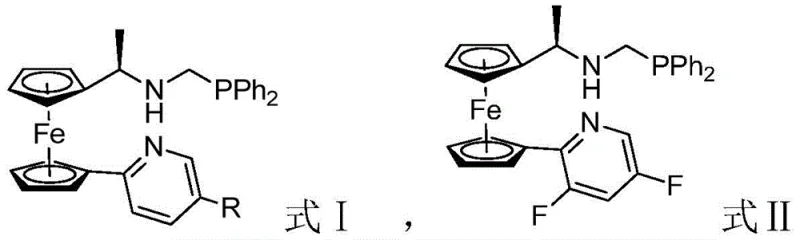
In stark contrast to these conventional limitations, the technology disclosed in CN110845547A utilizes a structurally unique ferrocene-based bidentate ligand system, defined generally by Formula I and Formula II. These ligands feature a specific arrangement of a phosphine donor and a nitrogen-containing heterocycle attached to the chiral ferrocene core. This architecture creates a rigid and well-defined chiral pocket around the palladium active site. The result is a catalyst that maintains high activity and exceptional enantioselectivity even at ambient temperatures (25°C). Unlike previous generations of catalysts, this system does not require stringent exclusion of water or oxygen during the catalytic cycle, dramatically lowering the barrier to entry for cost reduction in pharma intermediates manufacturing. The structural versatility allows for tuning via substituents such as methyl or tert-butyl groups, enabling precise optimization for specific substrate classes.
Mechanistic Insights into Pd-Catalyzed Asymmetric Alkylation
The efficacy of this catalytic system stems from the synergistic interaction between the soft phosphine donor and the harder nitrogen donor within the ligand framework. Upon coordination with palladium salts such as PdCl2 or Pd(OAc)2, the ligand forms a stable cationic or neutral complex that activates the allylic or alkylating species. The bulky ferrocene backbone acts as a chiral wall, effectively blocking one face of the approaching nucleophile (the indole derivative). This steric differentiation is crucial for inducing the formation of the quaternary carbon center with high fidelity. The electronic properties of the pyridine ring, modulated by substituents like fluorine or tert-butyl groups, further fine-tune the Lewis acidity of the metal center, optimizing the turnover frequency. This mechanistic robustness ensures that side reactions, such as racemization or oligomerization, are minimized, leading to cleaner reaction profiles and simplified downstream purification processes.
From an impurity control perspective, the ability to run the reaction at 25°C rather than -78°C is transformative. Low-temperature reactions often trap kinetic impurities or lead to incomplete conversions that require extensive recycling. By operating at thermodynamic equilibrium favoring the product at room temperature, the process ensures consistent quality. The catalyst loading is exceptionally low, ranging from 0.01 mol% to 0.05 mol%, which not only reduces the residual metal burden in the final API but also minimizes the formation of metal-associated degradation products. This level of control is essential for meeting the rigorous purity specifications demanded by global regulatory bodies for high-purity pharmaceutical intermediates.
How to Synthesize Chiral Ferrocene Ligands Efficiently
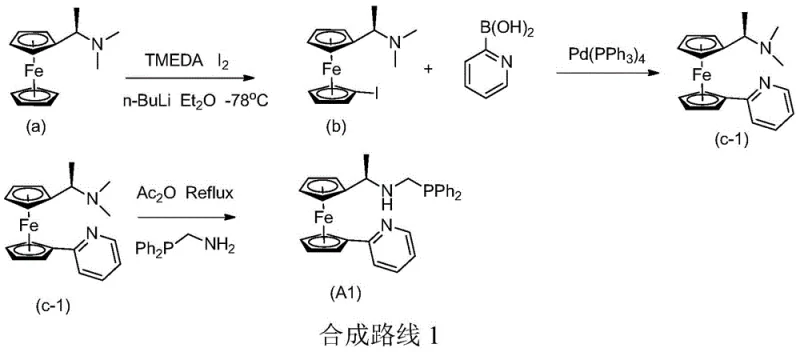
The preparation of these high-performance ligands follows a logical and scalable three-step sequence that begins with commercially available (R)-N,N-dimethylferrocenylamine. The process involves a directed ortho-lithiation followed by iodination to install a handle for cross-coupling. Subsequent Suzuki-Miyaura coupling with various pyridine boronic acids installs the necessary nitrogen donor arm. The final step involves a nucleophilic substitution to introduce the diphenylphosphine moiety. This modular approach allows for the rapid generation of a library of ligands to suit different catalytic needs. The detailed standardized synthesis steps for the most preferred embodiment, Ligand A1, are outlined below, demonstrating the feasibility of translating this chemistry from the bench to the pilot plant.
