Advanced Synthetic Route for 3-Aryl-4-Vinyl Oxazoline-2-Ketone Pharmaceutical Intermediates
Advanced Synthetic Route for 3-Aryl-4-Vinyl Oxazoline-2-Ketone Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust and scalable synthetic pathways for complex heterocyclic intermediates, particularly those serving as key scaffolds for drug discovery. Patent CN101429171B introduces a transformative methodology for the synthesis of 3-aryl-4-vinyl oxazoline-2-ketone, a valuable structural motif in medicinal chemistry. This innovation addresses critical bottlenecks in traditional manufacturing, such as the reliance on hazardous epoxides and multi-step sequences that plague conventional approaches. By leveraging simple butylene-1,2-diol as a starting feedstock, the disclosed process achieves high regioselectivity through strategic silyl protection, followed by efficient sulfonylation and a streamlined one-pot cyclization. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates while mitigating the risks associated with volatile raw material markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxazolinone core has relied heavily on the reaction between epichlorohydrin or substituted epoxides and aryl isocyanates. While conceptually straightforward, these legacy methods suffer from severe practical drawbacks that hinder commercial viability. The reaction conditions are often excessively harsh, requiring strict temperature control and specialized equipment to manage exotherms safely. Furthermore, the ring-opening of epoxides frequently leads to the formation of unwanted positional isomers, complicating downstream purification and drastically reducing overall yield. Alternative routes involving dibromo compound elimination are even more cumbersome, necessitating the preparation of unstable precursors through polystep reactions. These inefficiencies result in elevated production costs, extended lead times, and a larger environmental footprint due to excessive solvent and reagent consumption, making them less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a rational design centered on the differential reactivity of hydroxyl groups in butylene-1,2-diol. By selectively protecting the primary hydroxyl group with a silyl ether, the synthesis effectively masks one reactive site, directing subsequent functionalization exclusively to the secondary position. This strategy eliminates the formation of regioisomers at the source, ensuring a cleaner reaction profile. The subsequent conversion to a sulfonic acid ester creates an excellent leaving group, priming the molecule for the final cyclization step. This modular approach not only simplifies the operational workflow but also allows for the use of milder reagents and standard laboratory glassware. The versatility of the method is further enhanced by its compatibility with various silyl groups and solvents, providing process chemists with the flexibility to tune conditions for optimal performance.
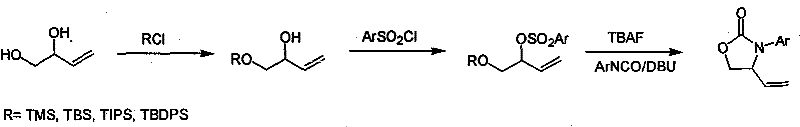
Mechanistic Insights into Silyl-Protection and DBU-Mediated Cyclization
The success of this synthetic route hinges on the precise control of steric and electronic factors during the initial protection phase. When butylene-1,2-diol is treated with silyl chlorides such as TBSCl or TMSCl in the presence of a base like imidazole, the less sterically hindered primary hydroxyl group reacts preferentially. This kinetic selectivity is crucial, as it generates the mono-protected intermediate with high fidelity, preventing the formation of di-silylated byproducts that would stall the synthesis. The choice of solvent, ranging from dichloromethane to tetrahydrofuran, plays a pivotal role in solubilizing the reagents while maintaining the necessary reaction kinetics at temperatures between 0°C and room temperature. This careful orchestration ensures that the intermediate retains the free secondary hydroxyl group required for the subsequent activation step, setting the stage for a high-yielding transformation.
The final cyclization step represents a masterpiece of base-mediated intramolecular nucleophilic substitution. Upon treatment with tetrabutylammonium fluoride (TBAF), the silyl protecting group is cleaved, regenerating the primary alcohol. In the presence of a strong non-nucleophilic base like DBU (1,8-diazabicyclo[5.4.0]undec-7-ene), the newly freed hydroxyl group attacks the carbonyl carbon of the aryl isocyanate. Simultaneously, the internal sulfonate leaving group is displaced, driving the formation of the five-membered oxazolinone ring. This tandem deprotection-cyclization sequence is highly efficient, minimizing the exposure of reactive intermediates to potential degradation pathways. The use of DBU is particularly advantageous as it facilitates the reaction under mild alkaline conditions, avoiding the need for strong acids or transition metal catalysts that could introduce difficult-to-remove impurities into the final API intermediate.
