Advanced Chiral Diboronate Esters for Efficient 1,2-Diamine Manufacturing
Introduction to Next-Generation Chiral Synthesis
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the need for more efficient and scalable routes to chiral building blocks. Patent CN111440205B introduces a groundbreaking class of biboric acid diol esters that serve as powerful inducing agents for the reductive coupling of imines. This technology addresses critical bottlenecks in the production of chiral 1,2-diamines, which are indispensable scaffolds in the development of active pharmaceutical ingredients (APIs), agrochemical intermediates, and specialized chiral catalysts. Unlike previous iterations of boron-mediated couplings that were restricted to specific cyclic structures like isoquinolines, this novel methodology expands the substrate scope to include a vast array of aromatic imines derived from readily available aldehydes and ammonia. The strategic implementation of these chiral diboronates allows for the direct construction of carbon-nitrogen bonds with exceptional stereocontrol, achieving enantiomeric excess values as high as 99% ee through simple recrystallization. This represents a paradigm shift for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering high-purity compounds without the prohibitive costs associated with traditional chiral pool synthesis.
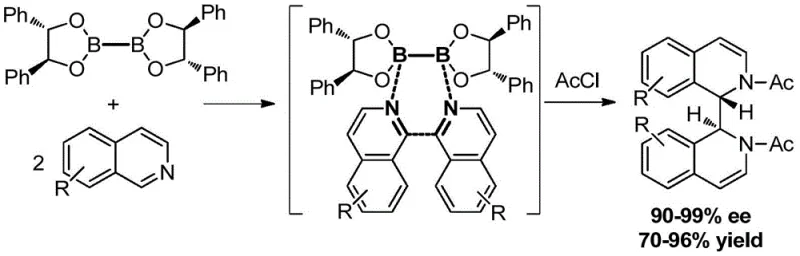
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 1,2-diamines has been plagued by inherent inefficiencies that drive up costs and complicate supply chains. The most common traditional approach involves the resolution of racemic mixtures, a process that is fundamentally flawed due to its maximum theoretical yield of only 50%. This limitation necessitates the disposal or recycling of half the produced material, creating significant waste and requiring extensive screening of resolution reagents for each new substrate. Furthermore, methods relying on chiral auxiliary groups often demand stoichiometric amounts of expensive reagents that are consumed during the reaction, leading to poor atom economy and high raw material costs. Another prevalent route involves nucleophilic substitution using chiral diols, which frequently requires hazardous reagents like sodium azide and involves multi-step sequences with protecting group manipulations. These conventional pathways are not only labor-intensive but also pose safety risks and environmental challenges that are increasingly unacceptable in modern green chemistry frameworks. As illustrated in the comparative analysis of synthetic strategies, the industry has long needed a method that bypasses these structural and economic constraints to enable true commercial viability.
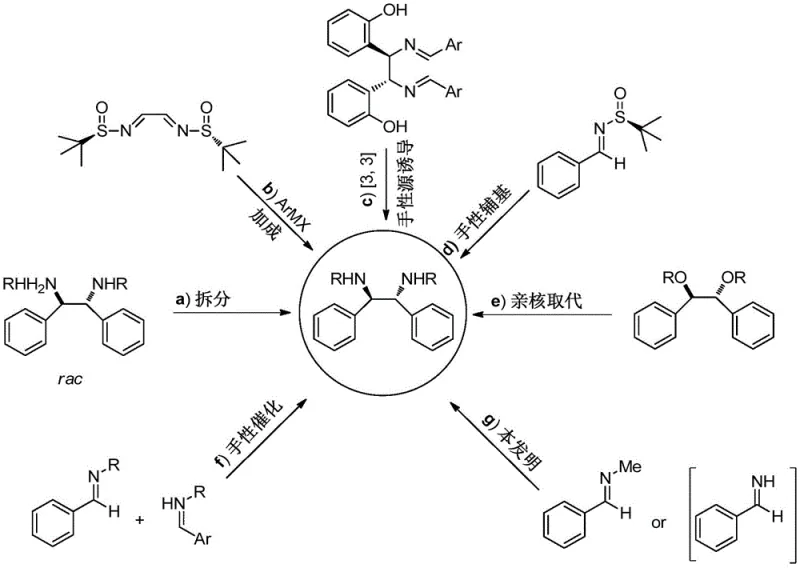
The Novel Approach
