Advanced Suzuki Coupling Strategy for Scalable Aryl Pyridine Derivative Production
The pharmaceutical industry continuously seeks robust synthetic pathways for heterocyclic compounds that serve as critical scaffolds in drug discovery, particularly for oncology applications. Patent CN103351333A introduces a significant advancement in the preparation of aryl pyridine derivative compounds, utilizing a highly efficient palladium-catalyzed Suzuki coupling reaction. This technology addresses long-standing challenges in constructing substituted pyridine frameworks by employing a specific combination of inorganic bases and phosphorus ligands to achieve superior selectivity. The disclosed method enables the direct coupling of halogenated pyridines with substituted phenyl boronic acids, resulting in disubstituted pyridine structures with remarkable purity profiles. By optimizing reaction parameters such as solvent composition and temperature control, this process minimizes the formation of unwanted by-products, thereby streamlining the downstream purification burden for process chemists. The strategic selection of mild reaction conditions not only enhances safety but also aligns with modern green chemistry principles required for sustainable pharmaceutical intermediate manufacturing.
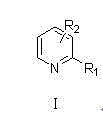
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating aryl-substituted pyridines often suffer from severe limitations regarding regioselectivity and functional group tolerance. Conventional methods frequently rely on harsh reaction conditions, such as strong organometallic reagents or extreme temperatures, which can lead to the decomposition of sensitive functional groups present on the pyridine ring. Furthermore, many existing protocols require expensive and difficult-to-remove transition metal catalysts that complicate the purification process and increase the overall cost of goods. The lack of systematic study on various poly-halogenated pyridines in prior art has resulted in inconsistent yields and unpredictable impurity profiles, creating significant bottlenecks for reliable pharmaceutical intermediate suppliers aiming to deliver consistent quality. These inefficiencies often necessitate multiple synthetic steps or extensive chromatographic purification, which are economically unsustainable for large-scale production campaigns.
The Novel Approach
The innovative methodology described in the patent overcomes these hurdles by implementing a optimized Suzuki-Miyaura cross-coupling strategy that operates under remarkably mild alkaline conditions. By utilizing inexpensive inorganic bases like sodium carbonate in a mixed solvent system of acetonitrile and methanol, the process achieves high conversion rates without compromising the integrity of the substrate. This approach significantly improves reaction selectivity, effectively suppressing side reactions that typically plague pyridine functionalization. The one-step synthesis capability allows for the direct assembly of complex molecular architectures from readily available starting materials, drastically simplifying the operational workflow. This streamlined process is particularly advantageous for cost reduction in API manufacturing, as it reduces both material consumption and processing time while ensuring high face shaping and yield consistency across diverse substrate scopes.
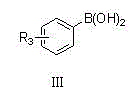
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The core of this synthetic breakthrough lies in the precise orchestration of the palladium catalytic cycle, which facilitates the carbon-carbon bond formation between the electron-deficient pyridine ring and the nucleophilic boronic acid. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the halogenated pyridine, a step that is critically influenced by the electronic nature of the pyridine nitrogen and the choice of ligand. The use of triphenylphosphine as a ligand stabilizes the active palladium species and accelerates the subsequent transmetallation step with the organoboron compound activated by the inorganic base. This careful balance of steric and electronic properties ensures that the catalytic turnover number remains high throughout the reaction duration, preventing catalyst deactivation which is a common issue in heterocyclic coupling. Understanding these mechanistic nuances is essential for R&D teams aiming to replicate these results for high-purity OLED material or pharmaceutical precursors.
Impurity control is inherently built into this mechanism through the suppression of homocoupling and protodeboronation side reactions. The specific solvent ratio of acetonitrile to methanol plays a pivotal role in solubilizing both the inorganic base and the organic substrates, creating a homogeneous reaction environment that promotes efficient collision frequency. Additionally, the mild basicity of sodium carbonate prevents the hydrolysis of sensitive ester or nitrile groups that might be present on the aryl boronic acid, preserving the structural diversity of the final product. The reaction proceeds under inert gas protection to prevent oxidation of the phosphine ligand and the palladium center, ensuring long-term catalyst stability. This rigorous control over the reaction microenvironment results in a clean crude product profile, significantly reducing the load on purification units and enhancing the overall throughput for commercial scale-up of complex polymer additives or fine chemicals.
