Industrial Scale-Up of Crosslinkable 6-Methylthioethyl Purine Deoxynucleoside for Advanced DNA Materials
The landscape of functional nucleic acid materials is undergoing a significant transformation, driven by the demand for specialized building blocks capable of site-directed mutagenesis and DNA encryption. A pivotal advancement in this domain is detailed in patent CN113461743A, which discloses a highly efficient preparation method for 6-methylthioethyl purine-2'-deoxynucleoside. This compound serves as a critical precursor for crosslinkable nucleosides, enabling the creation of stable DNA structures through interchain covalent bonding. Unlike traditional approaches that rely on expensive and poorly soluble natural nucleosides, this innovation leverages a synthetic ribose derivative to achieve superior process economics. For R&D directors and procurement specialists in the oligonucleotide sector, this technology represents a paradigm shift towards more accessible and scalable manufacturing of complex nucleoside analogues. The ability to produce high-purity intermediates without resorting to labor-intensive chromatographic separation addresses a major bottleneck in the supply chain of advanced genetic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of crosslinkable purine deoxynucleoside analogues has been hindered by significant economic and operational barriers. Prior art methods, such as those described in J. Am. Chem. Soc. 1999, typically utilize deoxyguanosine protected at the 3' and 5' positions with tert-butyldimethylsilane groups as the starting material. This specific protected nucleoside is not commercially available off-the-shelf, necessitating an additional synthesis step from deoxyguanosine itself. Deoxyguanosine is inherently expensive and suffers from poor solubility in common organic solvents, which severely limits reaction concentrations and complicates processing. Furthermore, the multi-step nature of these conventional routes often results in lower overall yields and generates substantial chemical waste. The reliance on chromatographic purification for intermediates further escalates production costs and extends lead times, making the commercial scale-up of complex nucleoside analogues economically challenging for many manufacturers.
The Novel Approach
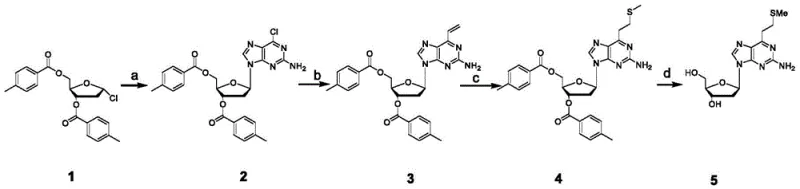
The methodology outlined in the patent introduces a streamlined four-step synthetic route that circumvents the drawbacks of natural nucleoside usage. By employing 1-chloro-2-deoxy-3,5-di-O-p-methylbenzoyl-D-ribose as the starting scaffold, the process utilizes a raw material that is not only significantly lower in cost but also exhibits excellent solubility and handling properties. The strategy involves an initial N-glycosylation to attach the purine base, followed by a palladium-catalyzed Stille coupling to introduce a vinyl group, and subsequently a thioetherification to install the methylthioethyl functionality. Crucially, the final deprotection step yields the target product in a form that can be purified via simple crystallization rather than column chromatography. This shift from chromatographic to crystalline purification is a game-changer for industrial viability, drastically reducing solvent consumption and processing time while ensuring high product purity suitable for sensitive biological applications.
Mechanistic Insights into Stille Coupling and Thioetherification
The core of this synthetic innovation lies in the precise manipulation of the purine ring substituents through transition metal catalysis and nucleophilic addition. The second step of the sequence employs a Stille coupling reaction, where the chlorine atom at the 6-position of the purine ring is displaced by a vinyl group using vinyl tributyltin and a tetrakis(triphenylphosphine)palladium catalyst. This transformation is critical as it installs the carbon backbone necessary for the subsequent functionalization. The reaction conditions, utilizing lithium chloride in ethyl acetate under reflux, are optimized to maximize conversion while minimizing the formation of organotin byproducts, which are notoriously difficult to remove. Following the vinyl installation, the third step involves a conjugate addition or radical-mediated thioetherification using sodium methyl mercaptide. This step effectively converts the reactive vinyl double bond into the stable methylthioethyl side chain, which acts as a latent crosslinking group. The mechanistic elegance of this sequence ensures that the sensitive glycosidic bond remains intact throughout the harsh conditions required for base modification.
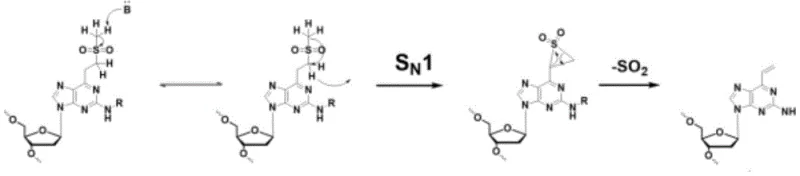
Beyond the synthesis of the monomer, the true value of this molecule is realized during its application in DNA assembly, where a unique elimination mechanism comes into play. Once incorporated into an oligonucleotide chain, the methylthioethyl group can be oxidized to a sulfone. Under basic conditions and elevated temperatures, this sulfone undergoes a specific elimination reaction. The mechanism involves the formation of a carbanion followed by an intramolecular rearrangement to generate a transient cyclic sulfone intermediate. This unstable species rapidly decomposes to release sulfur dioxide gas and generate a terminal olefin (vinyl group) directly on the DNA base. This in situ generation of the reactive alkene allows for subsequent Michael addition with amine groups on complementary strands, facilitating robust interchain crosslinking without the need for external crosslinking agents.