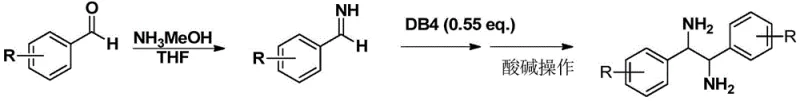
The innovative strategy disclosed in the patent utilizes a specifically designed chiral diboronic acid diol ester to induce the reductive coupling of imines directly. This approach leverages the unique reactivity of the boron-boron bond to facilitate the formation of the 1,2-diamine skeleton in a single catalytic or stoichiometric step with high fidelity. By starting from simple aromatic aldehydes and ammonia, the process accesses a wide variety of substrates that were previously difficult or impossible to functionalize with such high stereoselectivity. The reaction conditions are remarkably mild, typically proceeding at room temperature (20-30°C) in common solvents like tetrahydrofuran, which eliminates the need for cryogenic conditions or high-pressure equipment. Crucially, the workup procedure is drastically simplified; the product is isolated via straightforward acid-base extraction, completely avoiding the need for time-consuming and solvent-intensive column chromatography. This streamlined workflow not only accelerates the timeline from bench to kilogram scale but also ensures that the final product meets stringent purity specifications required for downstream applications in drug discovery and materials science.
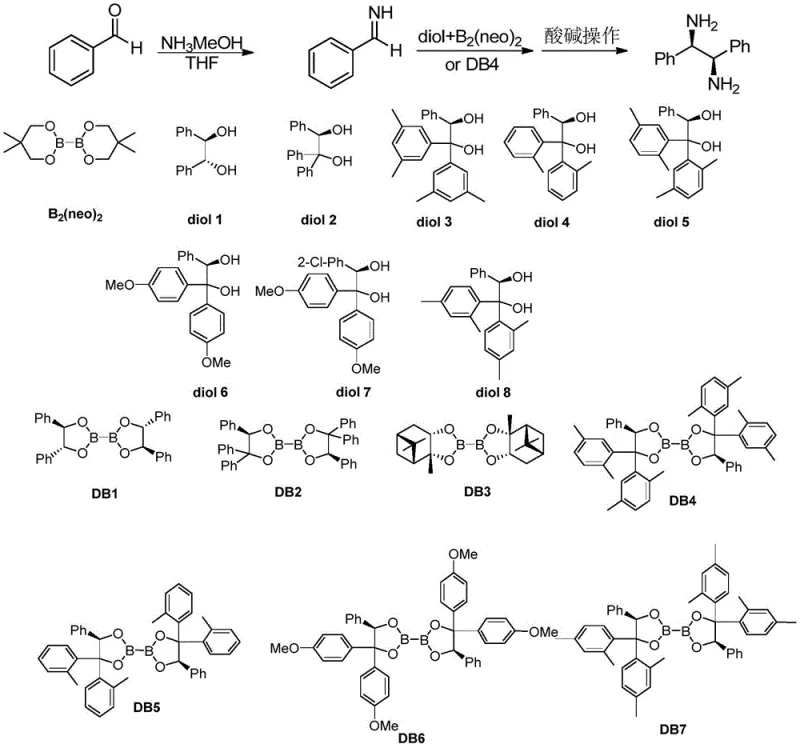
Mechanistic Insights into Diboronate-Mediated Reductive Coupling
The core of this technology lies in the precise interaction between the chiral diboronate ester and the imine substrate. The reaction mechanism involves the activation of the imine double bond by the Lewis acidic boron centers, followed by a stereoselective addition that establishes two new chiral centers simultaneously. The chirality of the diol ligand attached to the boron atoms creates a rigid chiral environment that directs the approach of the reactants, ensuring that one enantiomer is formed preferentially over the other. This high level of stereocontrol is evidenced by the consistent achievement of 95-99% ee across a diverse range of substrates, including those with electron-donating groups like methoxy and electron-withdrawing groups like halogens. The robustness of the catalyst system allows it to tolerate various functional groups without the need for protection, which is a significant advantage over organometallic methods that might be sensitive to moisture or reactive functionalities. Furthermore, the mechanistic pathway minimizes the formation of side products, resulting in a clean reaction profile that simplifies purification. The ability to tune the steric and electronic properties of the diol ligand, as seen in the variation of DB4 through DB7, provides chemists with a toolkit to optimize selectivity for specific challenging substrates, ensuring that the process remains effective even as molecular complexity increases.