How to Synthesize Aryl Pyridine Derivatives Efficiently
To implement this synthesis effectively, process engineers must adhere to strict stoichiometric ratios and environmental controls as outlined in the patent embodiments. The detailed standardized synthesis steps involve precise weighing of the halogenated pyridine and phenyl boronic acid, followed by the sequential addition of the base, catalyst, and ligand in the designated solvent mixture. Maintaining the reaction temperature within the specified range of 25°C to 80°C is crucial for balancing reaction kinetics with thermal stability. For comprehensive operational details, please refer to the structured guide below which breaks down the exact procedural requirements for laboratory and pilot plant execution.
- Prepare the reaction mixture by combining halogenated pyridine and substituted phenyl boronic acid in a mixed solvent system of acetonitrile and methanol.
- Add inorganic base such as sodium carbonate, along with a palladium catalyst like Pd(OAc)2 and a phosphorus ligand such as triphenylphosphine.
- Stir the reaction under inert gas protection at temperatures between 25°C and 80°C for 12 to 24 hours, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial economic benefits by leveraging commodity chemicals and minimizing the reliance on exotic reagents. The ability to use sodium carbonate as the base instead of more expensive or hazardous alternatives translates directly into lower raw material costs and simplified waste disposal protocols. Moreover, the high selectivity of the reaction reduces the need for costly chromatographic separation media, allowing for simpler crystallization or extraction workups that are far more scalable. This efficiency drives significant cost reduction in electronic chemical manufacturing and pharma sectors by lowering the overall cost of goods sold while maintaining premium quality standards. Supply chain managers will appreciate the robustness of the process, which tolerates minor variations in input quality without catastrophic failure, ensuring consistent output.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal scavengers and the use of low-cost inorganic bases drastically reduce the operational expenditure associated with catalyst removal and neutralization steps. By avoiding harsh conditions, the lifespan of reactor vessels and ancillary equipment is extended, further contributing to long-term capital savings. The high yield reported in the embodiments implies less raw material waste per kilogram of product, optimizing the atom economy of the entire production line. These factors combine to create a highly competitive cost structure that allows suppliers to offer aggressive pricing without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted phenyl boronic acids and halogenated pyridines, are widely available from global chemical vendors, mitigating the risk of single-source dependency. The mild reaction conditions reduce the energy intensity of the process, making it less susceptible to fluctuations in utility costs or availability. This resilience ensures that production schedules can be maintained even during periods of resource constraint, providing customers with dependable delivery timelines. The simplicity of the one-step process also shortens the manufacturing cycle time, enabling faster response to market demand spikes.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste due to the absence of toxic heavy metal residues and the use of recyclable solvent systems. The mild temperature profile reduces the cooling and heating load on industrial reactors, aligning with corporate sustainability goals and regulatory emissions standards. Scaling this reaction from gram to tonnage levels is straightforward because the heat transfer and mixing requirements are not extreme, facilitating a smooth technology transfer from R&D to production. This environmental compatibility simplifies permitting processes and reduces the liability associated with chemical handling and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these specifics helps in evaluating the feasibility of adopting this route for specific project requirements.
Q: What are the key advantages of this Suzuki coupling method over traditional pyridine synthesis?
A: This method utilizes mild inorganic bases like sodium carbonate instead of harsh conditions, significantly improving reaction selectivity and reducing side products while maintaining high yields suitable for industrial scaling.
Q: Which catalyst and ligand systems are preferred for optimal yield in this process?
A: The patent specifies palladium acetate (Pd(OAc)2) as the preferred catalyst paired with triphenylphosphine (PPh3) as the ligand, providing a balance of cost-effectiveness and catalytic efficiency.
Q: Can this synthesis route be adapted for large-scale commercial production?
A: Yes, the one-step synthesis procedure operates under relatively mild temperatures (25°C to 80°C) and uses common solvents, making it highly amenable to commercial scale-up with simplified operational protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Pyridine Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at adapting the Suzuki coupling protocols described in CN103351333A to meet stringent purity specifications required by top-tier pharmaceutical clients. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch, ensuring that our aryl pyridine derivatives meet the highest industry standards. Our commitment to quality assurance means that every shipment is accompanied by comprehensive documentation, providing total transparency and traceability for your supply chain.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your development pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall project costs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality intermediates reliably. Let us be your partner in turning complex chemical challenges into commercial successes.