How to Synthesize 6-Methylthioethyl Purine-2'-Deoxynucleoside Efficiently
The practical execution of this synthesis requires careful control of reaction parameters to ensure high yields and purity without the need for chromatographic intervention. The process begins with the activation of the purine base using a strong non-nucleophilic base like sodium bis(trimethylsilyl)amide in tetrahydrofuran, followed by the addition of the chloro-ribose derivative. Subsequent steps involve rigorous removal of tin residues after the coupling reaction, typically achieved through aqueous workups with fluoride salts or saline solutions. The final deprotection is carried out in a sealed vessel with ammonia in methanol at elevated temperatures to ensure complete removal of the benzoyl protecting groups. The resulting crude product is then subjected to ultrasonic treatment and crystallization from isopropyl ether to obtain the final off-white crystalline solid. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to replicate this high-efficiency route, please refer to the standardized guide below.
- Perform N-glycosylation using sodium bis(trimethylsilyl)amide and 2-amino-6-chloro-purine in THF to form the protected chloro-intermediate.
- Execute a Stille coupling reaction with vinyl tributyltin and palladium catalyst to introduce the vinyl group at the 6-position of the purine ring.
- React the vinyl intermediate with sodium methyl mercaptide in acetonitrile to generate the methylthioethyl side chain.
- Remove the p-methylbenzoyl protecting groups using ammonia in methanol under heated conditions to yield the final crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the need for silica gel column chromatography, manufacturers can significantly reduce the consumption of high-purity solvents and stationary phases, which are major cost drivers in fine chemical production. This simplification also translates to shorter batch cycles, allowing for faster turnover and improved responsiveness to market demand. Additionally, the use of a readily available ribose derivative as the starting material mitigates the supply risk associated with sourcing expensive, protected natural nucleosides that may have limited vendor availability. This stability in raw material sourcing is crucial for maintaining continuous production schedules in the competitive oligonucleotide therapeutics market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the replacement of costly starting materials and the removal of chromatographic purification steps. The starting material, 1-chloro-2-deoxy-3,5-di-O-p-methylbenzoyl-D-ribose, is substantially cheaper than the protected deoxyguanosine used in legacy methods. Furthermore, the ability to purify intermediates and the final product through crystallization rather than chromatography leads to substantial cost savings in terms of both consumables and labor. The reduced usage of toxic organotin reagents compared to prior art also lowers waste disposal costs and enhances the overall environmental profile of the manufacturing process.
- Enhanced Supply Chain Reliability: Relying on synthetic sugar derivatives rather than modified natural products insulates the supply chain from the volatility of biological sourcing. The reagents required for the Stille coupling and thioetherification steps are commodity chemicals with robust global supply networks. This ensures that production is not held hostage by the availability of niche biochemicals. Moreover, the simplified process flow reduces the number of potential failure points in the manufacturing line, thereby increasing the reliability of delivery timelines for critical research and development projects dependent on these specialized nucleoside building blocks.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, moving away from batch-limited chromatographic separations to continuous or large-batch crystallization operations. This scalability is essential for meeting the growing demand for DNA-based materials in therapeutics and data storage. From an environmental perspective, the reduction in solvent waste and the minimization of heavy metal catalyst loading align with increasingly stringent green chemistry regulations. The safer and more reliable synthesis process reduces the operational risks associated with handling hazardous materials, ensuring compliance with modern safety standards in chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 6-methylthioethyl purine-2'-deoxynucleoside. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on the process capabilities and product performance. Understanding these details is essential for evaluating the suitability of this intermediate for your specific oligonucleotide synthesis requirements.
Q: What are the primary cost advantages of this synthesis route compared to traditional deoxyguanosine methods?
A: The novel route utilizes 1-chloro-2-deoxy-3,5-di-O-p-methylbenzoyl-D-ribose as a starting material, which is significantly cheaper and more soluble than the tert-butyldimethylsilane-protected deoxyguanosine required in conventional methods. Furthermore, the process eliminates the need for column chromatography, relying instead on efficient crystallization for purification.
Q: How does the methylthioethyl group facilitate DNA crosslinking applications?
A: During DNA synthesis, the methylthioethyl group can be oxidized to a methylsulfonyl ethyl unit. Subsequent high-temperature ammonolysis triggers an elimination reaction that converts this unit into a reactive terminal olefin (vinyl group), which then undergoes Michael addition with amino groups on complementary DNA bases to form interchain crosslinks.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It involves only four reaction steps with robust conditions, avoids complex purification techniques like chromatography, and utilizes reagents like vinyl tributyltin in reduced quantities compared to prior art, making it safer and more environmentally friendly for mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methylthioethyl Purine-2'-Deoxynucleoside Supplier
The technological breakthroughs presented in patent CN113461743A highlight the immense potential of streamlined nucleoside synthesis for the next generation of genetic medicines and materials. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory routes are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that the consistency of nucleoside intermediates is paramount for the success of downstream oligonucleotide synthesis, and our manufacturing protocols are designed to deliver exactly that level of reliability.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this critical crosslinkable nucleoside for your research and commercial applications.