- Perform directed lithiation of (R)-N,N-dimethylferrocenylamine at -78°C followed by iodination to generate the key iodo-intermediate.
- Execute a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction with substituted pyridine boronic acids to install the heterocyclic moiety.
- Complete the ligand synthesis via nucleophilic substitution using diphenylphosphinomethylamine under reflux conditions to yield the final bidentate structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers profound strategic benefits beyond mere chemical elegance. The primary driver for value creation is the drastic simplification of the process infrastructure. By eliminating the need for cryogenic cooling systems and rigorous inert atmosphere gloveboxes for the catalytic step, capital expenditure (CAPEX) for new production lines is significantly reduced. Furthermore, the operational expenditure (OPEX) is lowered through reduced energy consumption and shorter cycle times, as reactions proceed rapidly at room temperature without the lag time associated with cooling and warming large batches. This translates directly into a more competitive cost structure for the final intermediates.
- Cost Reduction in Manufacturing: The ultra-low catalyst loading of 0.01-0.05 mol% represents a massive saving in precious metal costs. Palladium is a high-value commodity, and reducing its usage by orders of magnitude compared to traditional 1-5 mol% loadings directly improves the gross margin. Additionally, the simplified workup procedures, which do not require complex metal scavenging steps due to the low loading, further reduce the cost of goods sold. The elimination of expensive cryogens like liquid nitrogen or specialized cooling brines for the main reaction step adds another layer of financial efficiency to the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of the catalyst against moisture and oxygen variations makes the supply chain more resilient. Traditional air-sensitive catalysts often suffer from batch-to-batch variability due to minor leaks or handling errors, leading to production delays and wasted materials. This new system's tolerance ensures consistent output regardless of minor environmental fluctuations, guaranteeing on-time delivery to downstream customers. The starting materials, such as ferrocene derivatives and substituted boronic acids, are readily available from multiple global suppliers, mitigating the risk of single-source bottlenecks.
- Scalability and Environmental Compliance: Scaling asymmetric reactions is notoriously difficult due to heat transfer and mixing issues at low temperatures. This room-temperature protocol removes those engineering barriers, facilitating a seamless transition from kilogram to multi-ton scale. Moreover, the high atom economy and reduced solvent usage associated with the higher concentrations possible at ambient temperatures contribute to a greener manufacturing footprint. This aligns with modern ESG (Environmental, Social, and Governance) goals, making the supply chain more attractive to sustainability-conscious partners and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferrocene-based catalytic system. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this catalyst over traditional Trost or Overman systems?
A: Unlike traditional systems that require cryogenic temperatures to maintain enantioselectivity, this novel ferrocene-based catalyst operates efficiently at room temperature (25°C) without the need for rigorous water or oxygen exclusion, significantly simplifying process engineering.
Q: What is the typical catalyst loading required for industrial application?
A: The patent specifies an ultra-low catalyst loading ranging from 0.01 mol% to 0.05 mol%, which drastically reduces the cost of goods sold (COGS) associated with precious metal usage compared to standard protocols requiring 1-5 mol%.
Q: Can this methodology be applied to substrates other than 5-methoxy-3-methylindole?
A: Yes, the catalyst system demonstrates broad substrate scope, successfully catalyzing the asymmetric alkylation of various indole derivatives including 5-methoxy-3-ethylindole, maintaining high yields and enantiomeric excess across different steric environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bidentate Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in CN110845547A for the synthesis of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic chemistry, ensuring that the stringent purity specifications and rigorous QC labs standards required for chiral ligands are met with precision. We understand that the successful commercialization of such advanced intermediates relies not just on the chemistry, but on the reliability and consistency of the supply partner.
We invite you to collaborate with us to leverage this cutting-edge technology for your pipeline projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how switching to this room-temperature catalytic protocol can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog ligands or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your development timeline with superior chemical solutions.