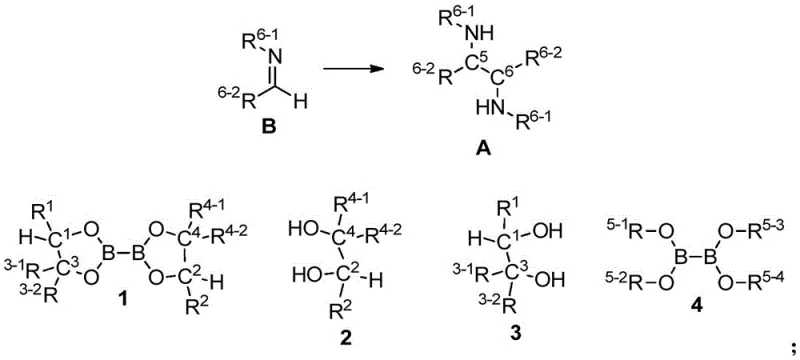
Impurity control is another critical aspect where this methodology excels. In traditional syntheses, trace metals from catalysts or byproducts from protecting group removal can persist in the final API, requiring costly scavenging steps. In contrast, the boron-mediated coupling generates benign byproducts that are easily removed during the aqueous workup. The hydrolysis of the boron species yields boric acid derivatives which are water-soluble and separate cleanly from the organic amine product upon pH adjustment. This intrinsic cleanliness of the reaction reduces the burden on quality control laboratories and ensures that the impurity profile of the final diamine is well within regulatory limits. Additionally, the stability of the intermediate imine and the diboronate reagent allows for flexible processing times, reducing the risk of batch failures due to strict timing constraints. The combination of high selectivity and clean reaction profiles makes this technology particularly attractive for the commercial scale-up of complex pharmaceutical intermediates where consistency and purity are paramount.
How to Synthesize Chiral 1,2-Diamines Efficiently
The practical implementation of this synthesis route is designed for ease of operation and scalability. The process begins with the preparation of the chiral diboronate ester, which can be synthesized by reacting the corresponding chiral diol with tetrahydroxydiboron in the presence of molecular sieves to remove water. Once the coupling reagent is prepared, the imine substrate is generated in situ by mixing the aromatic aldehyde with an ammonia source, such as an ammonia-methanol solution. The addition of the diboronate ester to this mixture initiates the reductive coupling, which proceeds smoothly at ambient temperatures. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during laboratory and pilot plant operations.
- Prepare the chiral diboronate ester by reacting a specific chiral diol with tetrahydroxydiboron in THF at 70°C with molecular sieves.
- Generate the imine substrate in situ by reacting an aromatic aldehyde with an ammonia methanol solution in THF at room temperature.
- Add the diboronate coupling reagent to the imine mixture, stir for 12-24 hours, and isolate the product via simple acid-base extraction without chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this diboronate-mediated synthesis offers substantial strategic benefits beyond mere technical performance. The primary driver for cost reduction in pharmaceutical intermediate manufacturing is the efficiency of raw material utilization and the simplicity of the isolation process. By eliminating the 50% yield ceiling inherent in resolution processes and avoiding the use of stoichiometric chiral auxiliaries, this method significantly lowers the cost of goods sold. The ability to recover and reuse the chiral diol ligand with up to 95% efficiency further amplifies these savings, turning what would be a consumable expense into a recyclable asset. Moreover, the removal of column chromatography from the purification workflow drastically reduces solvent consumption and processing time, leading to lower operational expenditures and a smaller environmental footprint. These factors combine to create a supply chain that is not only more cost-effective but also more resilient to fluctuations in raw material prices.