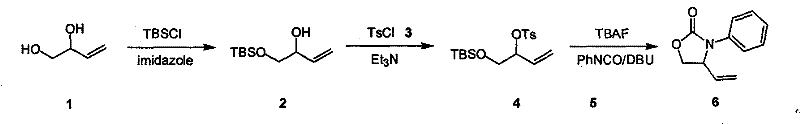
How to Synthesize 3-Aryl-4-Vinyl Oxazoline-2-Ketone Efficiently
Implementing this synthesis requires attention to detail regarding reagent stoichiometry and temperature management to maximize yield and purity. The process begins with the protection step, where strict control of the addition rate of silyl chloride is necessary to prevent local overheating and ensure selective mono-protection. Following isolation of the silyl ether, the sulfonylation is conducted under similar mild conditions, typically utilizing triethylamine as a scavenger for the generated acid. The final step combines deprotection and cyclization, where the sequential addition of TBAF, aryl isocyanate, and DBU must be carefully timed to drive the reaction to completion without side reactions. Detailed standardized operating procedures for each stage, including specific workup protocols involving aqueous washes and drying agents, are essential for reproducibility. For a comprehensive guide on executing these steps with precision, please refer to the technical protocol below.
- Selective protection of the primary hydroxyl group of butylene-1,2-diol using silyl chlorides (TMSCl, TBSCl, TIPSCl, or TBDPSCl) in solvents like DCM or THF.
- Reaction of the protected intermediate with aryl sulfonyl chloride (e.g., TsCl or p-chlorobenzenesulfonyl chloride) in the presence of a base like triethylamine.
- Deprotection of the silyl group using TBAF followed by immediate cyclization with aryl isocyanate under alkaline conditions using DBU to form the final oxazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers profound advantages that directly impact the bottom line and supply chain resilience. By shifting the starting material from specialized epoxides to commodity chemicals like butylene-1,2-diol, manufacturers can significantly reduce raw material procurement costs and mitigate supply risks. The elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, which are often a bottleneck in GMP manufacturing. Furthermore, the high atom economy and reduced number of unit operations translate to lower energy consumption and decreased solvent waste, aligning with increasingly stringent environmental regulations. These factors collectively contribute to a more sustainable and cost-effective production model that enhances competitiveness in the global marketplace.
- Cost Reduction in Manufacturing: The substitution of complex, multi-step precursor synthesis with a direct route from bulk diols drastically lowers the cost of goods sold. By avoiding the use of precious metal catalysts and reducing the total number of isolation steps, the process minimizes material loss and labor hours. The ability to use common solvents like dichloromethane and ethyl acetate further reduces operational expenses compared to specialized solvent systems required by older methods. Additionally, the high yields reported in the embodiments suggest that less starting material is wasted, leading to substantial cumulative savings over large production batches.
- Enhanced Supply Chain Reliability: Reliance on widely available feedstocks such as butylene glycol and standard reagents like TBSCl and TsCl ensures a stable supply chain不受 geopolitical or niche market fluctuations. Unlike proprietary catalysts or custom-synthesized epoxides that may have long lead times, the reagents for this process are commoditized and accessible from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, further securing consistent output.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of hazardous reagents make this process inherently safer and easier to scale from kilogram to tonne quantities. The simplified workup procedures, involving standard aqueous extractions and crystallization, are well-suited for existing infrastructure in multipurpose chemical plants. From an environmental standpoint, the reduction in waste generation and the avoidance of toxic heavy metals simplify wastewater treatment and disposal compliance. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. Understanding these details is crucial for project managers evaluating the feasibility of adopting this technology for their specific pipeline candidates. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-making processes.
Q: What are the advantages of using butylene-1,2-diol over epichlorohydrin for this synthesis?
A: Butylene-1,2-diol is a simple, easily obtained raw material that avoids the harsh reaction conditions and positional isomer issues associated with traditional epoxide-based routes, leading to higher purity and easier purification.
Q: Which silyl protecting groups are compatible with this synthetic route?
A: The process is highly versatile and supports various silyl protecting groups including TMS, TBS, TIPS, and TBDPS, allowing flexibility in optimizing reaction conditions and cost based on specific substrate requirements.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method utilizes common solvents like dichloromethane and THF, operates at mild temperatures (0°C to room temperature), and avoids complex catalytic systems, making it highly amenable to scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-4-Vinyl Oxazoline-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-aryl-4-vinyl oxazoline-2-ketone meets the highest international standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering consistent quality that supports your clinical and commercial manufacturing goals.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall project costs.