- Cost Reduction in Manufacturing: The economic model of this process is superior because it maximizes atom economy and minimizes waste. Traditional methods often require expensive chiral starting materials or catalysts that are lost during the reaction. In contrast, this novel approach uses readily available aldehydes and ammonia as starting materials, which are commodity chemicals with stable pricing. The high recovery rate of the chiral diol means that the initial investment in the chiral inducer is amortized over many batches, driving down the unit cost per kilogram of the final diamine. Additionally, the avoidance of protective group chemistry reduces the number of synthetic steps, which directly correlates to lower labor costs and reduced equipment usage time. This streamlined synthesis allows manufacturers to offer competitive pricing for high-value chiral intermediates without compromising on quality or margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on scarce or specialized reagents. This technology mitigates such risks by utilizing a robust set of starting materials that are globally sourced and widely available. The simplicity of the reaction conditions, which do not require exotic catalysts or extreme temperatures, ensures that production can be maintained consistently across different manufacturing sites. The high yield and selectivity reduce the likelihood of batch failures, which is a common cause of supply disruptions in fine chemical production. Furthermore, the scalability of the process from gram to multi-ton levels has been demonstrated, providing confidence that supply can be ramped up quickly to meet market demand. This reliability is crucial for pharmaceutical companies that require guaranteed delivery schedules to maintain their own production timelines for final drug products.
- Scalability and Environmental Compliance: As regulatory pressure mounts for greener manufacturing processes, this method stands out for its environmental compatibility. The reduction in solvent usage, particularly the elimination of large volumes of chromatography solvents, aligns with sustainability goals and reduces the cost of waste disposal. The aqueous workup generates minimal hazardous waste compared to heavy metal-catalyzed alternatives, simplifying compliance with environmental regulations. The process is inherently safe, operating at near-ambient temperatures and pressures, which reduces the energy load on the facility and minimizes safety risks associated with high-energy reactions. These attributes make the technology ideal for large-scale commercial production, where environmental, health, and safety (EHS) considerations are just as critical as economic ones. Manufacturers adopting this route can position themselves as leaders in sustainable chemistry while enjoying the operational efficiencies of a simplified process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diboronate technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows. The clarity provided here aims to remove ambiguity regarding the capabilities and limitations of the synthesis route.
Q: What is the primary advantage of this diboronate method over traditional resolution?
A: Unlike traditional resolution which is limited to a maximum theoretical yield of 50%, this asymmetric reductive coupling method achieves high yields (up to 82% demonstrated) with excellent stereoselectivity (up to 99% ee), effectively doubling the potential output from the same amount of starting material.
Q: Can the chiral diol ligand be recovered after the reaction?
A: Yes, a key economic driver of this process is the recyclability of the chiral diol. Through simple acid-base operations and pulping with n-hexane, the diol can be recovered from the reaction mixture with a recovery rate reaching up to 95%, significantly reducing the cost of goods for large-scale production.
Q: Is column chromatography required for purification?
A: No, one of the major operational benefits is the elimination of column chromatography. The product can be separated from the reaction system solely through acid-base extraction and washing operations, which simplifies the workflow and makes the process highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,2-Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the diboronate-mediated reductive coupling described in CN111440205B. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality intermediates. Our rigorous QC labs and stringent purity specifications guarantee that every batch of chiral 1,2-diamine meets the exacting standards required for pharmaceutical applications. We are committed to leveraging cutting-edge chemistry to solve complex supply chain challenges, offering solutions that balance performance with cost-efficiency. Our team is ready to assist you in navigating the transition from laboratory discovery to commercial manufacturing with confidence.
We invite you to contact our technical procurement team to discuss how this innovative technology can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this novel synthesis route. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your target molecules. Let us partner with you to accelerate your development timelines and secure a reliable supply of high-purity chiral intermediates for your future success.
